-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariérní portál
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaMalaria Burden and Artemisinin Resistance in the Mobile and Migrant Population on the Thai–Myanmar Border, 1999–2011: An Observational Study
Background:
The Shoklo Malaria Research Unit has been working on the Thai–Myanmar border for 25 y providing early diagnosis and treatment (EDT) of malaria. Transmission of Plasmodium falciparum has declined, but resistance to artesunate has emerged. We expanded malaria activities through EDT and evaluated the impact over a 12-y period.Methods and Findings:
Between 1 October 1999 and 30 September 2011, the Shoklo Malaria Research Unit increased the number of cross-border (Myanmar side) health facilities from two to 11 and recorded the number of malaria consultations. Changes in malaria incidence were estimated from a cohort of pregnant women, and prevalence from cross-sectional surveys. In vivo and in vitro antimalarial drug efficacy were monitored. Over this period, the number of malaria cases detected increased initially, but then declined rapidly. In children under 5 y, the percentage of consultations due to malaria declined from 78% (95% CI 76–80) (1,048/1,344 consultations) to 7% (95% CI 6.2–7.1) (767/11,542 consultations), p<0.001. The ratio of P. falciparum/P. vivax declined from 1.4 (95% CI 1.3–1.4) to 0.7 (95% CI 0.7–0.8). The case fatality rate was low (39/75,126; 0.05% [95% CI 0.04–0.07]). The incidence of malaria declined from 1.1 to 0.1 episodes per pregnant women-year. The cumulative proportion of P. falciparum decreased significantly from 24.3% (95% CI 21.0–28.0) (143/588 pregnant women) to 3.4% (95% CI 2.8–4.3) (76/2,207 pregnant women), p<0.001. The in vivo efficacy of mefloquine-artesunate declined steadily, with a sharp drop in 2011 (day-42 PCR-adjusted cure rate 42% [95% CI 20–62]). The proportion of patients still slide positive for malaria at day 3 rose from 0% in 2000 to reach 28% (95% CI 13–45) (8/29 patients) in 2011.Conclusions:
Despite the emergence of resistance to artesunate in P. falciparum, the strategy of EDT with artemisinin-based combination treatments has been associated with a reduction in malaria in the migrant population living on the Thai–Myanmar border. Although limited by its observational nature, this study provides useful data on malaria burden in a strategically crucial geographical area. Alternative fixed combination treatments are needed urgently to replace the failing first-line regimen of mefloquine and artesunate.
Please see later in the article for the Editors' Summary
Published in the journal: . PLoS Med 10(3): e32767. doi:10.1371/journal.pmed.1001398
Category: Research Article
doi: https://doi.org/10.1371/journal.pmed.1001398Summary
Background:
The Shoklo Malaria Research Unit has been working on the Thai–Myanmar border for 25 y providing early diagnosis and treatment (EDT) of malaria. Transmission of Plasmodium falciparum has declined, but resistance to artesunate has emerged. We expanded malaria activities through EDT and evaluated the impact over a 12-y period.Methods and Findings:
Between 1 October 1999 and 30 September 2011, the Shoklo Malaria Research Unit increased the number of cross-border (Myanmar side) health facilities from two to 11 and recorded the number of malaria consultations. Changes in malaria incidence were estimated from a cohort of pregnant women, and prevalence from cross-sectional surveys. In vivo and in vitro antimalarial drug efficacy were monitored. Over this period, the number of malaria cases detected increased initially, but then declined rapidly. In children under 5 y, the percentage of consultations due to malaria declined from 78% (95% CI 76–80) (1,048/1,344 consultations) to 7% (95% CI 6.2–7.1) (767/11,542 consultations), p<0.001. The ratio of P. falciparum/P. vivax declined from 1.4 (95% CI 1.3–1.4) to 0.7 (95% CI 0.7–0.8). The case fatality rate was low (39/75,126; 0.05% [95% CI 0.04–0.07]). The incidence of malaria declined from 1.1 to 0.1 episodes per pregnant women-year. The cumulative proportion of P. falciparum decreased significantly from 24.3% (95% CI 21.0–28.0) (143/588 pregnant women) to 3.4% (95% CI 2.8–4.3) (76/2,207 pregnant women), p<0.001. The in vivo efficacy of mefloquine-artesunate declined steadily, with a sharp drop in 2011 (day-42 PCR-adjusted cure rate 42% [95% CI 20–62]). The proportion of patients still slide positive for malaria at day 3 rose from 0% in 2000 to reach 28% (95% CI 13–45) (8/29 patients) in 2011.Conclusions:
Despite the emergence of resistance to artesunate in P. falciparum, the strategy of EDT with artemisinin-based combination treatments has been associated with a reduction in malaria in the migrant population living on the Thai–Myanmar border. Although limited by its observational nature, this study provides useful data on malaria burden in a strategically crucial geographical area. Alternative fixed combination treatments are needed urgently to replace the failing first-line regimen of mefloquine and artesunate.
Please see later in the article for the Editors' SummaryIntroduction
The past decade has seen encouraging progress in the control of malaria worldwide. According to the World Health Organization's World Malaria Report 2011 [1], several countries in sub-Saharan Africa have registered very significant decreases in the number of Plasmodium falciparum malaria cases. In other continents, malaria has receded too, particularly in Southeast Asia, although the precise number of cases in some countries of the subcontinent may have been grossly underestimated [2]. Economic development, urbanisation, and unprecedented financial support have all contributed to these successes against malaria. Renewed efforts in vector control, insecticide-treated net distribution, and the deployment of artemisinin-based combination treatments (ACTs) have also contributed to the reduction of morbidity and mortality associated with P. falciparum, and this trend is particularly prominent in areas of low transmission such as in Southeast Asia.
These trends have boosted efforts towards malaria elimination. However, these gains are now threatened by the emergence in Southeast Asia of P. falciparum isolates that exhibit resistance to artesunate [3]. This resistance is characterised by a slow parasite clearance rate observed in patients treated with artesunate, but the precise mechanism remains unknown. Whether or not this particular form of resistance has spread beyond Southeast Asia is currently uncertain, but confirmed resistance surrounding the Thai–Myanmar border is attributable to changes in the parasite's genetic make-up [4]. It is not known whether the resistant parasites on the Thai–Myanmar border are related to those in Cambodia or whether resistance to artemisinin has emerged independently, but there is considerable concern that if this resistance to artemisinin spreads or emerges elsewhere because of widespread use, it will seriously compromise malaria control programs and threaten the lives of millions worldwide. During the previous decades, the spread of chloroquine - and sulphadoxine-pyrimethamine-resistant parasites from Southeast Asia to Africa resulted in a dramatic rise in mortality from increased perennial or epidemic transmission [5],[6]. Resistance to artemisinin could trigger a catastrophic resurgence in malaria in many parts of the world and compromise the progress made in the treatment of severe malaria [7],[8]. With substantial international support, containment efforts have started in Cambodia and bordering Thailand. The strategy is based on active detection of parasite carriers using molecular tools, but the impact is difficult to measure in the absence of a molecular marker of artemisinin resistance.
The largest focus of P. falciparum malaria in this region is situated in Myanmar, with a reported annual caseload of 70,941 in 2010 [1]. It is therefore essential to suppress malaria transmission as much as possible on the border between Myanmar and Thailand to avoid the spread of resistance to artemisinin to neighbouring countries and beyond. Between 1994 and 1999 we observed the impact of early detection and treatment (EDT) with ACT in populations of refugees living on the Thai–Myanmar border [9]. Ten years later we reported a similar impact in a larger population living in the region, including villagers, migrant workers, and refugees [10]. In the meantime, as per national policies, Thailand (in 1995), Vietnam (in 1995), Cambodia (in 2000), and Myanmar (in 2002) all adopted ACT as first-line therapy for P. falciparum infections. Malaria control in border areas is particularly challenging, especially if effective control measures are not deployed on both sides of the border. The combination of a constant reservoir of malaria in Myanmar, where the disease burden is higher than in Thailand, and frequent population movement mean that providing control measures based on EDT alone to migrants on the Myanmar side of the border might be expected to have a lower impact on local malaria transmission and incidence than in a more enclosed population, such as in large refugee camps or in villages on the Thai side of the border, where similar measures have been highly successful.
We evaluated malaria prevalence and incidence in the mobile and migrant population on the Myanmar side of the border between 1 October 1999 and 30 September 2011 to assess whether increasing access to EDT with ACT for this population was associated with a decline in the malaria burden.
Methods
Setting
The border between Thailand and Myanmar (Burma) is 2,107 km long and is mostly forested and mountainous. It is inhabited by a mosaic of ethnic groups and is characterised by intense migration fluxes between the two countries. Decades of internal conflicts in Myanmar have resulted in massive population displacements, and over 150,000 refugees now live in camps in Thailand. Economic stagnation has also prompted the migration of millions of people to Thailand in search of work, especially since the mid-1990s. This region is endemic for malaria (all four types, namely, P. falciparum, P. vivax, P. ovale, and P. malariae), and P. falciparum is highly drug resistant. The migration of gem miners from western Cambodia to Myanmar in the 1960s is thought to have played a major role in the spread of resistance to chloroquine [11]. The main vectors of malaria are the mosquitoes Anopheles minimus, A. dirus, and A. maculatus, and the transmission is low but unstable. The population has very little naturally acquired immunity against malaria, and all age groups are affected. In 1985, malaria was the main cause of consultations and mortality in the Karen refugee camps [12].
Population
The catchment area of the five Shoklo Malaria Research Unit (SMRU) clinics extends over 120 km of the border (see Figure 1). The population living along the border is diverse and is composed of three main groups: a relatively stable population living in established villages inside Myanmar for a number of years; a much more mobile group, mainly adult workers migrating according to work availability; and newly arrived people either displaced or in search of employment.
Fig. 1. Location of SMRU clinics and cross-border health posts run by village health workers. 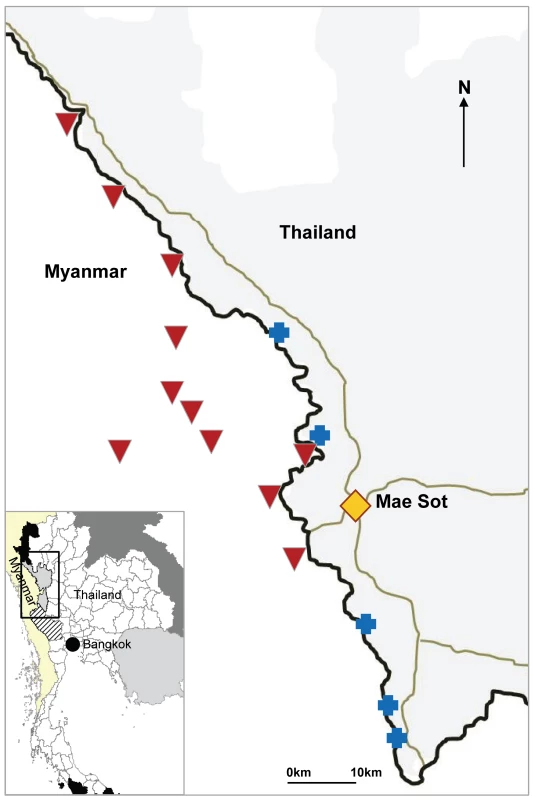
Blue crosses indicate SMRU clinics; inverted red triangles indicate SMRU health posts. The Thai–Myanmar border is represented by a black line, and the main roads by grey lines. Based on village population lists, the population living in villages on the Myanmar side has been estimated to be 42,000 people, with numbers updated during malariometric surveys and bed net distributions. This population size has grown steadily through the years. The ratio of men to women is estimated at 1, and ∼40% of the population is under 15 y of age, a population structure similar to that of the refugee camps. Villages in the catchment area are mainly located along the Moei river (the border between Myanmar and Thailand) but occasionally are as far as 30–40 km inside Myanmar.
The size of the mobile population is more difficult to assess; in 2004, the Thai Ministry of Interior estimated the number of registered migrants living in Tak province at ∼125,000. Another 50,000–100,000 migrants are thought to be living along the border without registration (data from the Tak public health services). The majority of those migrants are living in the three districts where SMRU clinics are located (namely, Mae Ramat, Mae Sot, and Phop Phra districts). In 2010, the Tak Industrial Federation estimated that 154,000 to 190,000 migrants (both registered and unregistered) were living in Tak province.
Expanding Cross-Border Access to EDT
Access to malaria diagnosis and treatment on the Myanmar side of the border is poor, because of travel limitations and insecurity. Health care is provided by local organisations, and coverage is limited. In general, patients with more severe illness seek treatment in Thailand. From 1999 to 2005, two “health posts” were run by locally selected health workers; they were trained by the SMRU staff to use rapid diagnostic tests (RDTs) and microscopy and to provide appropriate treatment following the protocols used in the SMRU clinics. After 2008, nine health posts were established on the Myanmar side but without microscopy. (see Figure 1). The health posts are operated and supported as follows. Severe cases are referred to the nearest medical facility. Antimalarial drugs (artemisinin-based combinations as well as chloroquine for the treatment of non-falciparum malaria) and antipyretics are supplied on a regular basis to avoid shortages or stock-outs. Supervision of the work is done routinely, and quality control (QC) of malaria diagnosis is performed at the SMRU laboratory in Mae Sot. Pregnant women seeking care at the health posts are encouraged to attend antenatal care at the nearest SMRU clinic on the Thai side. Insecticide-impregnated bed nets are distributed to all patients with confirmed malaria seen in SMRU clinics, as per the requirements of the clinic's funding agencies.
Study Design
In order to evaluate changes in the malaria burden since access to EDT was expanded, we recorded the number of consultations in all clinics with confirmed malaria diagnosis, changes in the prevalence of malaria in the populations on the Myanmar side of the border by means of cross-sectional surveys, and the incidence of malaria in a cohort of pregnant women living on both sides of the border but followed in clinics on the Thai side. Other factors that can affect malaria incidence were monitored, i.e., in vivo and in vitro antimalarial drug efficacy, bed net distribution, and climatic conditions. The observation period extended from 1 October 1999 to 30 September 2011.
Monitoring the Number of Malaria Cases
In the cross-border health posts (Myanmar side), malaria cases were detected by village health workers. Data were obtained weekly, and compiled on site by age group and malaria species. No gender distribution was available. All cases of P. falciparum reported were confirmed by either RDT (nine health posts) or microscopy (two health posts).
For each individual malaria case detected in the SMRU clinics (Thai side), demographic characteristics and type of malaria test and its result were recorded. Species were confirmed systematically by slide microscopy only for children under 5 y and pregnant women because some of the RDTs used can only detect P. falciparum. However, one week each month, all patients presenting at the SMRU clinics with malaria symptoms were tested by blood smear microscopy, allowing the calculation of changes in the P. falciparum/P. vivax ratio over time. The presence of gametocytes was reported routinely for each malaria smear done in the SMRU clinics. Hospitalisations due to severe malaria have been systematically recorded since 2003.
QC of the malaria diagnostic tests was implemented at all sites. All RDTs performed in the field by village health workers, mostly Paracheck-Pf, an HRP-II-based RDT detecting only P. falciparum, and Optimal-IT, a pLDH-based test detecting falciparum and non-falciparum species, were retained in zip-sealed plastic bags on site and reread later by a senior technician in Mae Sot. Although, this QC method has limitations, as RDT results do not always remain stable over time, it enables a check on the accuracy of data recording and can detect major errors in test result interpretation in the field that warrant further investigation or refresher training.
For SMRU clinics and health posts with a field laboratory performing microscopy, a standardised protocol for internal QC has been in place since 1997. Briefly, each field laboratory stores all slides in zip-sealed plastic bags, separating positive from negative slides. These are sampled several times a year and sent to the central laboratory in Mae Sot for cross-checking and quality assessment of the sensitivity, specificity, and negative and positive predictive values of the tests. Any discrepancies between results required a third and final reading by the head of the laboratory department.
Malaria Incidence: Follow-Up of the Cohort of Pregnant Women
Pregnant women attended weekly antenatal care clinics until delivery. In addition to routine antenatal care, a malaria smear was done at each consultation, whether the woman was symptomatic or not, and her haematocrit was measured every fortnight. Pregnant women were also encouraged to seek care immediately in case of fever or symptoms suggestive of malaria. Women found to be parasitaemic were treated for malaria, regardless of their symptoms.
Malariometric Cross-Sectional Surveys
Cross-sectional surveys in villages and migrant communities were conducted annually in order to detect changes in the transmission of P. falciparum and P. vivax. We surveyed one or two villages each year between 2000 and 2003; in 2006–2008, prior to expanding malaria control on the Myanmar side, we surveyed up to five villages annually. Most of the surveys were done during the rainy season, when malaria is at its peak. Exhaustive population surveys were done in villages with fewer than 500 inhabitants; for larger villages, 25% of the houses were randomly selected, and all residents in those houses were screened. Participation to the survey was voluntary. Each participant was given a unique identifier, was asked basic demographic information, and had a malaria smear performed. Anyone presenting with fever or a history of fever in the previous days also underwent an RDT for malaria, and if the test was positive, the individual was immediately treated. Malaria smears were considered negative if the technician did not find any parasites in 100 oil-immersion fields (1,000×) on a thick blood film. Results of the malaria smear screening were obtained within 24–48 h, and all people with a positive malaria smear result were treated.
In Vivo Efficacy of Mefloquine-Artesunate
We monitored the efficacy of mefloquine (8 mg/kg/d) and artesunate (4 mg/kg/d) over 3 d for the treatment of patients with uncomplicated P. falciparum malaria confirmed microscopically annually. After giving written consent, patients were followed daily until resolution of their parasitaemia and then weekly for 6 wk. Recrudescence between days 5 and 42 was differentiated from a new infection using parasite genotyping by PCR [13]. Patients with indeterminate genotyping results and new infections were censored at the date of the reappearance of P. falciparum parasites. Delayed parasite clearance was considered to be present if patients were still parasitaemic 72 h (day 3) after the baseline positive malaria smear.
In Vitro Antimalarial Drug Studies
Parasite isolates were obtained from non-pregnant patients with uncomplicated acute P. falciparum malaria primary infection attending the SMRU clinics if they had at least five parasites per 1,000 red blood cells and consented to give 5 ml of blood. Drug susceptibility was tested using the hypoxanthine uptake method. The reproducibility of IC50 measurements was assessed using cloned K1 isolates of P. falciparum.
Climate
Monthly climatic conditions (rainfall and mean temperature) were obtained from Mae Sot (central area) and Umphang (southern area) meteorological stations and from the Tha Song Yang health department (northern area). Annual trends were obtained by averaging monthly rainfall (in millimetres) and mean temperature (in degrees Celsius) from the different meteorological stations.
Statistical Analysis
Annual incidence rates in pregnancy for P. falciparum and for P. vivax were calculated separately. Only the first positive malaria smear of each species was considered and counted to calculate the number of new malaria episodes per pregnant woman. Women with negative malaria smears throughout their pregnancy were considered free of malaria; the period between the first antenatal care visit and the pregnancy outcome was considered the period “at risk” and was measured in weeks. In addition to incidence, the annual cumulative proportion of women who experienced at least one malaria episode during their pregnancy was calculated for both P. falciparum and P. vivax. Anaemia was reported as present if the haematocrit was <30% at least once during the pregnancy, and is presented as a proportion of the total number of women per year.
Proportions of positive cases found during malariometric surveys were stratified by plasmodial species, age, and gender, and were compared by chi-square test with Bonferroni correction.
Mefloquine-artesunate (MAS3) PCR-adjusted cure rates were estimated using Kaplan-Meier survival analysis. Patients who did not complete the 3-d treatment course or did not have at least 3 d of follow-up were excluded from the parasite clearance time analysis.
In vitro antimalarial dose–response curves were measured by a nonlinear regression function to determine the IC50 (expressed in nanograms/millilitre) using WinNonlin (version 4.1, Pharsight Corporation), and data were analysed using SPSS for Windows (version 15, SPSS). Temporal trends in geometric mean IC50 values of mefloquine, dihydroartemisinin, and artesunate were analysed (ANOVA method).
Results
Population
The changes in the estimated population figures are shown in Table 1. Although these are only estimates, they indicate that the mobile population living on the Thai side has remained stable (approximately 200,000), while the population living in established villages in the area covered by SMRU clinics and the health posts (including the settlements on the Myanmar side) has increased 4-fold.
Tab. 1. Population estimates between 2000 and 2011. 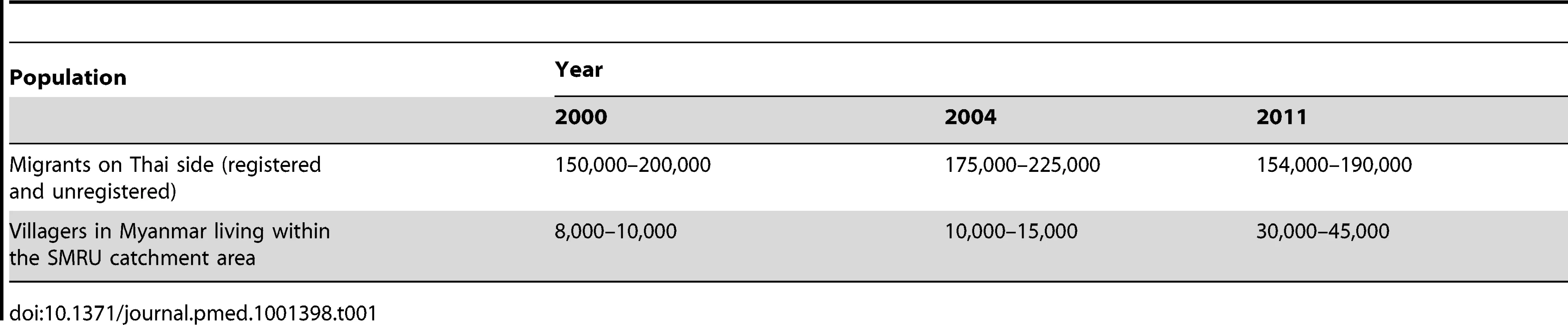
Number of P. falciparum Infections
The total number of P. falciparum infections we detected per year in SMRU clinics and in the village health posts is shown in Table 2. The number of cases detected increased initially with the opening of new facilities but thereafter gradually decreased, while the overall number of consultations (malaria-related or not) steadily increased from 25,345 to 69,000, reflecting the rise of the general population living in the area. Table 3 shows the distribution by age and gender of the P. falciparum cases detected in the SMRU clinics. The most striking finding is the predominance of infections (50,316/90,321; 55.7% [95% CI 55.4–56.0]) in young adult males, probably reflecting a higher exposure because of their occupations. The proportion of patients who presented with gametocytes increased significantly from 3.0% (95% CI 2.3–3.5) (149/5,010) in 2000 to 12.1% (95% CI 10.9–13.5) (296/2,446) in 2011 (chi-square test for trend, p<0.001) (Figure 2).
Fig. 2. Proportion of malaria smears with P. falciparum or P. vivax gametocytes. 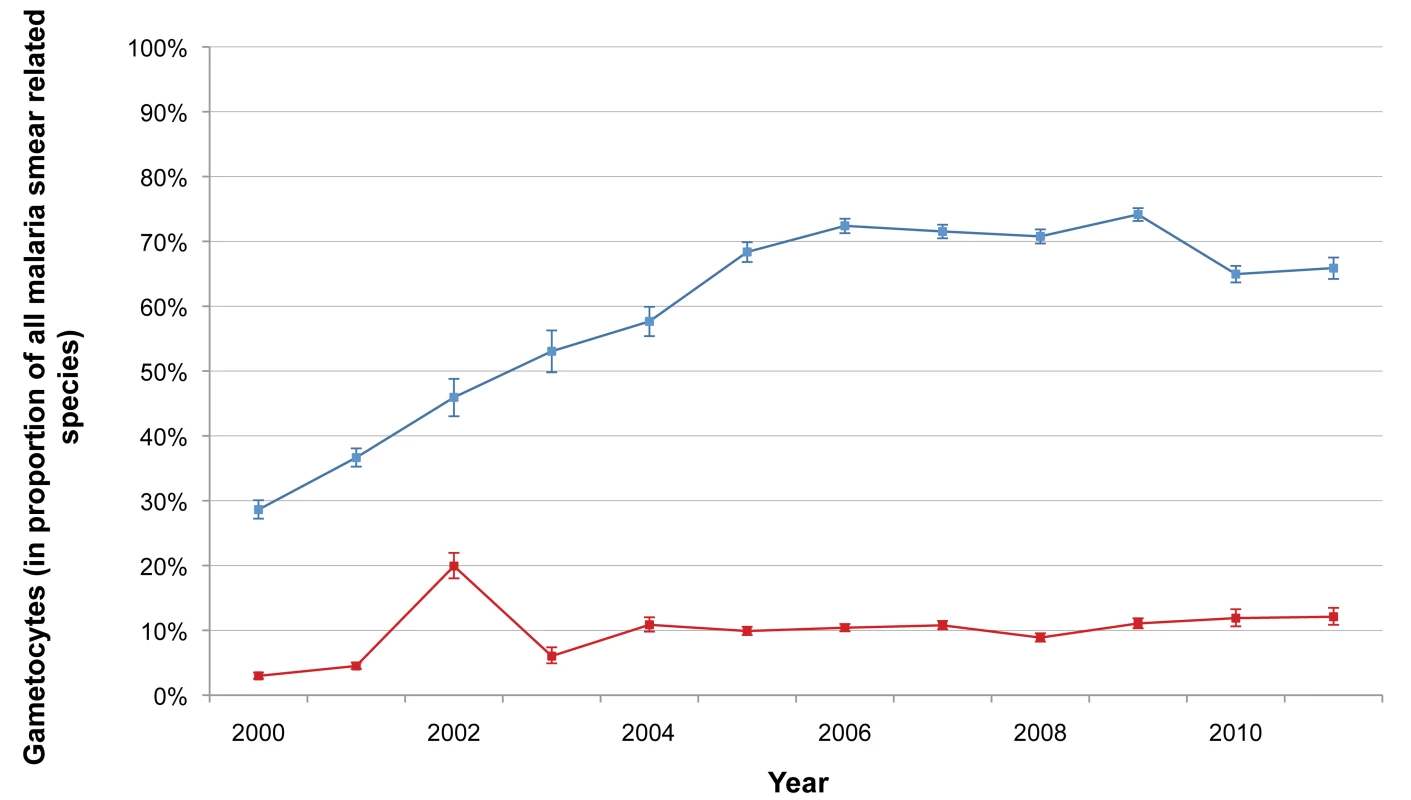
Annual proportion of all patients whose smears were positive for P. falciparum malaria who had P. falciparum gametocytes present on their malaria smear (red), and those whose smears were positive for P. vivax malaria who had P. vivax gametocytes present on their malaria smear (blue). Bars indicate 95% CIs. Tab. 2. Annual total number of consultations and number of P. falciparum infections detected in SMRU clinics and health posts. 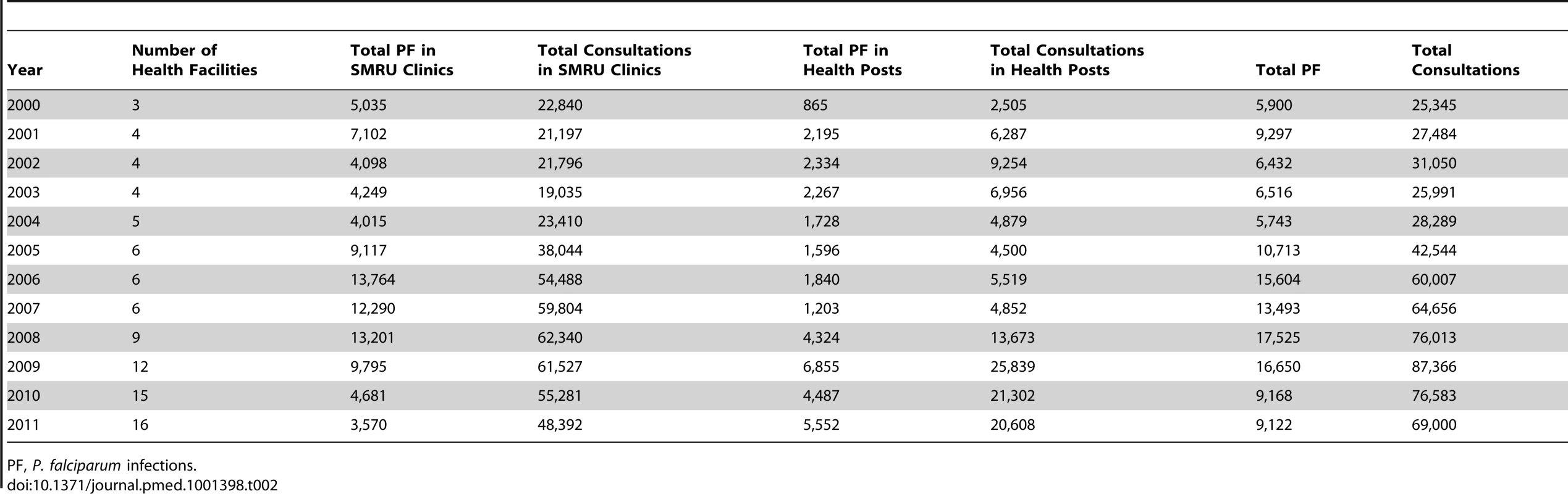
PF, P. falciparum infections. Tab. 3. Total P. falciparum malaria cases confirmed and treated in SMRU clinics by sex and age group (and as percentage of total annual number of P. falciparum cases). 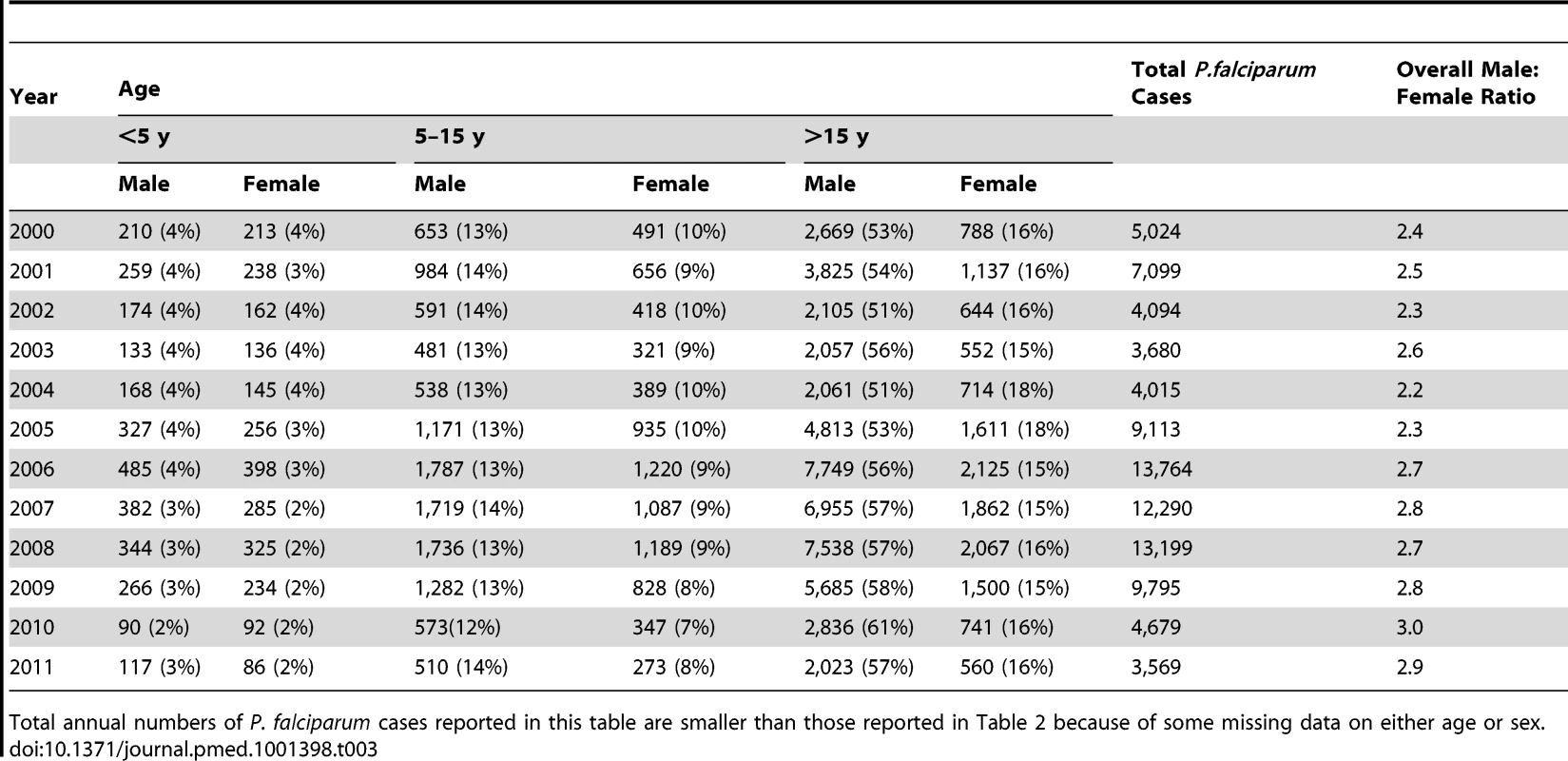
Total annual numbers of P. falciparum cases reported in this table are smaller than those reported in Table 2 because of some missing data on either age or sex. Number of P. vivax Infections
The annual numbers of P. vivax (and P. falciparum) infections in children under 5 y that presented at the SMRU clinics are shown in Table 4. The percentage of consultations due to malaria decreased over time (from 1,048/1,344, 78% [95% CI 76–80], to 767/11,542, 7% [95% CI 6.2–7.1]; chi-square test for trend, p<0.001).There was a significant decrease in the ratio of P. falciparum infections to P. vivax infections in the children during the study period, from 0.7 (95% CI 0.6–0.7) to 0.3 (95% CI 0.2–0.3) (chi-square test for trend, p<0.001). This finding was also evident (in all age groups) in the data compiled during the weeks when all patients presenting at SMRU clinics with malaria symptoms had a malaria smear, as shown in Table 5. The largest change was in the adult population, in which the ratio of P. falciparum infections to P. vivax infections decreased from 1.8 (95% CI 1.7–1.9) to 0.8 (95% CI 0.8–0.8) (chi-square test for trend, p<0.001). The proportion of patients who presented with P. vivax gametocytes steadily increased over the years to a plateau in 2006 (difference in proportion between 2000 and 2006 43.8% [95% CI 41.9–45.6]; chi-square test, p<0.001) (Figure 2).
Tab. 4. Total P. falciparum and P. vivax malaria cases confirmed by malaria smear among all children under 5 y old treated in SMRU clinics. 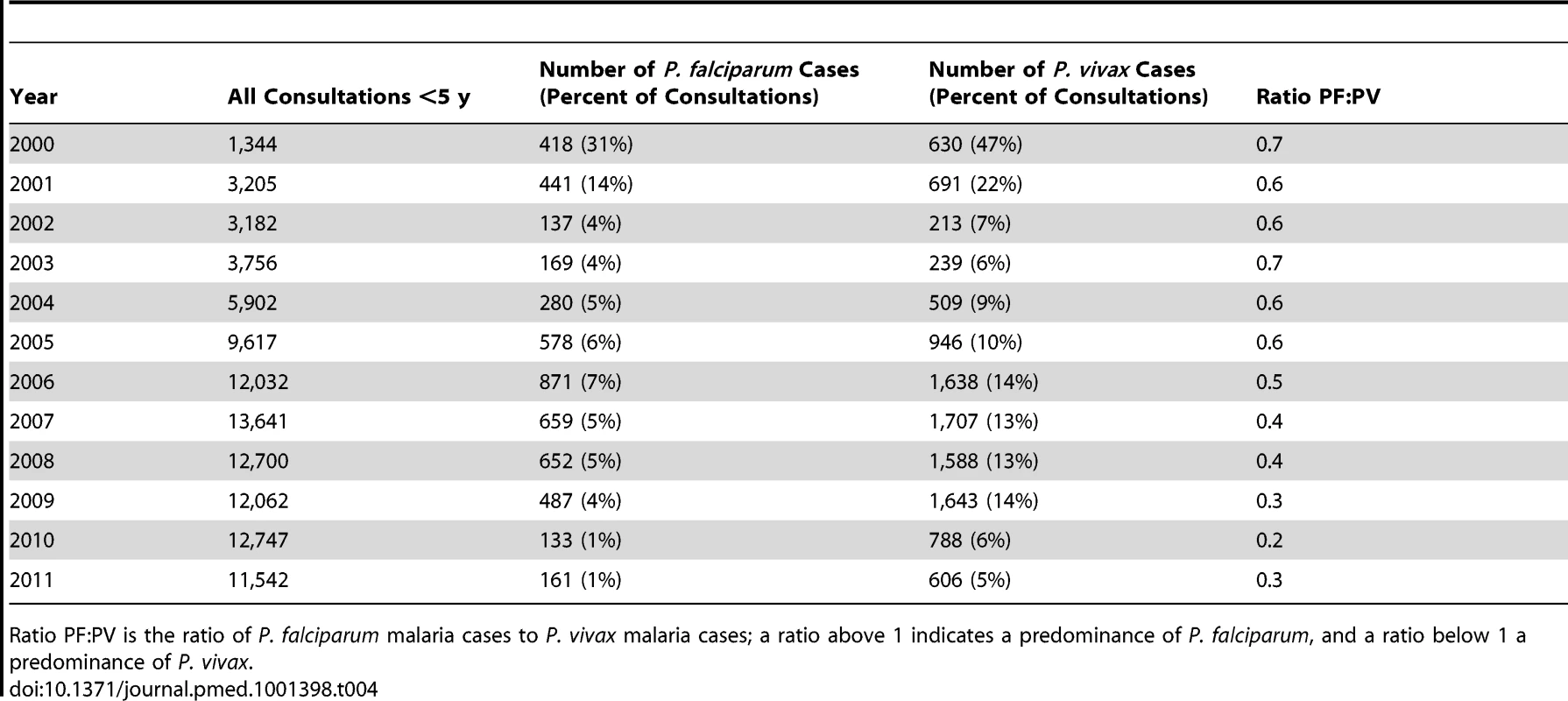
Ratio PF:PV is the ratio of P. falciparum malaria cases to P. vivax malaria cases; a ratio above 1 indicates a predominance of P. falciparum, and a ratio below 1 a predominance of P. vivax. Tab. 5. Ratio of P. falciparum to P. vivax malaria cases confirmed during weeks when all patients presenting to SMRU clinics with malaria symptoms had a malaria smear, by age group. 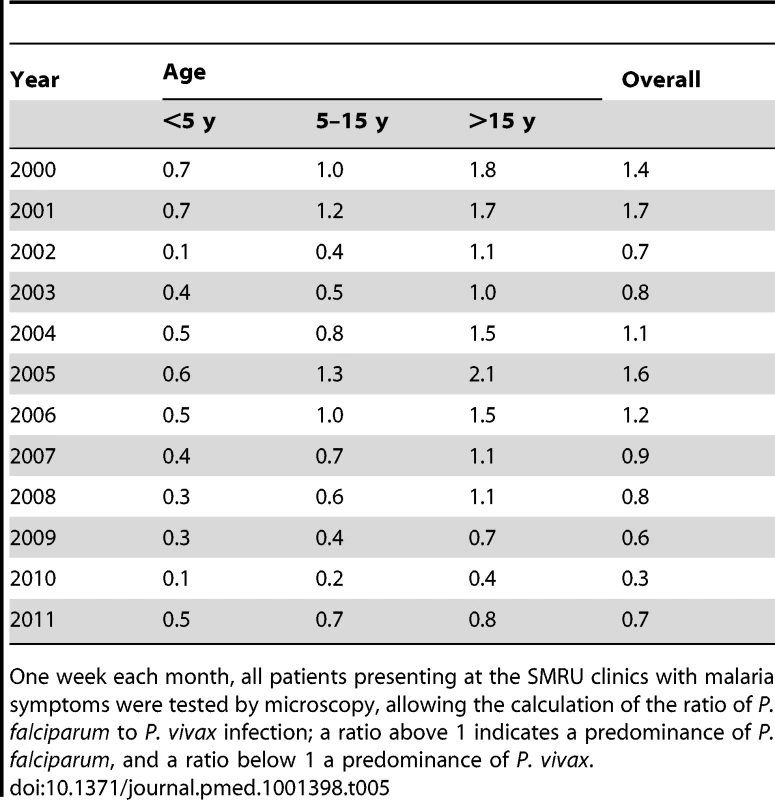
One week each month, all patients presenting at the SMRU clinics with malaria symptoms were tested by microscopy, allowing the calculation of the ratio of P. falciparum to P. vivax infection; a ratio above 1 indicates a predominance of P. falciparum, and a ratio below 1 a predominance of P. vivax. Hospitalised Cases and Mortality
Table 6 shows the number of patients hospitalised for malaria during the study period and the case fatality rate. The proportion of patients hospitalised for either uncomplicated hyperparasitaemia or severe disease was stable, 4.0% (95% CI 3.9–4.2) (3,022 hospitalisations/75,126 total patients with falciparum malaria). The number of deaths remained very low: the overall case fatality rate was 0.05% (95% CI 0.04–0.07), ranging across years from 0.01% (95% CI 0.00–0.06) to 0.11% (95% CI 0.05–0.25). Overall, only two patients (0.003% [95% CI 0.000–0.010] of 72,104 patients treated in ambulatory care in the outpatient department) died after receiving an oral artemisinin-based treatment for an uncomplicated P. falciparum infection. Forty-eight patients were hospitalised with a P. vivax infection and a concomitant condition between 2007 and 2011, but none were fatal or associated with signs of severe malaria.
Tab. 6. Hyperparasitaemia and severe malaria cases hospitalised in SMRU clinics, and case fatality rate among non-pregnant patients, from 2003 to 2011. 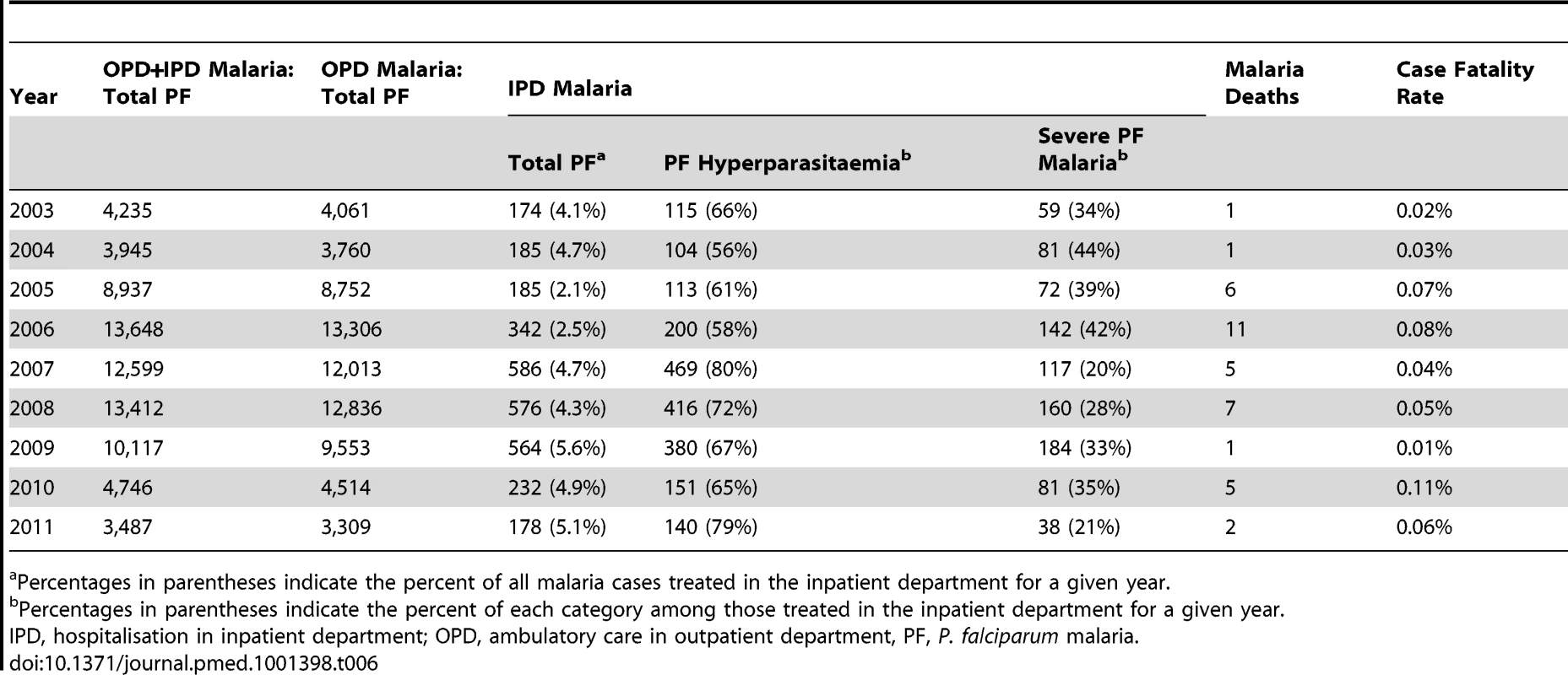
Percentages in parentheses indicate the percent of all malaria cases treated in the inpatient department for a given year. Laboratory Quality Control
Between 1 January 2000 and 31 December 2011 a total of 42,806 malaria smears were rechecked. This figure corresponds to an average of 3.6 unannounced rounds of QC per site per year. The mean sensitivity, specificity, positive predictive value, negative predictive value, and Kappa value for species differentiation were 95.7%, 99.8%, 99.7%, 98.0%, and 0.95, respectively. Out of 14,426 Paracheck-Pf tests that could be verified, 2.3% (95% CI 2.1–2.6) (335/14,426) had an interpretation error. Out of 9,569 Optimal-IT tests verified, 1.1% (95% CI 1.0–1.4) (109/9,569) were wrongly interpreted. However, delays in double-checking the RDTs may have affected the reliability of these results.
Malaria in Pregnancy
The incidence of P. falciparum and P. vivax infection in pregnant women is shown in Figure 3. There was a marked decline in the incidence of P. falciparum, from 1.09 to 0.14 infections·woman−1·year−1 (Student's t test, p<0.001), while P. vivax incidence declined only after 2003 (from 1.70 to 0.26 infections·woman−1·year−1; Student's t test, p<0.001). The cumulative proportion of women infected during pregnancy is illustrated in Figure 4 and clearly shows the decline in malaria exposure in this group from 24.3% (95% CI 21.0–28.0) (143/588) to 3.4% (95% CI 2.8–4.3) (76/2,207) for P. falciparum and from 24.0% (95% CI 20.7–27.6) (141/588) to 6.2% (95% CI 5.3–7.3) (137/2,207) for P. vivax, during the 12-y period. During the same period, the proportion of pregnant women with at least one haematocrit measurement below 30% also declined significantly (from 52.4% [95% CI 48.3–56.5] to 26.0% [95% CI 24.2–28.0]; chi-square test for trend, p<0.001) (Figure 5).
Fig. 3. P. falciparum and P. vivax malaria incidence among pregnant women. 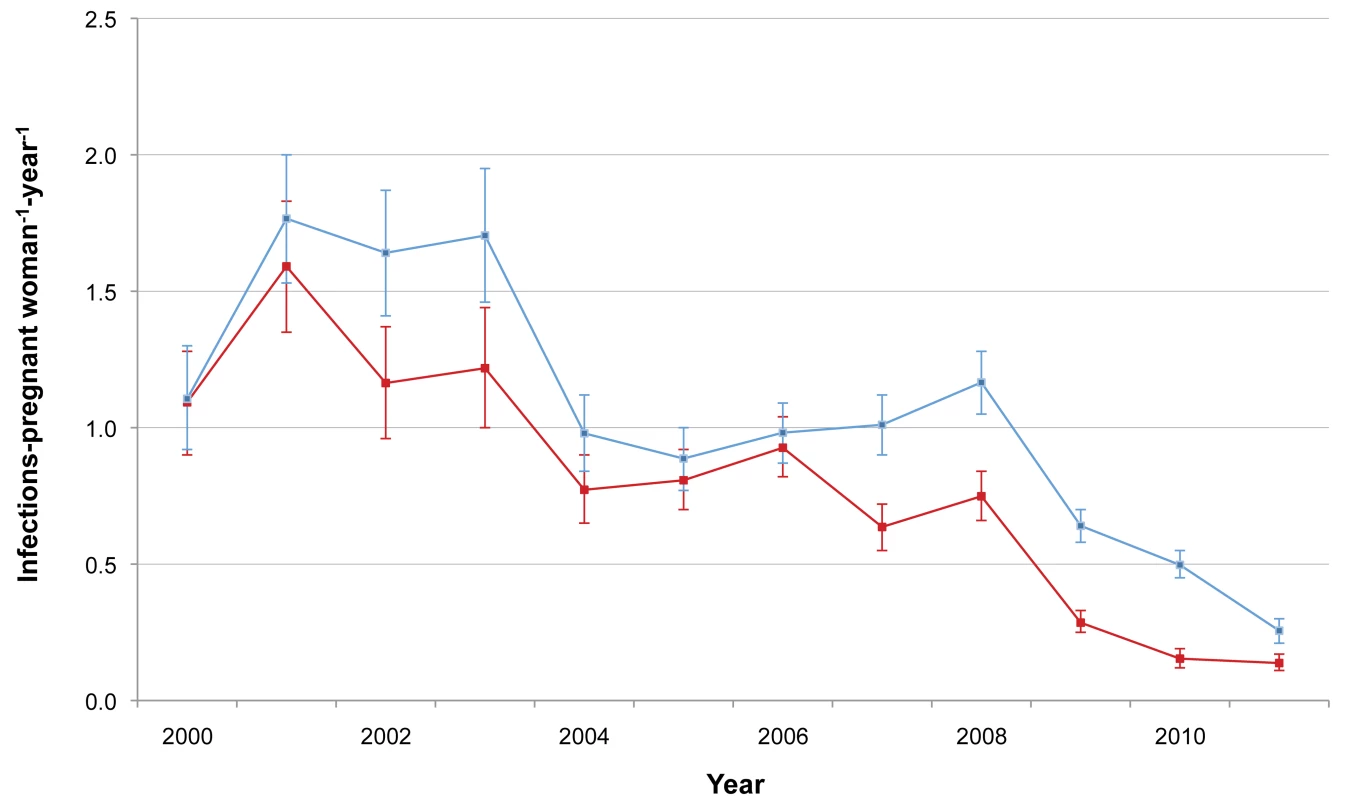
Red line indicates P. falciparum; blue line indicates P. vivax. Bars indicate 95% CIs. Fig. 4. Annual proportion of P. falciparum and P. vivax infections among pregnant women. 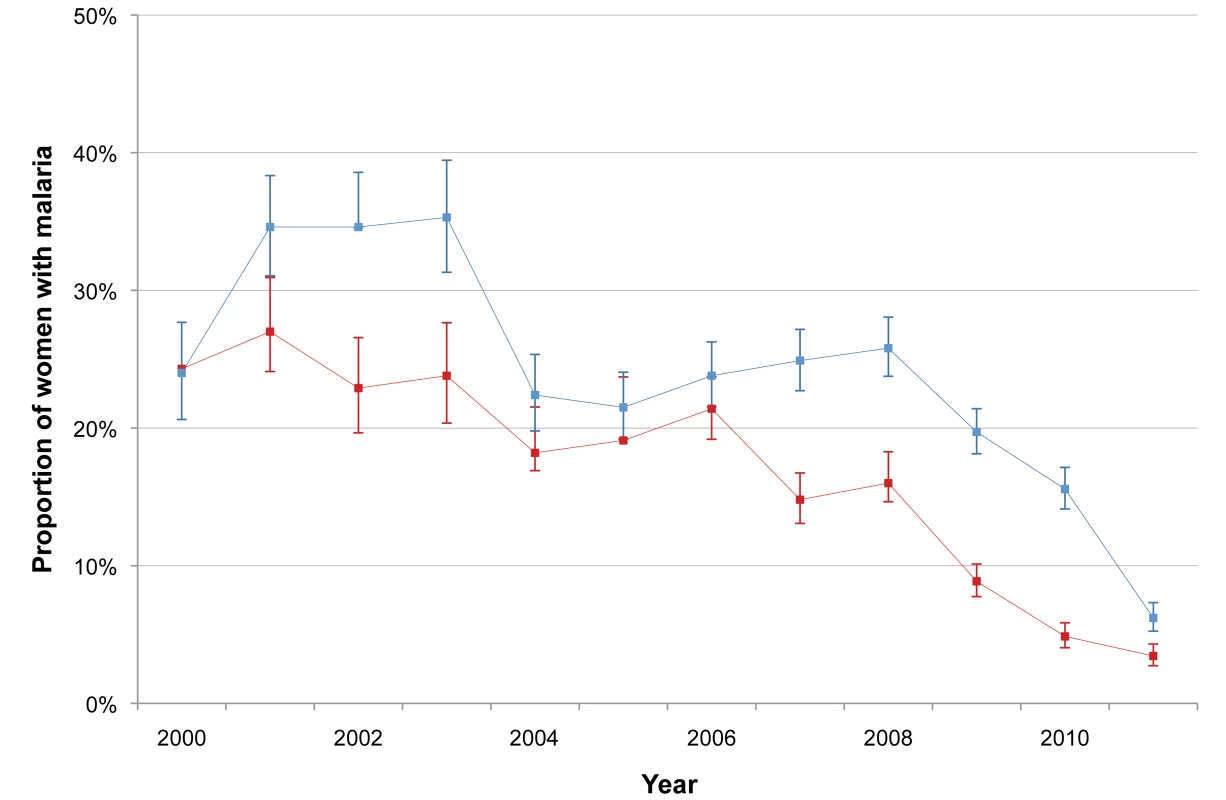
Red line indicates P. falciparum; blue line indicates P. vivax. Bars indicate 95% CIs. Fig. 5. Proportion of pregnant women with anaemia during their pregnancy. 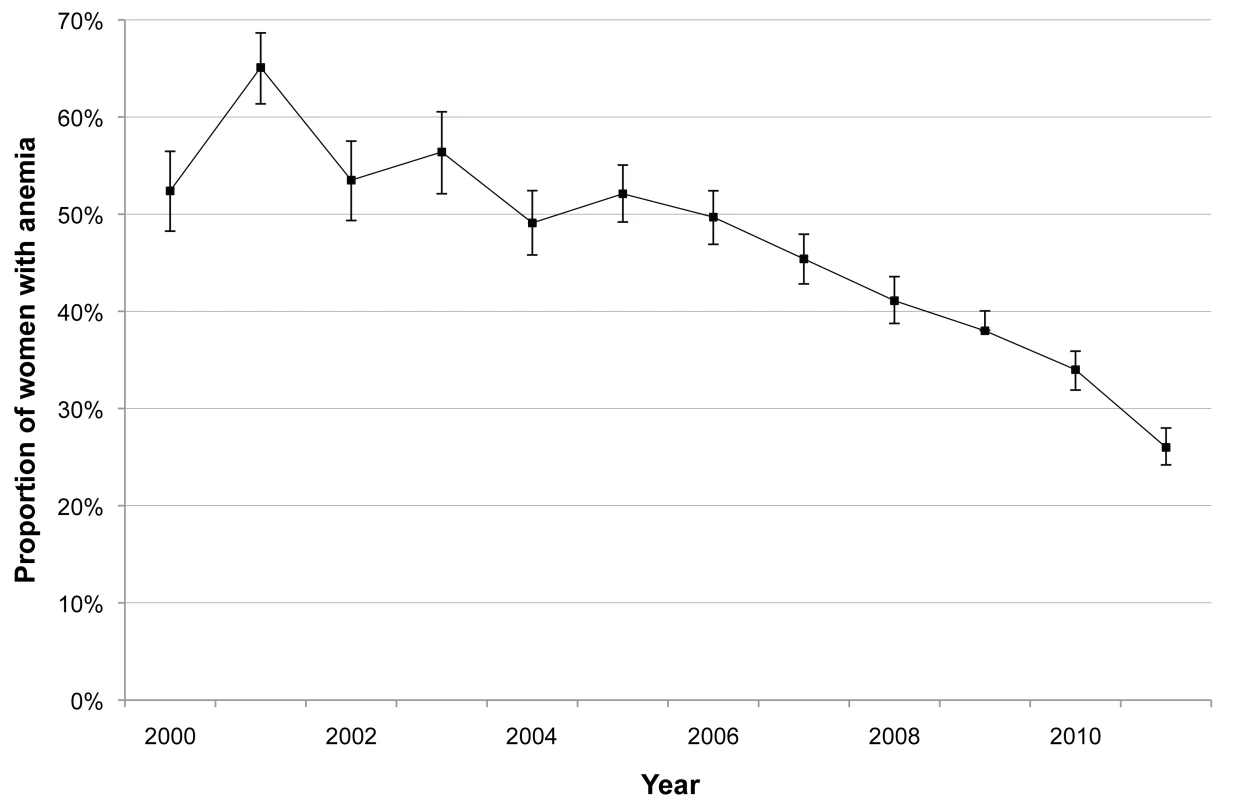
Anaemia was considered present if there was at least one haematocrit value<30% during the course of the pregnancy. Bars indicate 95% CIs. Cross-Sectional Malariometric Surveys
We conducted 24 cross-sectional surveys during the study period in 8,106 people from communities and villages on the Myanmar side of the border. The results are summarised in Table 7, and the ratio of P. falciparum to P. vivax infection is shown in Figure 6. P. falciparum was the predominant species during surveys conducted in Myanmar villages prior to 2003, with an annual prevalence ranging from 8.0% (95% CI 5.7–11.3) (29/361) to 24.6% (95% CI 20.7–28.9) (103/419). In 2006, P. falciparum prevalence decreased by 2/3 and continued to decrease until 2010, when 12/900 (1.3% [95% CI 0.8–2.3]) persons tested positive for P. falciparum. In contrast, P. vivax prevalence remained relatively constant over the years, with an averaged prevalence of 8.9% (95% CI 8.4–9.6) (728/8,106), and as of 2010 was the predominant species along the border area. As already observed among patients with confirmed malaria seeking care at the SMRU clinics, there was a predominance of P. falciparum infections in young adult males (41% [95% CI 36–46] of all P. falciparum infections found). P. vivax, by contrast, was predominant in the age group 5–15 y (12.8% [95% CI 11.6–14.2] [315/2,457] versus 6.5% [95% CI 5.4–7.9] [100/1,530] in age group <5 y and 7.6% (95% CI 6.8–8.5) (313/4,119) in age group >15 y; chi-square test, p<0.001).
Fig. 6. P. falciparum infection/P. vivax infection ratio from village surveys in Myanmar within SMRU clinic/health post catchment area. 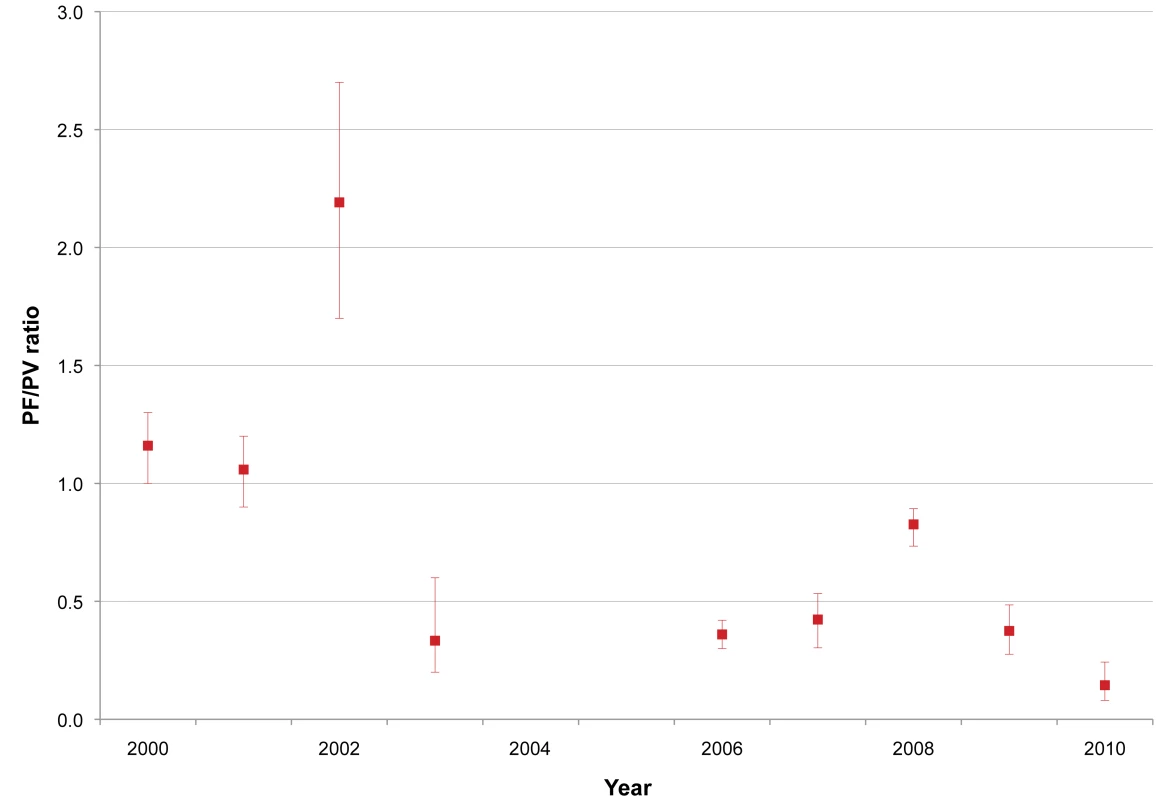
Red squares indicate the P. falciparum infection/P. vivax infection (PF/PV) ratio; bars indicate 95% CIs. A ratio above 1 indicates the predominance of P. falciparum over P. vivax; a ratio below 1 indicates the predominance of P. vivax. Tab. 7. Prevalence of P. falciparum and P. vivax infection and of gametocyte carriage during surveys of Myanmar villages within SMRU clinics and health posts catchment area. 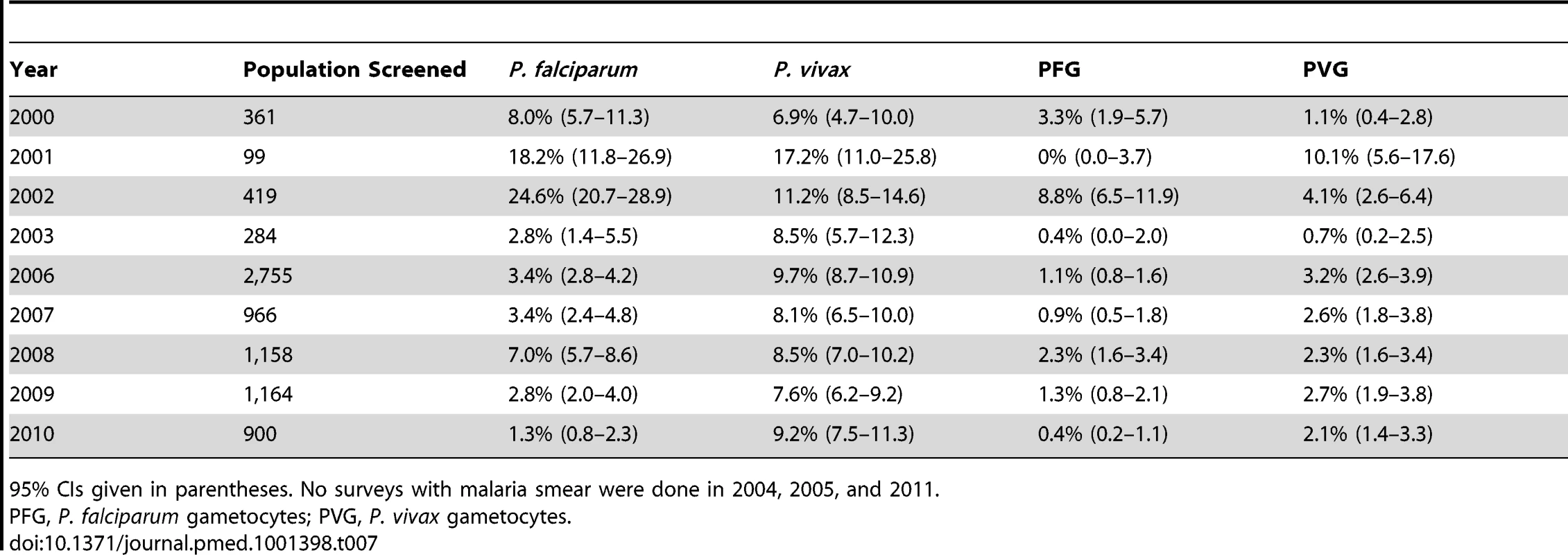
95% CIs given in parentheses. No surveys with malaria smear were done in 2004, 2005, and 2011. In Vivo Efficacy of Mefloquine-Artesunate
We enrolled 1,926 people in studies to determine the efficacy of MAS3 between 1 October 1999 and 30 September 2011; 75/1,926 people (3.9%) did not complete the 3-d treatment course or did not have at least 3 d of follow-up and were excluded from the parasite clearance time analysis, but they remained included for the cure rate analysis. The day-42 PCR-corrected cure rates and the proportion of patients still parasitaemic at day 3 are shown in Figure 7. There was a significant decline in the efficacy of MAS3 over the years (hazard ratio = 1.12 [95% CI 1.04–1.21], p<0.001) and a significant lengthening of the parasite clearance time (none of the 46 patients had a delayed [>3 d] clearance of parasitaemia in 2000, but 8 of 29 [28% (95% CI 15–46)] did in 2011; chi-square test for trend, p = 0.003).
Fig. 7. Day-42 PCR-adjusted MAS3 parasitological efficacy and proportion of patients still parasitaemic at day 3. 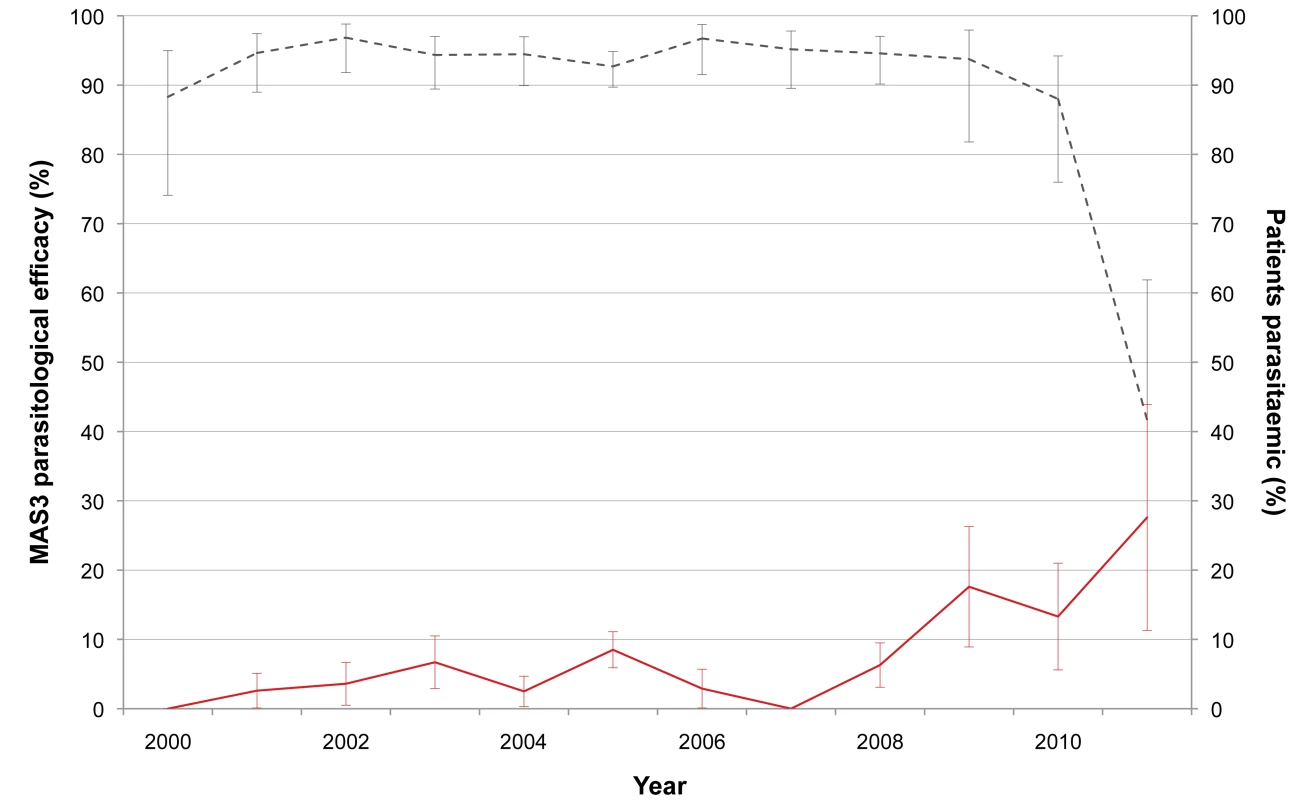
This graph shows the changes in day-42 PCR-adjusted MAS3 parasitological efficacy (black dashed line) and proportion of patients still parasitaemic at day 3 (red line) from 2000 to 2011. Bars indicate 95% CIs. In Vitro Sensitivity
The in vitro sensitivity (IC50) of P. falciparum isolates to mefloquine, artesunate, and dihydroartemisinin is shown in Figure 8. There were no specific trends over the study period.
Fig. 8. P. falciparum isolate in vitro sensitivity to mefloquine, artesunate, and di-hydroartemisinin from 2000 to 2010. 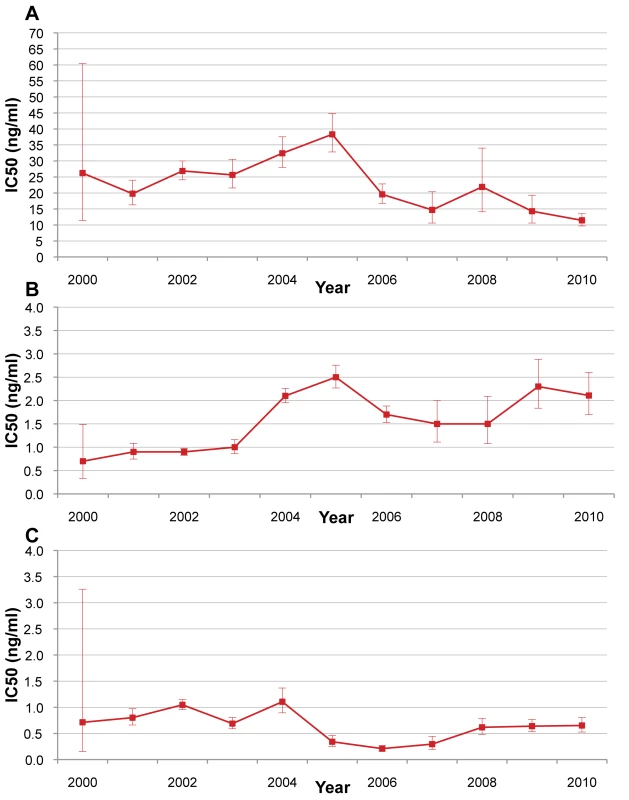
Red lines indicate in vitro sensitivity of P. falciparum isolates to (A) mefloquine, (B) artesunate, and (C) dihydroartemisinin. Bars indicate 95% CIs. No data were available for 2011. Climate
The annual rainfall and the mean annual temperature are shown in Figure 9. There was a steady increase in the mean annual temperature of 0.1°C between October 1999 and September 2011, but expected seasonal variations in temperature and rainfall. Over the study period, the mean annual rainfall ranged from 1,600 mm to 2,600 mm. The mean temperature ranged from 22.2°C in December to 28.3°C in April, and annual relative humidity was above 75%. The start of the rains was delayed in 2010, resulting in an overall reduction of annual mean rainfall by half. However, rainfall increased again in 2011.
Fig. 9. Annual rainfall and mean annual temperature between 2000 and 2011. 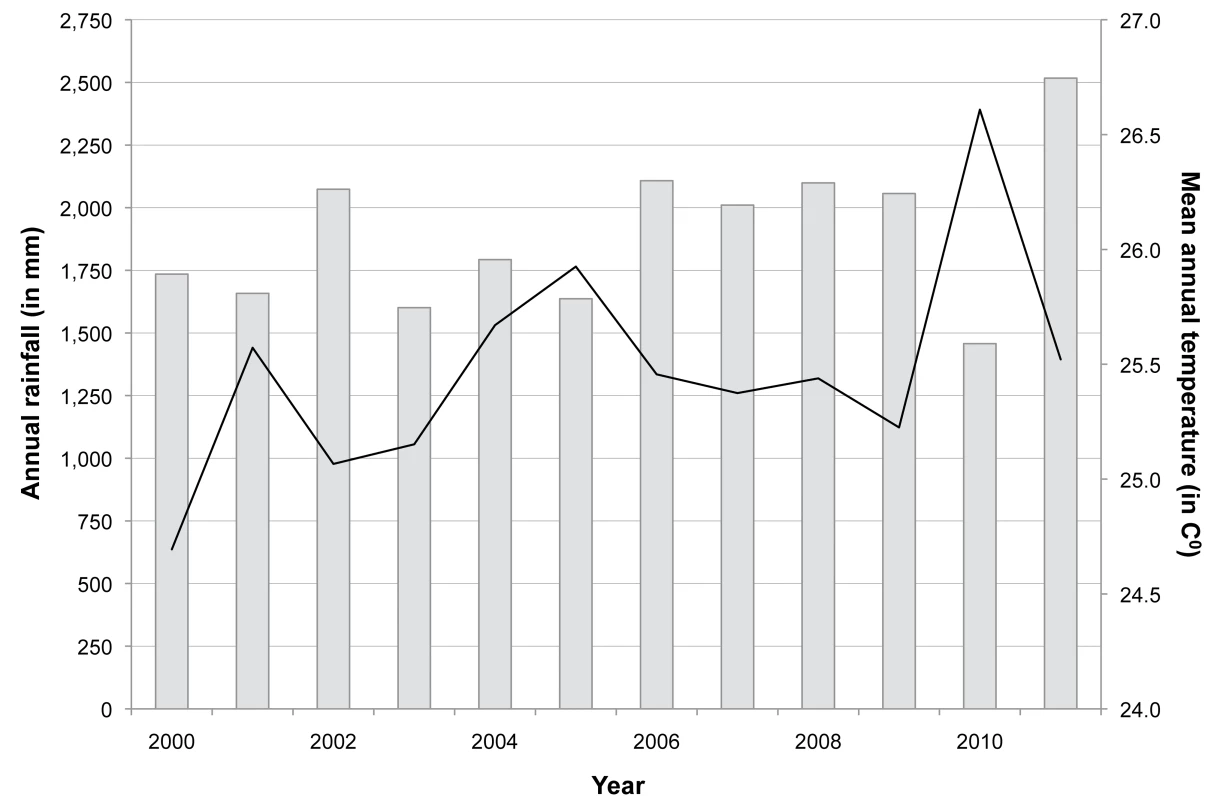
Black line indicates mean annual temperature in degrees Celsius, and grey bars indicate annual rainfall in millimetres. Discussion
Since 2000 the transmission of P. falciparum has declined substantially in the population in Myanmar living on the border with Thailand in Tak province (the most populated part of the border, with intense migration), despite being a well-known focus of highly drug-resistant P. falciparum malaria and an area of documented emergence of P. falciparum resistance to artesunate [4]. The decline is evident from the number of cases detected and treated in the SMRU clinics situated on the Thai side of the border. Longitudinal studies in a cohort of pregnant women confirm this decline. The decline has mainly been attributed to improved access to early detection of malaria and treatment with highly effective artemisinin-based antimalarial therapies.
This approach used by SMRU as well as other health providers in the area had proven effectiveness first in the refugee camps on the Thai border in the 1990s, then in Thai villages of Tak province and elsewhere in the region [14], and even in low transmission areas of Africa [15]. This effectiveness is explained by the rapid killing of sexual stages of P. falciparum and the activity of artemisinins on gametocytes [16].
Expanding EDT to the mobile and migrant population on the Myanmar side of the border also appears to have been successful in reducing malaria transmission, as evidenced by the decrease in the number of consultations for malaria and the results of the cross-sectional prevalence surveys. The financial investment was relatively modest, consisting mainly of drug and diagnostic test provision.
There are other factors that may have contributed to this reduction in the transmission of P. falciparum. There was a sharp decline in rainfall in 2010, and this may have contributed to lower vector abundance. However, the levels of precipitation in 2011 were higher than average, and this finding did not translate into a significant rise in caseload. In general, there was not much seasonal climatic variation from year to year over the period of study, so we do not believe that climate was a major confounding factor for changes in malaria prevalence or incidence observed over time (see detailed climate information in Figure S1). Over 46,000 insecticide-impregnated bed nets were also distributed between 2000 and 2011; however, the vectors in this area (A. minimus, A.dirus, and A. maculatus) are forest mosquitoes feeding early in the evening and outdoors, such that the impact of impregnated nets is limited [17]. These vector characteristics also explain the high proportion of cases in young adult males, because they are exposed while working in the forested areas. Vector control measures would also not explain the differences observed between P. falciparum and P. vivax case reductions. The latter species has become dominant, and the ratio of P. falciparum to P. vivax infections has declined in all areas where the patients had access to EDT (Figure 6). The slow decline in the number of P. vivax infections may be explained by the decline in P. falciparum infections, as many P. vivax parasitaemic episodes are relapses from previous infections, and P. falciparum infection is associated with P. vivax relapse [18]. So the reduction in the number of clinical episodes of P. falciparum may have contributed to lower numbers of P. vivax relapses [19].
Antimalarial drugs are the cornerstone of EDT, and this situation puts the parasite population under selection pressure for resistance. Indeed, we have shown that the proportion of patients with multiple copies of the gene Pfmdr1 (known to mediate resistance to mefloquine) is increasing, which may have contributed to the decline in the in vivo efficacy of the MAS3 combination recently reported, although the number of participants recruited in 2011 was very small (n = 32) [10]. If this sharp drop in efficacy is confirmed, an alternative ACT will have to be registered and deployed. The effectiveness of the current treatment for failures (7 d of quinine and tetracyclines) is poor. More worryingly, resistance to artemisinin is also emerging. This situation translates into a reduction of the parasite clearance rate following treatment with artesunate and may be explained by parasite genetics [20]. Unfortunately, resistance to artemisinin is not detected by conventional in vitro susceptibility testing methods, as shown in this study. The observation of increasing gametocyte carriage in patients presenting with P. falciparum infection is of great concern. The mechanisms that confer resistance to artemisinin are unknown, but it has been suggested that the use of substandard drugs and monotherapies may be contributing factors [3]. However, in the border population described here, self-treatment and monotherapies are uncommon. Alternatively, the use of quality drugs, free of charge, even in combination, may have selected parasite clones resistant to artesunate. This situation does not question the rationale behind using ACTs [16] but may be related to the phenomenon of dormancy [21]. When ring stages of P. falciparum are exposed to artesunate (or its metabolite, dihydroartemisinin), a small proportion becomes metabolically inactive and insensitive to drugs, and these are not removed by the spleen, unlike those affected by the drug [22] and unlike what is normally seen in patients [23],[24]. Whether the two observations (dormancy and the slow clearance phenotype) are related is unknown but cannot be excluded [25],[26]. To complicate matters further, P. vivax is becoming more resistant to chloroquine in this area [27], and this finding may explain the increasing proportion of patients presenting with vivax gametocytes (Figure 2). It may be necessary to adopt a unified ACT for both species, as in Papua province, Indonesia [28].
The observational nature of some of these data is an obvious limitation, and there are potential biases. The unstable nature of the population and the difficulties of working across an international border mean that the cases detected may not be representative of the overall population. Pregnant women, the group in whom we defined malaria incidence, may have a different risk of exposure and may not reflect the disease burden in the general population. However, it should be acknowledged that in most malarious regions (in particular in border areas), data are either nonexistent or much less detailed than in this study. We cannot eliminate all sources of bias, but the decline in P. falciparum cases in all age groups and both genders remains robust.
The benefit of increased access to EDT for the population is obvious, as illustrated by the decrease in maternal malaria-related anaemia. Efforts are underway to try to contain the spread of artemisinin resistance from western Cambodia. Our results and results from elsewhere indicate that an aggressive strategy based on EDT of cases, combined with vector control and information to the population, is the way forward to eliminate malaria. However, it is uncertain whether this strategy alone could achieve complete elimination over time. Adjunctive approaches should be evaluated, such as the use of low-dose primaquine to reduce transmission further, mass drug administrations, more sensitive detection of sub-microscopic infections using molecular methods, or targeted use of the new malaria vaccine, if it is approved. Addition of all these interventions may not be cost-effective, and so this approach should also be assessed.
Considerable support and strong leadership are urgently needed to step up the malaria control program in Myanmar and elsewhere. In practice, this means increasing the population coverage and training of village health workers in the use of RDTs and ACTs plus primaquine. Eliminating P. falciparum or dramatically reducing the number of cases is feasible, but the main obstacle is the difficulty of accessing populations living in remote, sometimes dangerous areas and across international borders.
Supporting Information
Zdroje
1. World Health Organization (2011) World malaria report 2011. Geneva: World Health Organization.
2. HaySI, GuerraCA, TatemAJ, NoorAM, SnowRW (2004) The global distribution and population at risk of malaria: past, present, and future. Lancet Infect Dis 4 : 327–336.
3. DondorpAM, NostenF, YiP, DasD, PhyoAP, et al. (2009) Artemisinin resistance in Plasmodium falciparum malaria. N Engl J Med 361 : 455–467.
4. PhyoAP, NkhomaS, StepniewskaK, AshleyEA, NairS, et al. (2012) Emergence of artemisinin-resistant malaria on the western border of Thailand: a longitudinal study. Lancet 379 : 1960–1966.
5. KorenrompEL, WilliamsBG, GouwsE, DyeC, SnowRW (2003) Measurement of trends in childhood malaria mortality in Africa: an assessment of progress toward targets based on verbal autopsy. Lancet Infect Dis 3 : 349–358.
6. MarshK (1998) Malaria disaster in Africa. Lancet 352 : 924.
7. DondorpA, NostenF, StepniewskaK, DayN, WhiteN (2005) Artesunate versus quinine for treatment of severe falciparum malaria: a randomised trial. Lancet 366 : 717–725.
8. DondorpAM, FanelloCI, HendriksenIC, GomesE, SeniA, et al. (2010) Artesunate versus quinine in the treatment of severe falciparum malaria in African children (AQUAMAT): an open-label, randomised trial. Lancet 376 : 1647–1657.
9. NostenF, van VugtM, PriceR, LuxemburgerC, ThwayKL, et al. (2000) Effects of artesunate-mefloquine combination on incidence of Plasmodium falciparum malaria and mefloquine resistance in western Thailand: a prospective study. Lancet 356 : 297–302.
10. CarraraVI, SirilakS, ThonglairuamJ, RojanawatsirivetC, ProuxS, et al. (2006) Deployment of early diagnosis and mefloquine-artesunate treatment of falciparum malaria in Thailand: the Tak Malaria Initiative. PLoS Med 3: e183 doi:10.1371/journal.pmed.0030183.
11. VerdragerJ (1986) Epidemiology of the emergence and spread of drug-resistant falciparum malaria in South-East Asia and Australasia. J Trop Med Hyg 89 : 277–289.
12. DecludtB, PecoulB, BibersonP, LangR, ImivithayaS (1991) Malaria surveillance among the displaced Karen population in Thailand April 1984 to February 1989, Mae Sot, Thailand. Southeast Asian J Trop Med Public Health 22 : 504–508.
13. BrockmanA, PaulRE, AndersonTJ, HackfordI, PhaiphunL, et al. (1999) Application of genetic markers to the identification of recrudescent Plasmodium falciparum infections on the northwestern border of Thailand. Am J Trop Med Hyg 60 : 14–21.
14. DelacolletteC, D'SouzaC, ChristophelE, ThimasarnK, AbdurR, et al. (2009) Malaria trends and challenges in the Greater Mekong Subregion. Southeast Asian J Trop Med Public Health 40 : 674–691.
15. BarnesKI, DurrheimDN, LittleF, JacksonA, MehtaU, et al. (2005) Effect of artemether-lumefantrine policy and improved vector control on malaria burden in KwaZulu-Natal, South Africa. PLoS Med 2: e330 doi:10.1371/journal.pmed.0020330.
16. WhiteNJ (1999) Delaying antimalarial drug resistance with combination chemotherapy. Parassitologia 41 : 301–308.
17. DolanG, ter KuileF, JacoutotV, WhiteNJ, LuxemburgerC, et al. (1993) Bed nets for the prevention of malaria and anaemia in pregnancy. Trans R Soc Trop Med Hyg 87 : 620–626.
18. LooareesuwanS, WhiteNJ, ChittamasS, BunnagD, HarinasutaT (1987) High rate of Plasmodium vivax relapse following treatment of falciparum malaria in Thailand. Lancet 2 : 1052–1055.
19. WhiteN (2011) Determinants of relapse periodicity in Plasmodium vivax malaria. Malar J 10 : 297.
20. CheesemanIH, MillerBA, NairS, NkhomaS, TanA, et al. (2012) A major genome region underlying artemisinin resistance in malaria. Science 336 : 79–82.
21. Kyle DE, Webster HK (1996) Postantibiotic effect of quinine and dihydroartemisinin derivatives on Plasmodium falciparum in vitro: implications for a mechanism of recrudescence [abstract]. XIVth International Congress for Tropical Medicine and Malaria; 17–22 November 1996; Nagasaki, Japan.
22. TeuscherF, GattonML, ChenN, PetersJ, KyleDE, et al. (2010) Artemisinin-induced dormancy in plasmodium falciparum: duration, recovery rates, and implications in treatment failure. J Infect Dis 202 : 1362–1368.
23. SaralambaS, Pan-NgumW, MaudeRJ, LeeSJ, TarningJ, et al. (2011) Intrahost modeling of artemisinin resistance in Plasmodium falciparum. Proc Natl Acad Sci U S A 108 : 397–402.
24. MokS, ImwongM, MackinnonM, SimJ, RamaodossR, et al. (2011) Artemisinin resistance in Plasmodium falciparum is associated with an altered temporal pattern of transcription. BMC Genomics 12 : 391.
25. NostenF (2010) Waking the sleeping beauty. J Infect Dis 202 : 1300–1301.
26. ChengQ, KyleDE, GattonML (2012) Artemisinin resistance in Plasmodium falciparum: a process linked to dormancy? Int J Parasitol Drugs Drug Resist 2 : 249–255.
27. PhyoAP, LwinKM, PriceRN, AshleyEA, RussellB, et al. (2011) Dihydroartemisinin-piperaquine versus chloroquine in the treatment of Plasmodium vivax malaria in Thailand: a randomized controlled trial. Clin Infect Dis 53 : 977–984.
28. DouglasNM, AnsteyNM, AngusBJ, NostenF, PriceRN (2010) Artemisinin combination therapy for vivax malaria. Lancet Infect Dis 10 : 405–416.
Štítky
Interní lékařství
Článek vyšel v časopisePLOS Medicine
Nejčtenější tento týden
2013 Číslo 3- Červená fermentovaná rýže účinně snižuje hladinu LDL cholesterolu jako vhodná alternativa ke statinové terapii
- Berberin: přírodní hypolipidemikum se slibnými výsledky
- Příznivý vliv Armolipidu Plus na hladinu cholesterolu a zánětlivé parametry u pacientů s chronickým subklinickým zánětem
- Alternativní léčebné možnosti u hypercholesterolemie při intoleranci statinů
- Vliv kombinace nutraceutik na remodelaci levé komory srdeční u osob s metabolickým syndromem
-
Všechny články tohoto čísla
- Malaria Burden and Artemisinin Resistance in the Mobile and Migrant Population on the Thai–Myanmar Border, 1999–2011: An Observational Study
- Getting Closer to a Fully Correctable and Connected Research Literature
- Surveillance Programme of IN-patients and Epidemiology (SPINE): Implementation of an Electronic Data Collection Tool within a Large Hospital in Malawi
- Using Routine Surveillance Data to Estimate the Epidemic Potential of Emerging Zoonoses: Application to the Emergence of US Swine Origin Influenza A H3N2v Virus
- Adapting Standards: Ethical Oversight of Participant-Led Health Research
- Strengthening the Expanded Programme on Immunization in Africa: Looking beyond 2015
- Changing Patterns in Place of Cancer Death in England: A Population-Based Study
- Policies and Initiatives Aimed at Addressing Research Misconduct in High-Income Countries
- Young Children's Probability of Dying Before and After Their Mother's Death: A Rural South African Population-Based Surveillance Study
- Research Misconduct in Low- and Middle-Income Countries
- Dissemination of Research Findings to Research Participants Living with HIV in Rural Uganda: Challenges and Rewards
- The Cost and Impact of Scaling Up Pre-exposure Prophylaxis for HIV Prevention: A Systematic Review of Cost-Effectiveness Modelling Studies
- Adjunctive Atypical Antipsychotic Treatment for Major Depressive Disorder: A Meta-Analysis of Depression, Quality of Life, and Safety Outcomes
- Number of Patients Studied Prior to Approval of New Medicines: A Database Analysis
- PLOS Medicine
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Surveillance Programme of IN-patients and Epidemiology (SPINE): Implementation of an Electronic Data Collection Tool within a Large Hospital in Malawi
- Adjunctive Atypical Antipsychotic Treatment for Major Depressive Disorder: A Meta-Analysis of Depression, Quality of Life, and Safety Outcomes
- Strengthening the Expanded Programme on Immunization in Africa: Looking beyond 2015
- The Cost and Impact of Scaling Up Pre-exposure Prophylaxis for HIV Prevention: A Systematic Review of Cost-Effectiveness Modelling Studies
Přihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



