-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaThe Rsb Phosphoregulatory Network Controls Availability of the Primary Sigma Factor in and Influences the Kinetics of Growth and Development
Chlamydia trachomatis is the leading cause of both bacterial sexually transmitted infection and infection-derived blindness world-wide. No vaccine has proven protective to date in humans. C. trachomatis only replicates from inside a host cell, and has evolved to acquire a variety of nutrients directly from its host. However, a typical human immune response will normally limit the availability of a variety of essential nutrients. Thus, it is thought that the success of C. trachomatis as a human pathogen may lie in its ability to survive these immunological stress situations by slowing growth and development until conditions in the cell have improved. This mode of growth is known as persistence and how C. trachomatis senses stress and responds in this manner is an important area of research. Our report characterizes a complete signaling module, the Rsb network, that is capable of controlling the growth rate or infectivity of Chlamydia. By manipulating the levels of different pathway components, we were able to accelerate and restrict the growth and development of this pathogen. Our results suggest a mechanism by which Chlamydia can tailor its growth rate to the conditions within the host cell. The disruption of this pathway could generate a strain incapable of surviving a typical human immune response and would represent an attractive candidate as an attenuated growth vaccine.
Published in the journal: . PLoS Pathog 11(8): e32767. doi:10.1371/journal.ppat.1005125
Category: Research Article
doi: https://doi.org/10.1371/journal.ppat.1005125Summary
Chlamydia trachomatis is the leading cause of both bacterial sexually transmitted infection and infection-derived blindness world-wide. No vaccine has proven protective to date in humans. C. trachomatis only replicates from inside a host cell, and has evolved to acquire a variety of nutrients directly from its host. However, a typical human immune response will normally limit the availability of a variety of essential nutrients. Thus, it is thought that the success of C. trachomatis as a human pathogen may lie in its ability to survive these immunological stress situations by slowing growth and development until conditions in the cell have improved. This mode of growth is known as persistence and how C. trachomatis senses stress and responds in this manner is an important area of research. Our report characterizes a complete signaling module, the Rsb network, that is capable of controlling the growth rate or infectivity of Chlamydia. By manipulating the levels of different pathway components, we were able to accelerate and restrict the growth and development of this pathogen. Our results suggest a mechanism by which Chlamydia can tailor its growth rate to the conditions within the host cell. The disruption of this pathway could generate a strain incapable of surviving a typical human immune response and would represent an attractive candidate as an attenuated growth vaccine.
Introduction
Chlamydia trachomatis is the leading cause of bacterial sexually transmitted infection worldwide [1], as well as the leading cause of infection-associated blindness [2]. Members of the Chlamydiaceae are obligate intracellular parasites that must complete a unique intracellular development cycle in order to propagate. This cycle is characterized by phenotypic variation between an Elementary Body (EB) that is infectious but metabolically quiescent, and a Reticulate Body (RB) that is replicative but not infectious. Cellular infection is initiated by the EB, which attaches and induces its own intake via translocation of effector cargo. Once inside the cell, the EB differentiates into an RB, which replicates via binary fission. During the late stages of infection, RBs asynchronously re-differentiate into the EB form (reviewed in [3]). Alternatively, certain stress conditions mediate the onset of a separate growth mode (termed persistence), which was defined as a ‘viable but non-cultivable’ state, in which chlamydiae fail to complete the development cycle and instead differentiate into aberrant, enlarged particles; this phenotype is reversible upon abatement of the mediating stress [4]. While modulations in transcript levels are associated with the stages of acute development [5–7] and the onset of persistence [8,9], the regulatory mechanisms that govern these phenotypic shifts are not understood.
Sigma factors are responsible for the recruitment of the core RNA polymerase (RNAP) to cognate promoter elements, and, thus, their function dictates the subset of genes transcribed within a cell. Chlamydia encodes three sigma factors, σ66, σ54, and σ28, whose individual expression patterns [10–12] fail explain the stage-specific transcription profiles observed in acute chlamydial development [12]. Assuming these sigma factors participate in distinct functions, then post-expression mechanisms of regulation must theoretically be employed.
Switch-protein kinase modules are common effectors of energy and stress responses in bacteria. One of the best studied is the ‘Regulator of SigmaB’ or Rsb system in Bacillus subtilis. Within this type of module, a component called the switch-protein can either bind to and affect the function of a target protein, or function as a kinase in the phosphorylation of a network antagonist. Phosphorylation of the antagonist prevents further interaction with the switch-protein. The function of a competing PP2C-like phosphatase controls the level of active antagonist. In the absence of active (i.e. non phosphorylated) antagonist, the switch protein is driven towards its regulatory function. Commonly, the target of the switch-protein is a sigma factor, as is the case for the RsbW protein in B. subtilis (reviewed in [13]). Analogues of a switch-protein kinase regulatory module are conserved in the C. trachomatis genome [14] and were named after the B. subtilis module.
Putative components of the Rsb switch-protein regulatory system, outlined in Table 1, include one analogue of the switch-protein kinase (RsbW), two analogues of the module antagonist (RsbV1 and RsbV2), and three proteins that contain recognized PP2C-like phosphatase domains (RsbU, CT589, and CT259) [14]. Henceforth, chlamydial analogues with common family-member protein names will be designated with a ‘Ct’ subscript, e.g. RsbWCt. Previous studies have revealed that all potential module members are expressed [12,15] and that RsbWCt is a kinase specific for RsbV1 and RsbV2 - although a ‘switch’ function regulatory target has not been identified [16,17].
Tab. 1. Rsb analogues in the Chlamydiaceae. 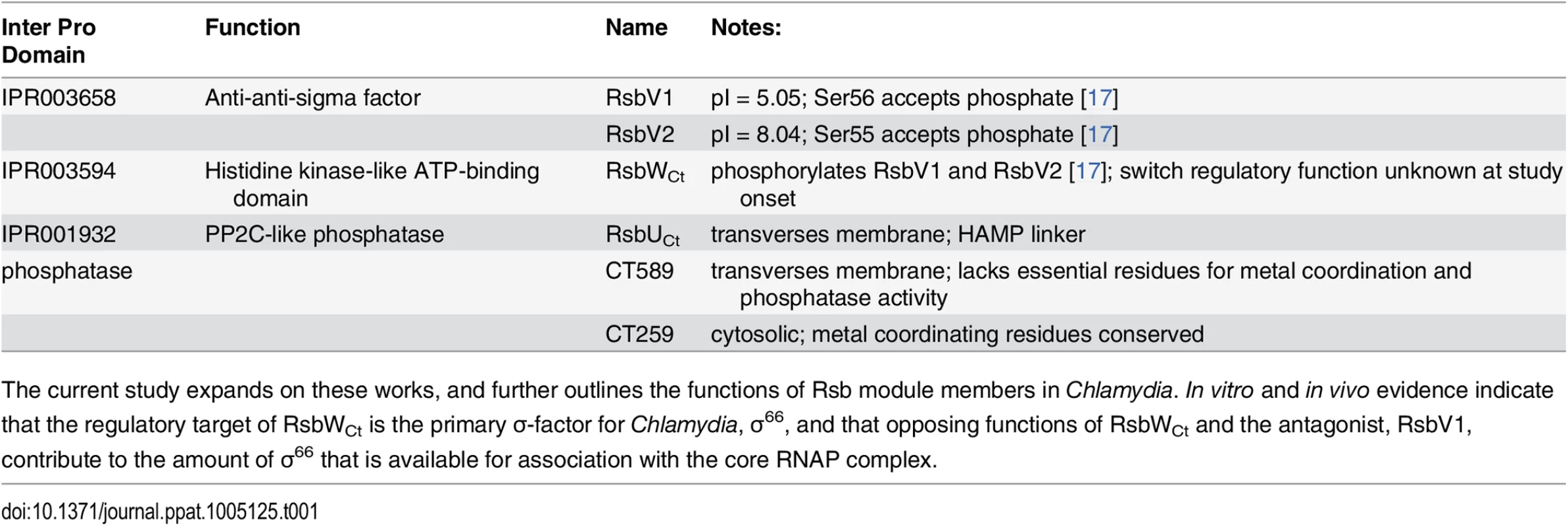
The current study expands on these works, and further outlines the functions of Rsb module members in Chlamydia. In vitro and in vivo evidence indicate that the regulatory target of RsbWCt is the primary σ-factor for Chlamydia, σ66, and that opposing functions of RsbWCt and the antagonist, RsbV1, contribute to the amount of σ66 that is available for association with the core RNAP complex. Results
RsbW associates directly with σ66
The regulated target of the putative switch-protein kinase analogue in Chlamydia (RsbWCt) had not been identified at the onset of this study. Therefore, a bacterial adenylate cyclase two-hybrid (BACTH) system [18] was employed to screen for interactions between RsbWCt and the three chlamydial sigma factors: σ28, σ54, and σ66 (Fig 1A). In order to gauge the relevance of this method for the screening of RsbW-type interactions, homologous proteins of known function from B. subtilis [19,20] were used as positive (σB + RsbWBs) and negative (σB + RsbTBs) control combinations. As expected, interaction of σB with its cognate anti-sigma factor, RsbW-Bs, complemented adenylate cyclase (AC) activity, and expression of the cAMP-dependent lacZ reporter cassette was detected via the Miller Assay. In contrast, co-expression of σB with RsbTBs (a paralogue of RsbWBs that does not bind to σB) did not exhibit complementation. When RsbWCt was expressed with the chlamydial alternative sigma factors, σ28 and σ54, similar activities to the empty vector controls were observed (t = 0.124 and t = 0.023, respectively; One-way ANOVA, Bonferroni's multiple comparison post test). In contrast, an interaction between RsbWCt and σ66 was clear (t = 3.633), as LacZ levels approached those of the positive control (GCN4 leucine zipper domains; ‘zip’). Interaction between σ66 and RsbWCt was observed in both cloning permutations of the BACTH assay, and expression of σ66 did not artificially activate the cAMP dependent reporter LacZ cassette in the absence of an RsbWCt fusion protein (S1 Fig).
Fig. 1. RsbWCt associates with σ66, but not σ54 or σ28. 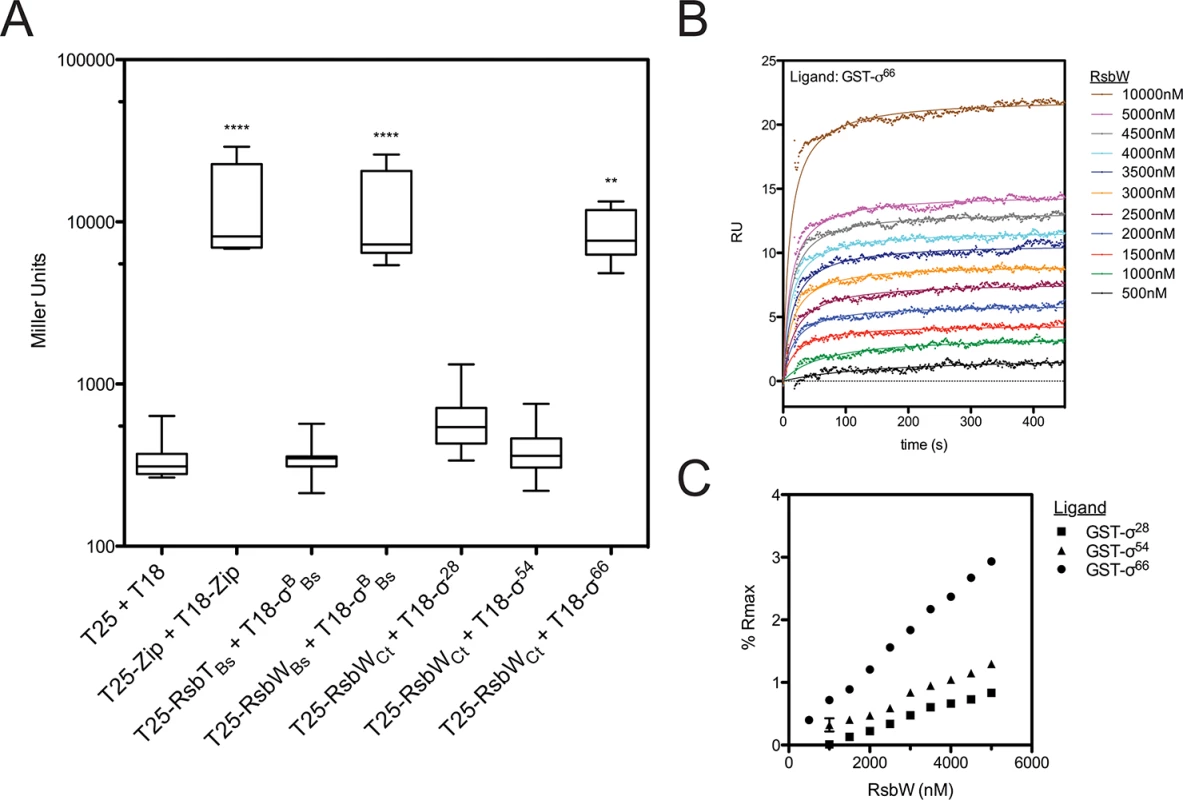
Interactions between RsbWCt and the chlamydial σ-factors were screened by BACTH (A). Mean β-galactosidase activity is displayed in Miller Units (MU). Each box represents the upper/lower quartiles transected by the arithmetic mean (N = 9). Whiskers represent the 5–95% confidence interval (** represents p<0.01, **** represents p<0.0001; One-way ANOVA with Bonferroni’s post-test against the negative control). SPR analysis confirmed interaction of RsbWCt and σ66. Overlay of sensorgrams from a single flow-cell in which σ66 had been immobilized prior to injection of increasing concentrations of RsbWCt show a dose-dependent response (B). Flowcells in which σ54 and σ28 were immobilized were assessed in parallel. Equilibrium rates were calculated by curve fitting and these values were transformed by the theoretical maximum binding for each ligand (Rmax). Percent of Rmax was plotted as a function of RsbWCt concentration (C). Error bars (typically miniscule) represent the 95% confidence interval of the Req derived from curve fitting analysis. The overlay and Req vs. [RsbWCt] plots are representative of 3 experiments. To validate the BACTH screen, molecular interactions were monitored in real time by surface plasmon resonance (SPR). Recombinant, purified σ-factors (σ66, σ28, and σ54) were immobilized to consecutive flowcells of an individual CM5 sensorchip, before the chip was charged with RsbWCt analyte at various concentrations. The change in response units (RU) over time, relative to a reference flowcell (immobilized Glutathione-S-transferase; GST), served as a function of binding between the ligand (sigma factor) and analyte (RsbWCt). Curves of best fit were applied to RU sensorgrams and the rate of equilibrium (Req) for each curve was determined (for example, a σ66-binding plot overlay is shown in Fig 1B). In order to compare the relative binding of RsbWCt to the three σ-factors, Req values were normalized by the theoretical maximum RU (Rmax) for each ligand, which was based on the original immobilization levels for each sigma factor. At every concentration tested, RsbWCt bound a greater percentage of immobilized σ66 than either of the alternative sigma factors (Fig 1C). This trend was consistent in multiple experiments (n = 3), although the overall percentage of theoretical Rmax bound appeared to decrease with the age of the immobilized ligand on each chip (from 1 to 3 days), indicating that the activity or conformation of the immobilized ligands decreased over time. When a GST-antibody capture system was used, in which GST-tagged sigma factors were captured by immobilized anti-GST immunoglobulin immediately prior to addition of RsbWCt, up to 40% of the σ66 Rmax was bound at a concentration of 5 μM (S2 Fig). Thus, these data indicate that RsbWCt associates with the sigma factor, σ66.
Interaction of RsbW with RsbV1 or RsbV2 depends on ATP and the phosphorylation state of the antagonist
RsbWCt is a kinase specific for RsbV1 and RsbV2, and the phosphorylated residues of the antagonists have been mapped to Serine-56 and Serine-55, respectively [17]. In order to gain insight into the kinetics of the kinase activity for RsbWCt against RsbV1 and RsbV2, aliquots from phosphorylation reactions were removed iteratively and resolved on a polyacrylamide gel supplemented with Phos-tag, an agent which causes an electromobility shift of phosphorylated proteins. When incubated in the presence of ATP, RsbWCt induced a mobility shift of both RsbV1 and RsbV2, although there was a clear distinction in the rates at which the two antagonists were phosphorylated; after only 10 minutes, 100% of RsbV1 migration was shifted, whereas only a fraction of RsbV2 was shifted after 2 hours of incubation with RsbWCt (Fig 2A and 2B).
Fig. 2. Association of RsbW-Ct with antagonist depends on ATP and the phosphorylation state of the antagonist. 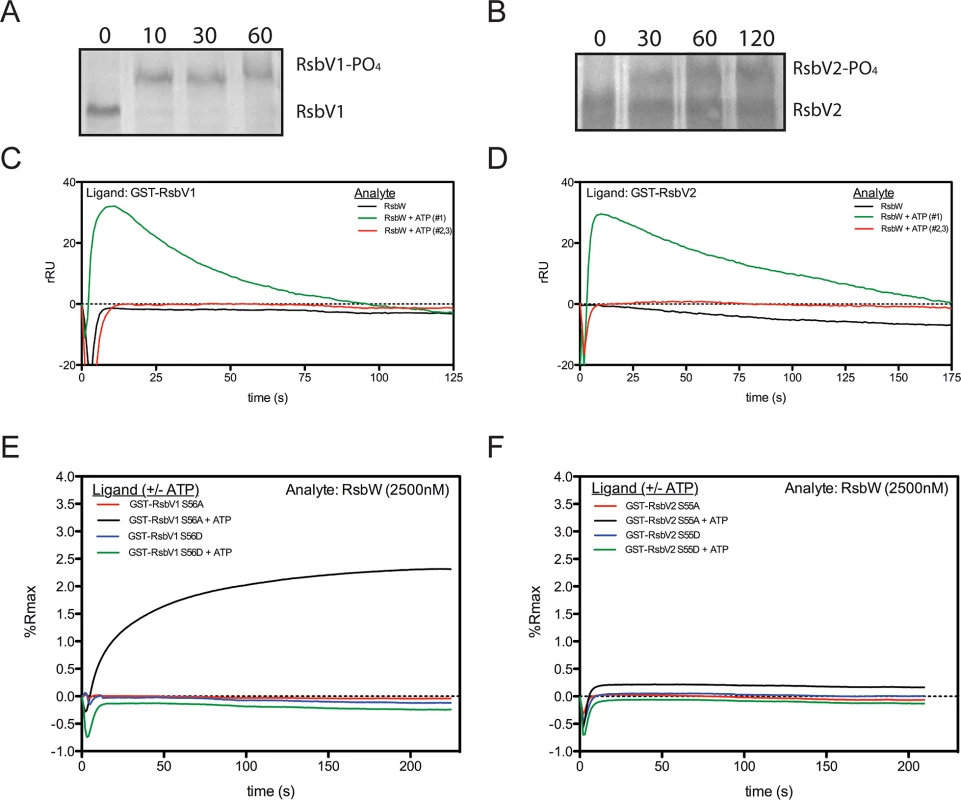
Purified RsbV1 (A) and RsbV2 (B) were incubated in the presence of RsbWCt + ATP and monitored longitudinally (minutes) for phosphorylation, detected via electromobility shift during Phos-tag supplemented gel electrophoresis. Each panel represents two experiments in which the same results were obtained. For SPR analysis, antagonists RsbV1 (C) and RsbV2 (D) were immobilized and charged with 2500nM RsbWCt- in the absence (black) or presence (first attempt, green; repeated attempts, red) of ATP. Response over time was normalized by the response in an empty flow-cell. Sensorgrams shown represent two experiments (separate sensorchips) in which the same trends were observed. Derivative mutants, in which the phospho-accepting residues were changed to aspartic acid (S56D, S55D) or alanine (S56A, S55A) for RsbV1 (E) and RsbV2 (F), respectively, were tested for their ability to associate with RsbWCt. Serine to aspartic acid mutations, mimicking a permanently phosphorylated state, prevented the association of RsbWCt in both the absence (blue) and presence (green) of ATP for both antagonists. Alanine derivatives of both antagonists, which cannot be phosphorylated and mimic a permanently active state, were bound in the presence of ATP (black), but not without ATP (red). To determine the direct effects of phosphorylation on the interaction between RsbWCt and antagonist, SPR was again utilized; RsbV1 and RsbV2 antagonists were immobilized onto individual flow-cells of a sensorchip and RsbWCt was then supplied in the absence or presence of ATP. Without ATP, RsbWCt did not bind to RsbV1 (Fig 2C) or to RsbV2 (Fig 2D). When ATP was supplied along with RsbWCt, both ligands produced abnormal binding curves, in which an initial increase in response gradually returned to the baseline level. Subsequent applications of RsbWCt plus ATP yielded no response with either ligand. We reasoned that upon addition of ATP, RsbWCt was able to associate initially with, then phosphorylate RsbV1 and RsbV2, but that phosphorylation prevented their further interaction.
To explore this hypothesis, similar SPR experiments were carried out on derivatives of RsbV1 and RsbV2, in which the phospho-accepting residues were mutated to mimic either an immutable non-phosphorylated state (serine to alanine), or a permanent phosphorylation state (serine to aspartic acid). The addition of RsbWCt without ATP exhibited no binding response to any of the four antagonist derivatives (Fig 2E and 2F). Supplementation of ATP with RsbWCt yielded no binding response to the RsbV1 S56D or RsbV2 S55D derivatives, suggesting that a negative charge (from the phosphate group or, in this case, the aspartic acid residue) at position 56/55, respectively, of the antagonist abrogated association with RsbWCt. Supplementation of ATP with RsbWCt produced a typical saturation-binding curve to the RsbV1 S56A and, to a lesser extent, RsbV2 S55A antagonist derivatives. The S56A or S55A derivative antagonists gave a similar binding response on subsequent RsbWCt plus ATP injections, presumably because of their inability to accept phosphorylation and, rendering them inactive. Together these data indicate that the binding of RsbWCt to its RsbV1 and RsbV2 antagonists depends on both the phosphorylation state of RsbV1 or RsbV2 and ATP.
Phosphorylated RsbV1 is a substrate of the C-terminal PP2C-like domain of RsbUCt
Three proteins contain recognized PP2C-like phosphatase (IPR001932) domains in C. trachomatis: CT259, RsbUCt and CT589 (Table 1). RsbUCt and CT589 are paralogues, both predicted to transverse the membrane with a hypothetical domain localized in the periplasm and a PP2C-like domain localized in the cytoplasm [14,17]. In contrast, CT259 appears to consist of a single cytosolic domain (Fig 3A). When aligned with the PP2C-like domains of RsbUBs and SpoIIE from B. subtilis, the residues involved in two Mn2+ coordination sites, which are essential for phosphatase activity, are conserved in RsbUCt, but not in CT589 [17]. CT259 appears to maintain both divalent metal coordination sites, although the first site exhibits conservative aspartic acid to glutamic acid mutations (S3 Fig).
Fig. 3. Proteins containing putative PP2C-like phosphatase domains in C. trachomatis. 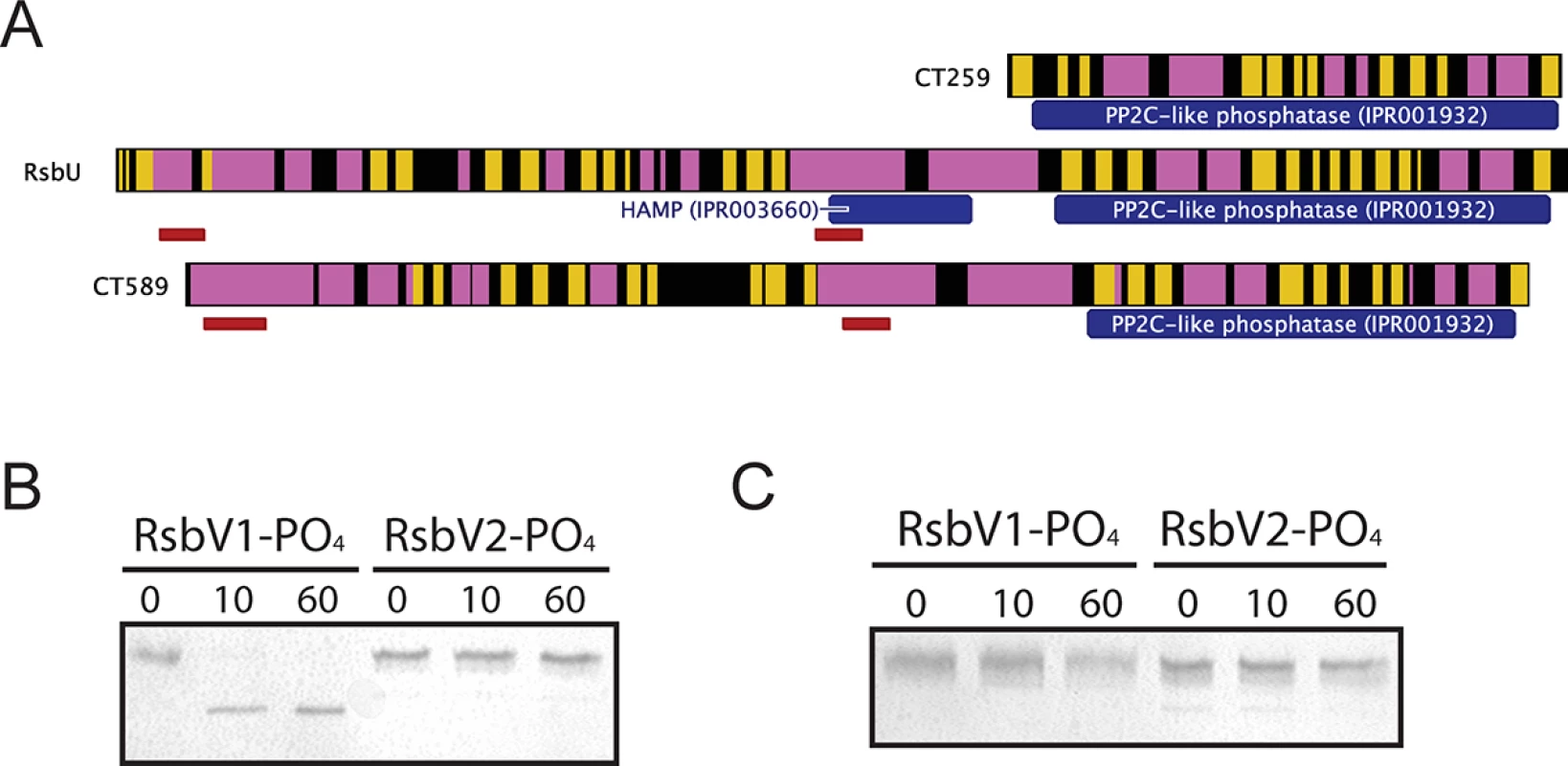
(A) Predicted secondary structure is shown for CT259, RsbU-Ct and CT589 (β-sheet represented by yellow, α-helix represented by pink). The RsbUCt and CT589 paralogues are both predicted to transverse the bacterial inner membrane (represented by red bars), whereas no transmembrane domains were predicted for CT259. Phosphatase assays revealed that the RsbUCt PP2C-like domain was capable of dephosphorylating RsbV1, but not RsbV2 (B). CT259 did not dephosphorylate either antagonist (C). Each panel is representative of two experiments in which the same results were obtained. To test if the carboxy-terminal PP2C-like domain of RsbUCt was an active phosphatase, the sequence corresponding to the final 258 amino acids was cloned into an expression vector for recombinant purification. This C-terminal domain of RsbUCt (referred to as C-RsbUCt) was incubated with RsbV1 or RsbV2 that had been previously phosphorylated by RsbWCt. The phosphorylation states of the antagonists were distinguished by resolution on a Phos-tag supplemented acrylamide gel. Introduction of C-RsbUCt caused a rapid shift from the phospho - to non-phosphorylated form of RsbV1, however phospho-RsbV2 remained phosphorylated throughout the entire time-course (Fig 3B). CT259 was also examined for phosphatase activity against the two phospho-antagonists, though no activity was observed (Fig 3C). Thus, while the C-terminal domain of RsbUCt appears to maintain phosphatase activity for phospho-RsbV1, no phosphatase capable of recognizing RsbV2 was identified.
Manipulation of Rsb pathway components in vivo
A chlamydial shuttle vector for the controlled expression of a target cassette in Chlamydia was engineered in order to further investigate the role of Rsb components in vivo. Briefly, the shuttle vector, pGFP::SW2 [21], was modified such that the constitutive promoter driving the gfp-cat cassette was replaced with the tetracycline inducible promoter system from pRPF185 [22], producing pCT308-GFP (S4A Fig). The gfp cassette was then replaced with sequences corresponding to rsbW or rsbV1 from C. trachomatis serovar D/UW/CX genomic DNA to make pCT1310-RsbW and pCT1310-RsbV1, respectively. These three shuttle vectors transformed C. trachomatis strain L2/25667R (originally lacking any plasmid; henceforth referred to as L2R) and were stable through several passages in penicillin-supplemented medium, prior to plaque purification, expansion, and density gradient harvest. Strains harboring these shuttle vectors were as resistant to penicillin as the original parent shuttle vector, pGFP::SW2 [21] with concentrations of 0.01–0.05 U/ml failing to have any effect on generation of infectious progeny, despite the same concentrations inhibiting the C. trachomatis L2R strain lacking any plasmid (S4B Fig). Endogenous fluorescence was not detected in Chlamydia harboring pCT308-GFP in the absence of ATc, but was detected upon supplementation of the growth medium (S4C Fig).
Additionally, a strain in which rsbV1 was inactivated using insertional mutagenesis [23] was engineered. Briefly, a GII intron carrying the aadA-marker (to confer resistance to spectinomycin) was targeted for insertion into the 5’ region of rsbV1, creating DFCT15 (rsbV1::GII[aadA], S5 and S6 Figs). The mutant strain was resistant to spectinomycin and sequencing of the disrupted rsbV1 locus confirmed the GII intron insertion, resulting in alteration of the wild type RsbV1 sequence after 10 amino acids. Serial passage in the absence of spectinomycin followed by PCR analysis of the insertion-site using rsbV1-specific primers confirmed marker stability in the absence of selection, as previously observed for the GII intron carrying the bla-marker [23]. Consequently, experiments were performed without spectinomycin when matched with non-spectinomycin resistant strains.
RsbWCt and RsbV1 affect transcription from σ66-dependent promoters in Chlamydia
Based on the results of in vitro binding assays and the respective functions of homologous proteins in other bacteria, we hypothesized that RsbWCt was a negative regulator of σ66 and that non-phosphorylated RsbV1 would act as a positive regulator of σ66 by antagonizing RsbWCt. To test this hypothesis in vivo, we monitored the expression of bona fide σ66-dependent genes in our L2R transformant and rsbV1 knockout strains. Normalization of transcript expression in Chlamydia is not trivial (e.g. [8,24]), especially considering our hypothesis of differential ‘housekeeping’ transcription. We tested a number of different normalization techniques that did not assume that any chlamydial gene would correlate unequivocally to the number of chlamydiae present within the sample. While use of C. trachomatis-specific gDNA from parallel samples as an exogenous control has been employed frequently (e.g. [8,25]), this type of normalization fails to account for variation in RNA loading / efficiency of reverse transcription as a source of error. We found that use of the geometric mean of Chlamydia-specific gDNA from parallel samples and endogenous host cell gapdh as a normalization factor for each sample produced a data set with the lowest cumulative intrasample error (S7 Fig). Primary Cq values, along with normalization calculations, are available in S1 Dataset. As expected, rsbW transcript expression was elevated in the L2R pCT1310-RsbW strain (Fig 4A), whereas rsbV1 transcript expression was elevated in the L2R pCT1310-RsbV1 strain (Fig 4B). Transcript of rsbV1 was not detected by RT-qPCR in DFCT15 (rsbV1::GII), confirming genetic analysis (S6 Fig). Expression of the gfp-cassette in L2R pCT308-GFP strain reached similar levels of the other expression cassettes (data available in S1 Dataset).
Fig. 4. Analysis of σ66-dependent gene expression in response to modified rsb component expression. 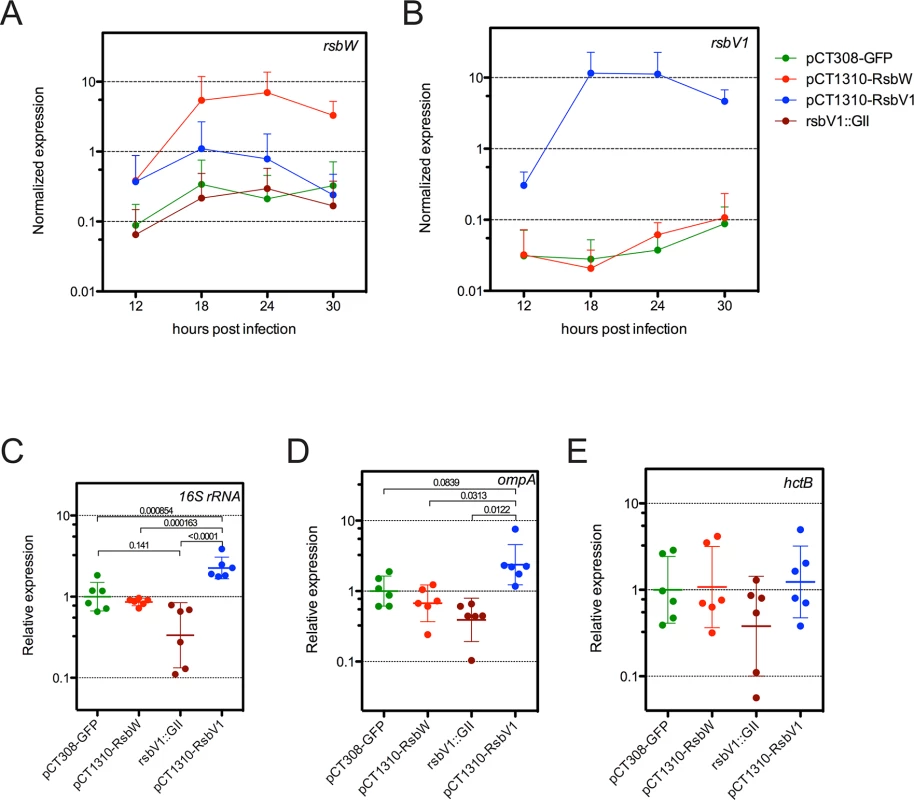
Transcripts levels of rsbW (A) or rsbV1 (B) were monitored in L2R strains harboring shuttle vector plasmids pCT308-GFP, pCT1310-RsbW, or pCT1310-RsbV1, as well as in strain DFCT15, in which rsbV1 had been inactivated via transposon insertion (rsbV1::GII). The expression levels of target cassettes were elevated in their respective expression strains, whereas no rsbV1 transcription was detected in the rsbV1::GII strain. Expression of target cassettes are shown as the mean normalized expression as a function of time (hours post infection). Error bars represent the 95% CI. Normalized expression of 16S rRNA (C), ompA (D), and hctB (E) were calibrated to the geometric mean of the GFP-expression control. Lines indicate the geometric mean of relative expression and error bars represent the 95% CI. P-values are derived from One-way ANOVA with Tukey’s multiple comparisons post-test performed in R. Transcription of ompA and 16S rRNA are σ66-dependent [26–29], whereas transcription of hctB is σ28-dependent [30]. Only σ66-dependent genes experienced differential expression. To test whether ‘housekeeping’ transcription was affected in the transformant / knockout mutant strains, we assessed the transcription of two genes with bona fide σ66-dependent promoter systems, 16S rRNA [26,27] and ompA [28,29], limiting our analysis to time-points prior to the typical RB to EB re-differentiation in order to avoid re-differentiation as a confounding variable. As predicted, σ66-dependent transcription was elevated in the L2R pCT1310-RsbV1 strain, with the pooled relative expression of ompA and 16S rRNA reaching a geometric mean of 2.32-fold over the control (95% CI = 1.72 - to 3.11-fold). Conversely, the relative expression of σ66-dependent transcription was reduced to 0.362 of control (95% CI = 0.224 to 0.583) in the rsbV1::GII strain. Notably, σ66-dependent transcription in the L2R pCT1310-RsbW strain was reduced from the control expression (95% CI = 0.585 to 0.988), however did not reach similar repression levels as the rsbV1::GII strain. This negative effect has been observed in each of two previous analysis of expression experiments (summary figures shown S8 and S9 Figs). The observation that the rsbV1::GII strain exhibits lower amounts of σ66-mediated transcription compared to the RsbWCt expression strain may indicate that the levels of rsbW achieved in these experiments were insufficient to completely overcome antagonism from endogenous RsbV1 levels, resulting in a subtle but consistent phenotype.
As a control, we also assessed whether σ28-dependent transcription of hctB [30], was affected in the transformant / knock-out mutant strains, with the caveat that all known σ28-dependent genes are transcribed during the late stages of the developmental cycle due to repression by an early-stage transcriptional repressor (EUO) [31,32]. Indeed, we were not able to detect hctB transcription in all samples at 12 hpi, and levels at 18 hpi neared the limit of detection. Thus, we analyzed expression of hctB during the late stages of the developmental cycle. At these time-points, no differential expression was observed. Taken together with the in vitro binding data, our results suggest RsbWCt is an anti-sigma factor of σ66, and that RsbV1 is a bona fide antagonist of the RsbWCt to σ66 association.
Ectopic expression of RsbWCt and RsbV1 affects chlamydial replication and development
We hypothesized that modulating the activity of the ‘housekeeping’ sigma factor would have detectable effects on Chlamydia, yet no obvious effect on growth or development was observed during the passage and selection of pCT1310-series transformants. Immunofluorescent analysis (IFA) revealed that all transformant strains exhibited a morphology consistent with acute development, and that no aberrant, enlarged chlamydial particles consistent with the persistent phenotype were observed. However, we did observe that inclusions from the L2R pCT1310-RsbV1 strain appeared much larger than the GFP or RsbWCt expression strains (p<0.0001; One-way ANOVA, Tukey’s multiple comparison post-test versus both other samples; S10 Fig). We reasoned that increased inclusion size observed in the L2R pCT1310-RsbV1 strain might be attributed to increased metabolic capacity upon elevated ‘housekeeping’ transcription, and if so, this may correlate to modulations in growth and development. To test this hypothesis, we analyzed the transformant and rsbV1 mutant strains for genomic replication (1-step growth), recoverable infectious progeny (2-step growth), and plaque expansion.
For both 1-step and 2-step growth analysis, data was normalized by the empirical IFU input for each experiment, such that the data presented accounts for the actual number of infection events for each sample. Differential genomic replication was not overtly evident between the strains, though analysis of the area underneath each growth curve did reveal a trend that mirrored levels of σ66-dependent transcription, with L2R pCT1310-RsbV1 exhibiting the highest chlamydial load, followed by L2R pCT308-GFP, and then L2R pCT1310-RsbW and the rsbV1 mutant (Fig 5A). As a second indicator of development, recoverable infectious progeny (2-step growth) was also monitored (Fig 5B). At 24 hpi, the L2R pCT1310-RsbV1 strain exhibited a 2.3-fold increase in infectious progeny compared to control (p = 0.0026; One-way ANOVA with Tukey’s multiple comparisons post-test), whereas L2R pCT1310-RsbW exhibited a 3.1-fold reduction (p = 0.181). The rsbV1 mutant also displayed a reduction in infectious progeny that exceeded that of L2 pCT1310-RsbW (p = 0.108 versus control). Thus recoverable infectious progeny and, to a lesser extent, genomic replication exhibited a pattern that correlated with observed σ66-dependent transcription.
Fig. 5. The effect of rsbW and rsbV1 expression on genomic replication and development of infectious progeny. 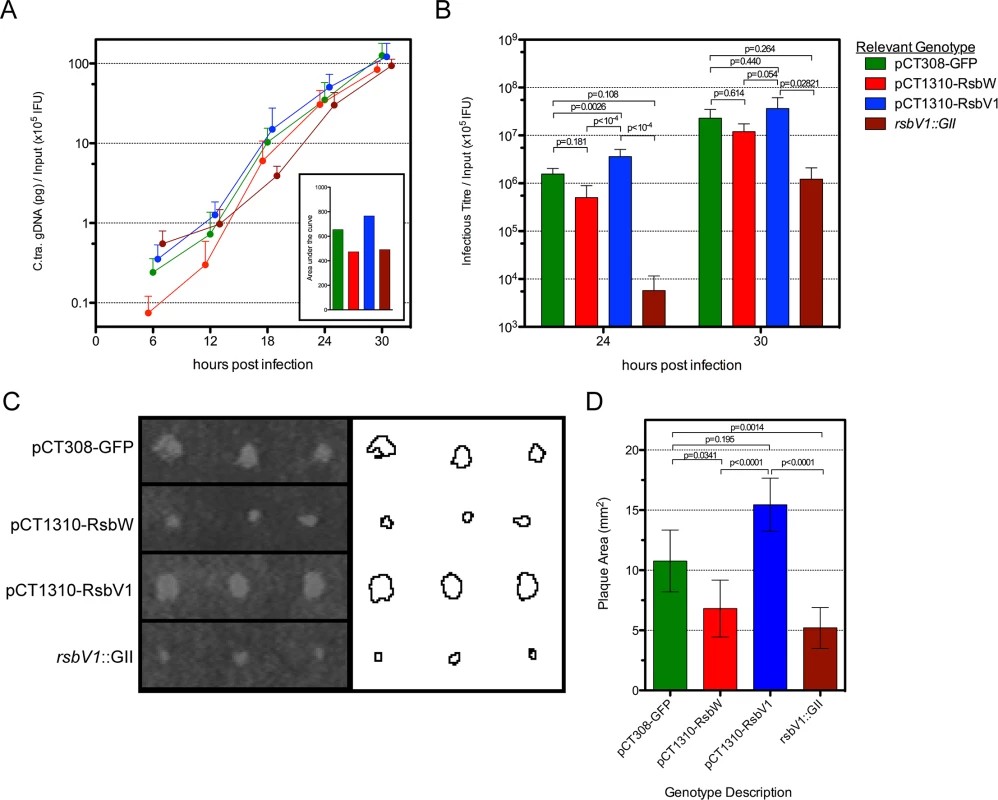
1-step and 2-step growth from time-course experiments were monitored for strain L2R harboring shuttle vector plasmids pCT308-GFP (green), pCT1310-RsbW (red), pCT1310-RsbV1 (blue), or from strain DFCT15 (rsbV1::GII; maroon). The mean of C. trachomatis gDNA, normalized by the empirical IFU input (x105) is plotted by hours post-infection (A). The inset shows the area under each curve. The mean of recoverable infectious progeny from the same experiments was also normalized by empirical input (B). Error bars for both panels represent the 95% confidence interval. P-values derived from One-way ANOVA with Tukey’s multiple comparisons test performed in R. Plaque expansion from infection foci were measured using FIJI freeware (C). Bars represent mean plaque size (mm2) from all foci at days 8 and 9 post-infection (D). Error bars represent 95% confidence intervals. P-values derived from One-way ANOVA with Dunn’s test for multiple comparisons using rank sums in R. However, because neither one-step or two-step growth analysis alone is a perfect measure of Chlamydia fitness, we chose to analyze transformant and mutant strains in a plaque expansion assay. Because plaque expansion depends on all aspects of chlamydial development (e.g. replication, re-differentiation, cell exit, and secondary host cell entry), the relative size of plaques can be used as an indicator of overall chlamydial fitness.
Specific locations within HeLa monolayers were inoculated with the modified Chlamydia strains and plaques were measured after 8–9 days, using a semi-automated procedure in FIJI [33]. An example of this process is shown in Fig 5C, in which plaques from day 9 post-inoculation are shown. In concordance with the 1-step and 2-step growth profiles, the L2R pCT1310-RsbV1 strain exhibited the highest rate of plaque expansion (Fig 5D). Moreover, the L2R pCT1310-RsbW and DFCT15 (rsbV1::GII) strains yielded smaller plaques than the GFP expression control. Thus, in concert with in vitro and transcript expression data, these results support a model in which the availability of σ66 is affected by relative levels of RsbW and RsbV1 and that their experimental manipulation was capable of influencing the rate at which C. trachomatis infection progresses in a cell culture model.
Discussion
The evidence presented in this report indicates that the primary target of the Rsb phosphoregulatory network in Chlamydia trachomatis is σ66, the main ‘housekeeping’ sigma factor of the pathogen. Interaction of RsbWCt with σ66 was shown indirectly by bacterial two-hybrid assay and directly by SPR analysis. Moreover, previous results that RsbWCt is a kinase for both RsbV1 and RsbV2 in vitro [17] were confirmed, although there is a difference in efficiency between these phosphorylation events, with RsbV1 being the preferred substrate. RsbWCt does not associate with RsbV1 or RsbV2 in their phosphorylated forms, or, interestingly, in the absence of ATP. We further observed in vitro phosphatase activity from the RsbUCt PP2C-like domain that is specific for phospho-RsbV1, but not for phospho-RsbV2. Thus, a complete signaling module (consisting of a system phosphatase, antagonist, switch-protein, and target) has been characterized in this report. To verify the model generated from in vitro assays, mutant and transformant strains of Chlamydia were engineered for in vivo analysis. Elevated expression of RsbV1 correlated with the enhanced expression of bona fide σ66-dependent transcripts and a more rapid growth profile in multiple assays. In contrast, elevated expression of RsbWCt and the inactivation of its antagonist both resulted in reduced transcription of representative σ66-dependent genes and a depressed growth profile.
Taken together, these results provide a mechanism by which the Rsb network could control σ66 availability, and perhaps growth rate, in response to various stimuli. We postulate the following working model (Fig 6). Under steady-state conditions, the equilibrium of the Rsb network provides a molecular ‘speed-limit’ on σ66 activity and subsequently on metabolic activity (Fig 6A). Upon experiencing increased levels of active (i.e. non phosphorylated) RsbV1, the equilibrium of RsbWCt function would be driven away from sequestration of σ66, resulting in amplified levels of ‘housekeeping’ transcription. Possible inputs for this pathway would be the increased activity/expression of RsbUCt, or increased expression of RsbV1 (Fig 6B). Alternatively, upon decreased expression or activity of RsbV1, the equilibrium of RsbWCt would be driven towards sequestration of σ66, limiting ‘housekeeping’ transcription and possibly restricting metabolism. Potential inputs for this shift would include decreased expression or activity of RsbUCt, decreased expression of RsbV1 (Fig 6C), or, intriguingly, decreased levels of ATP. A mechanism that links energy stress to decreased general levels of transcription seems plausible, however a procedure for the manipulation or even measurement of Chlamydia ATP levels (i.e. to distinguish host versus pathogen ATP pools) has not been elucidated, making exploration of this hypothesis difficult.
Fig. 6. Proposed model for the function of the Rsb regulatory system in Chlamydia. 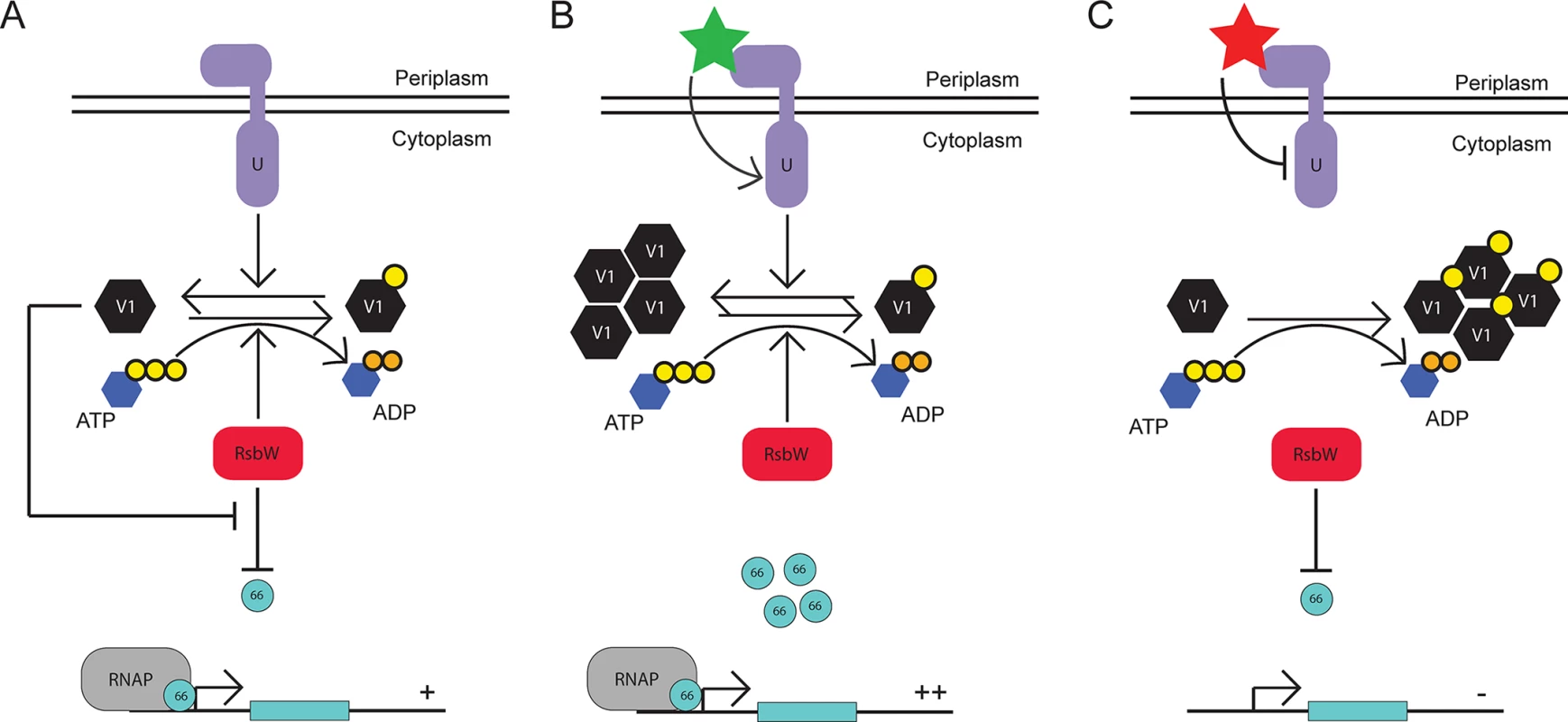
Under steady-state conditions, the expression levels and activies of RsbUCt (purple), RsbV1 (black), and RsbWCt (red) provide an equilibrium in which σ66 availability is ample for normal growth and development (A). Upon an accumulation of active (i.e. non phosphorylated) RsbV1 via increased expression or increased activity/expression of RsbUCt, the equilibrium of RsbWCt function shifts away from the anti-sigma factor role, allowing increased transcription of σ66-promoted genes (B). Upon the decreased availability of active RsbV1 (either through reduced expression or reduced RsbUCt expression/activity), the equilibrium of RsbWCt function is driven towards the anti-sigma factor function, limiting the amount of σ66 that is available for association with the core RNAP complex (C). We were initially surprised to observe that the association between RsbWCt and RsbV1 is dependent on ATP, which represents a mechanism distinct from Rsb components in the in B. subtilis module [34]. However, this distinction may reflect the differences in regulated targets with σB as an alternative sigma factor responsible for activating transcriptional response to stress, and σ66 as the primary sigma factor for Chlamydia. Under low ATP conditions in B. subtilis, RsbWBs is not able to inactivate RsbVBs such that they associate stably, liberating σB for the initiation of the stress response [34]. However, under low ATP conditions in Chlamydia, the theoretical association between anti-sigma factor and antagonist would not occur, driving the function of RsbWCt towards sequestration of σ66 and the reduction in ‘housekeeping’ transcription. We postulate that the ATP-dependence of anti-sigma to antagonist interaction theoretically must switch the Rsb system from one of stochastic activation in B. subtilis [35] to one of stochastic inactivation in Chlamydia.
This study extends the work of Hua et al [17], which characterized several interactions within this module, including kinase activity of RsbWCt and detailed in silico analysis of the module members. Yet, they failed to observe any interaction between RsbWCt and any of the chlamydial σ-factors in a yeast-two hybrid system. Furthermore, in vitro transcription assays showed no interaction between RsbWCt and σ28, and a separate study failed to observe any effect of RsbWCt in a heterologous σ28-mediated transcription assay in Salmonella enterica [16,17]. While we concur that σ28 does not interact with RsbWCt, we provide evidence that RsbWCt indeed interacts with σ66. One possible explanation for this discrepancy could be that the σ66-RsbW complex was not targeted properly to the yeast nucleus due to its size, stoichiometric ratio, etc. By using a bacterial two hybrid reliant on generation of a cytosolic metabolite, neither complex size nor nuclear import was an issue.
Interestingly, other proteins have been described as modulators of σ66 function in Chlamydia. For instance, CT663 has been likened to the Rsd protein in E. coli [36], which inhibits σ70-mediated transcription during stationary phase growth [37]. Another protein, GrgA, reported as a non-specific DNA binding protein, also associates with the non-conserved region of σ66. By interacting with both simultaneously, GrgA enhances the transcription of σ66-dependent promoter systems in vitro [38]. Thus, there is a clear precedent for the alteration of σ66 activity as an evolved strategy in Chlamydia. We add RsbWCt to this list of σ66 modulators characterized in vitro, and additionally provide in vivo evidence to support its function in this role.
We would like to note that we also attempted to transform C. trachomatis with shuttle vectors capable of expressing RsbV1_S56A, RsbV2, RsbV2_S55A, RsbU, CT589, and CT259, of which none transformed Chlamydia in at least 2 attempts (in which a positive control for transformation was employed and successful). We also attempted insertional inactivation of rsbV2, rsbU, and ct589, but were unsuccessful. As transformation methodologies are relatively new for Chlamydia, we are unsure whether these observations can be attributed to effects on chlamydial fitness (e.g. such manipulations are lethal or at least highly detrimental) or are simply technical failures. The use of a positive control vector for each transformation, as well as the use of multiple plasmid backbones (pCT1310 - and p2TK-SW2 [39]) for attempted transformations suggest the former may be more likely. Interestingly, of the genes targeted for manipulation, those that were successfully altered exhibited mild, but reproducible, phenotypic changes. Thus, it is plausible that any robust alteration in the function of Rsb module (e.g. the overexpression of constitutively active RsbV1 S56A) could be highly detrimental to chlamydial growth under selective cell culture conditions. Further insights into the process of chlamydial transformation should shed light onto the nature of these negative results.
What, then, is the evolutionary function of the Rsb system in Chlamydia? To our knowledge, there is no phenotype (occurring during acute or persistent modes of growth) that demonstrates a global modulation in σ66-mediated transcription. Perhaps this is not surprising considering that beyond basic expression and availability of sigma factors, other global transcriptional repressor proteins are likely to be influential at all stages of Chlamydia development. For instance, the protein EUO controls transcription of both σ66 - and σ28-dependent late genes via interaction with operating elements found in both promoter types during the early stages of infection [31]. Thus, even if the Rsb network dictated an increased availability of σ66, transcription of σ66-dependent late genes would be occluded by the presence of the EUO DNA-binding protein. While it is possible the Rsb module works in concert with other global regulators in order to actualize RB to EB re-differentiation, we favor a model where its function provides a molecular 'speed-limit' for housekeeping transcription (and subsequently metabolism) as a way to avoid overuse of potentially limited nutrients. Manipulation of expression levels of RsbWCt and RsbV1 via ectopic expression or insertional inactivation resulted in differential growth and development in a nutrient-replete cell culture model. The elucidation of the activities of the system components in response to inimical growth conditions could provide more resolution into the question of the evolutionary role of this system in Chlamydia trachomatis.
In conclusion, this report provides multiple lines of evidence that indicate the Rsb module is a bona fide molecular circuit capable of influencing the availability of the main sigma factor in Chlamydia. The potential of this network to accelerate and restrict growth rate and development substantiates it as an important pathway for further study, and may even constitute a novel target for generation of attenuated, or even accelerated, growth mutants.
Materials and Methods
Strains and parent plasmids
Chlamydia trachomatis serovar L2/25667R, pGFP::SW2 shuttle vector, and the BACTH vectors and DHM1 E. coli were provided by Dr. Scot Ouellette (University of South Dakota, Vermillion). The pRFP185 plasmid was received from Dr. Robert Fagan (University of Sheffield, Sheffield). Bacillus subtilis subspecies subtilis strain 168 was a received from Professor Neil Fairweather (Imperial College, London). Chlamydia trachomatis serovar D/UW/Cx was provided by Dr. Rey Carabeo (University of Aberdeen, Scotland).
Cloning
All PCR reactions intended for cloning purposes were performed with Phusion High-Fidelity Polymerase (NEB). Either C. trachomatis genomic DNA (Serovar D/UW/CX) or B. subtilis sbsp. subtilis 168 genomic DNA was used for template in reactions. Primer sequences are listed in S1 Table. Insert and vector ligations were either performed using traditional restriction endonuclease digest (NEB) and ligation (T4 Ligase; NEB), or in a one-step Gibson Assembly cloning reaction (NEB). Plasmids intended for recombinant protein expression are listed in S2 Table, and plasmid shuttle vectors for the transformation of C. trachomatis are listed in S3 Table. Site directed mutagenesis of pGEX expression vectors for the antagonist proteins was also carried out with PCR based Gibson Assembly. All plasmid insert sequences were verified by Sanger sequencing (GATC; Germany or Macrogen, USA).
Bacterial two hybrid assay
The bacterial adenylate cyclase two-hybrid method for assessment of protein-protein interaction was completed as per the instructions of the manufacturer (EuroMedex). Detailed methods are described in S1 Text. The Miller Assay determined expression of the reporter LacZ [40]. Sample groups in all graphs represent an equal number of biological replicates, indicated in figure legends.
Surface plasmon resonance
The Biacore 3000 instrument (GE Healthcare) revealed biomolecular interactions via surface plasmon resonance (SPR). Specifics for each run are described in supplemental information. CM5 sensorchips were used for all experiments. The optimal pH for pre-concentration of ligands was determined using the pH scouting wizard (Biacore 3000 software), and ligands were using the amine coupling kit. All experiments were carried out in a buffer of 10mM HEPES, 150mM NaCl, and 1mM MgCl2 at 30°C. All consumables were purchased from GE Healthcare.
Protein purification
Proteins were expressed from pGEX-derived vectors in PC2 E. coli [41]. Soluble proteins were purified by standard techniques. Insoluble proteins were liberated from inclusion bodies using sarkosyl as described in the S1 Text. All proteins were assessed for purity via SDS-PAGE and Coomassie-Blue stain. Detailed protocols are available in S1 Text.
Bioinformatics
All DNA and protein sequence diagrams were generated using Geneious version 7.0.2, created by Biomatters. Secondary structures were predicted by the Phyre2 algorithm [42]. Transmembrane regions were predicted using the Hidden Markov Model in Geneious. InterPro domains were identified using the European Bioinformatics Database (EBI). Accession numbers listed are from the UniProtKB database.
Kinase and phosphatase assays
In vitro kinase/phosphatase assays were performed using recombinant, purified preparations of the system components and are described in detail in the S1 Text. Phos-tag reagent (Alpha Laboratories) was utilized to shift the migration of phosphorylated RsbV1 or RsbV2 during SDS-PAGE.
Chlamydia trachomatis transformation and insertional mutategenesis
Transformation of C. trachomatis L2/25667R (plasmid-deficient) with ectopic expression vectors was carried out, as described [21,43] with slight modifications highlighted in the S1 Text. Every transformation attempt utilized pGFP::SW2 as a positive control (100% success rate). DFCT15 was generated from the transformation of C. trachomatis strain L2/434/Bu with plasmid pDFTT6aadA as described in [23] with the exception that spectinomycin was used for selection at 500 ug/ml instead of ampicillin for mutant selection and plaque isolation of clones. The intron was targeted to insert between base pairs 28 and 29 of rsbV1 (TCCCTTGTAAATGAAGGATGCCTGTTTGGC—intron–CTTGTTCTTCTTTCT) in an antisense orientation. The predicted insertion efficiency values were an E-value of 0.75 and a score of 8.51. Intron re-targeting was performed as directed by the TargeTron manual (Sigma-Aldrich). Both transformant and knock-out strains were considered plasmid-competent.
Chlamydia infections
HeLa cells (ATCC) at 80–95% confluence in a 6 - or 12-well cluster plate were inoculated with C. trachomatis diluted in Hank’s Balanced Salt Solution prior to centrifugation (500xg for 15 minutes at 20°C) and incubation at 37°C for 30 minutes. The inoculum was then aspirated before addition DMEM supplemented with 10% fetal bovine serum (FBS) and other supplements as noted. For cassette induction, anhydrous tetracycline (Sigma) was supplemented to a final concentration of 5 ng/ml at 6 hours post infection.
1-step growth analysis
C. trachomatis strains were grown on the same cluster plate for 6–30 hpi. At designated time points, infected monolayers were washed, trypsin-treated into suspension, pelleted and stored in PBS at -80°C. Total genomic DNA was extracted and diluted to a final concentration of 1 ng/ml. Chlamydia-specific gDNA was assayed twice using qPCR (see Quantitative PCR below) with primers amplifying a region of the hypothetical gene, ct652.1, or the 16S ribosomal subunit gene. Starting quantities were normalized against the empirical IFU input for each strain in order to account for any differences the actual versus intended MOI within each sample.
2-step growth analysis
Samples intended for quantification of recoverable infectious progeny were dislodged into 1ml SPG buffer (220mM Sucrose, 10mM Na2HPO4, 4mM KH2PO4, 5mM Glutamic Acid) using sterile glass beads, and stored at -80°C. Sample infectious titers were quantified as described previously [25]. A similar process was used to determine the empirical infectivity of primary sample infections. Titers were normalized by this empirical infection rate for each strain within the individual experiment to exclude any effects of variation in stock aliquots.
Plaque expansion assay
Uninfected HeLa cells (~85–90% confluence) in 6-well cluster plates were covered with 1X DMEM containing 0.8% agarose, which was kept at 42°C until use). Agarose medium was then allowed to congeal at room temperature for ~15 minutes. Strains were diluted to a concentration of 107 IFU/ml. A bevelled 20μl pipette tip was briefly incubated in the inoculum and then used to puncture the gel overlay at specific sites. After inoculation, gels were overlaid with 3ml of DMEM supplemented with 15% FBS, 1μg/ml cycloheximide, 5ng/ml ATc, and 1 μg/ml gentamicin. All inoculation sites were checked for infection on day 1. Liquid overlay media were changed every 3 days. For plaque area measurement, overlay medium was carefully aspirated and the monolayers fixed by adding 10% formaldehyde in PBS to each well. After ~2 hours, gels were carefully removed and the fixed monolayers washed with PBS twice. Each well then received 0.5 ml of a 1% crystal violet in 20% ethanol solution. Monolayers were stained for ~30 minutes, prior to gentle washing of the plate in a large reservoir of ddH2O. Images of plaques were captured from beneath the well using a GelDoc-It Imaging system (Bioimaging Systems) using the exact same zoom, aparature, and focus settings for each plate. Plaques were identified using FIJI [33], by adjusting the “Threshold” and then “Analyze Particles”. For infection foci where no plaque was observed, a measurement of 1 pixel was recorded. Image in Fig 5C are from samples harvested on day 9 post-infection. Mean values in Fig 5D represent measurements from day 8 and 9 post-infection.
Immunofluorescent analysis
Fluorescent imaging for empirical infectivity and for recoverable infectious progeny titer was carried out on a Nikon Eclipse TE2000 epifluoresence microscope. For morphology and inclusion size analysis, images were captured using a Leica SP5 Resonant inverted confocal microscope, using identical settings for each sample. Montage images and scale bars were generated in FIJI. Inclusion sizes were determined in FIJI, using elipses to circumscribe each inclusion within a field of view from which the measurement tool provided the inclusion area (pixels).
Nucleic acid extractions
Genomic DNA extractions were prepared using the DNeasy Blood and Tissue Kit (QIAGEN). For RNA extraction, RNALater fixative was removed from infected monolayers and RNA was extracted by Trizol reagent (Life Technologies). RNA samples were treated with DNase (Turbo DNA-free kit, Applied Biosystems) for 1 hour, before DNase inactivation via the instructions of the manufacturer. cDNA was then generated from 2.5 μg of RNA sample using the Superscript III Reverse Transcriptase kit (Invitrogen), and diluted 1 : 8 in nuclease-free water before storage at -20°C. No Reverse Transcriptase controls were generated for all samples.
Quantitative PCR
Quantitative PCR was conducted in triplicate on the BioRad CFX96 Real-time system. Each reaction consisted of 1X Power SYBR green Mastermix, 0.45μM of each primer, and 2μl of sample in a volume of 25μl. Each run for a Chlamydia gene contained a standard curve of L2/25667R gDNA to assess amplification efficiency. NRT controls were performed for all cDNA samples. Equal amounts of total nucleic acid were loaded for each assay type (2 ng for gDNA, and the cDNA generated from 37.5 ng of RNA).
Transcript expression analysis
To control for both the number of C. trachomatis particles and for differences in reverse transcription efficiency, target gene expression was normalized by the geometric mean of an exogenous (C. trachomatis specific gDNA) and endogenous (host gapdh transcript) controls. Normalized expression was calculated as in [44], using the formula: ETarget(-Cq[Target]) / Geometric mean(2(-Cq[GAPDH]), EgDNA(-Cq[gDNA])), where E represents the amplification efficiency (e.g. 100% = 2), Cq represents the mean cycle threshold of three technical replicates for the given run (thus, error presented is biological, not technical). All cDNA samples were assayed for contaminating gDNA with No Reverse Transcriptase (NRT) controls, of which none exhibited gDNA contamination to mathematically relevant levels.
Data analysis, statistics, and graphs
Graphs were generated using GraphPad Prism v5.0f. Statistical analysis of BACTH and SPR data were also completed with GraphPad Prism software. The data sets generated for gene expression, plaque expansion size, 1-step growth, and 2-step growth were analyzed and statistical analysis performed using R. R analysis scripts are deposited available at doi: 10.6084/m9.figshare.1466906 (1-step growth, 2-step growth, gene expression analysis) and doi: 10.6084/m9.figshare.1466907 (Plaque size analysis).
Accession numbers of relevant genes and proteins
Sigma66 (rpoD / ct615; P18333); Sigma28 (fliA / ct061; O84064); Sigma54 (rpoN / ct609; O84615); RsbWCt (rsbW / ct549; O84553); RsbV1 (rsbV_1 / ct424; O84431); RsbV2 (rsbV_2 / ct765; O84770); RsbUCt (rsbU / ct588; O84592); CT589 (ct589; O84593); CT259 (ct259; O84261); 16S rRNA (ctr01 / 16S rRNA_1; 884531); HctB (hctB / ct046; Q06280); OmpA (ompA / ct681; O84605); EUO (ct446; O84452); GrgA (ct504: O84512); RsbWBs (bsu04720; P17905); RsbVBs (bsu04710; P17903); RsbTBs (bsu04690; P42411); SigmaB (bsu04730; P06574).
Supporting Information
Zdroje
1. CDC. Chlamydia trachomatis Genital Infections—United States, 1995. Morbidity and Mortality Weekly Report. 1997; 46 : 193–198. 9072679
2. Resnikoff S, Pascolini D, Etya’ale D, Kocur I, Pararajasegaram R, Pokharel GP, et al. Global data on visual impairment in the year 2002. Bull World Health Organ. 2004;82 : 844–851. 15640920
3. AbdelRahman YM, Belland RJ. The chlamydial developmental cycle. FEMS Microbiol Rev. 2005;29 : 949–959. 16043254
4. Beatty WL, Morrison RP, Byrne GI. Persistent chlamydiae: from cell culture to a paradigm for chlamydial pathogenesis. Microbiol Rev. 1994;58 : 686–699. 7854252
5. Belland RJ, Zhong G, Crane DD, Hogan D, Sturdevant D, Sharma J, et al. Genomic transcriptional profiling of the developmental cycle of Chlamydia trachomatis. Proceedings of the National Academy of Sciences. 2003;100 : 8478–8483.
6. Nicholson TL, Olinger L, Chong K, Schoolnik G, Stephens RS. Global stage-specific gene regulation during the developmental cycle of Chlamydia trachomatis. J Bacteriol. 2003;185 : 3179–3189. 12730178
7. Shaw EI, Dooley CA, Fischer ER, Scidmore MA, Fields KA, Hackstadt T. Three temporal classes of gene expression during the Chlamydia trachomatis developmental cycle. Mol Microbiol. 2000;37 : 913–925. 10972811
8. Ouellette SP, Hatch TP, AbdelRahman YM, Rose LA, Belland RJ, Byrne GI. Global transcriptional upregulation in the absence of increased translation in Chlamydia during IFNgamma-mediated host cell tryptophan starvation. Mol Microbiol. 2006;62 : 1387–1401. 17059564
9. Timms P, Good D, Wan C, Theodoropoulos C, Mukhopadhyay S, Summersgill J, et al. Differential transcriptional responses between the interferon-gamma-induction and iron-limitation models of persistence for Chlamydia pneumoniae. J Microbiol Immunol Infect. 2009;42 : 27–37. 19424556
10. Shen L, Li M, Zhang Y-X. Chlamydia trachomatis sigma28 recognizes the fliC promoter of Escherichia coli and responds to heat shock in chlamydiae. Microbiology. 2004;150 : 205–215. 14702414
11. Mathews SA, Volp KM, Peter T. Development of a quantitative gene expression assay for Chlamydia trachomatis identified temporal expression of σ factors. FEBS Lett. 1999;458 : 354–358. 10570939
12. Douglas AL, Hatch TP. Expression of the transcripts of the sigma factors and putative sigma factor regulators of Chlamydia trachomatis L2. Gene. 2000;247 : 209–214. 10773461
13. Hughes KT, Mathee K. The anti-sigma factors. Annu Rev Microbiol. 1998;52 : 231–286. 9891799
14. Stephens RS, Kalman S, Lammel C, Fan J, Marathe R, Aravind L, et al. Genome sequence of an obligate intracellular pathogen of humans: Chlamydia trachomatis. Science. 1998;282 : 754–759. 9784136
15. Humphrys MS, Todd C, Yezhou S, Shetty AC, Chibucos MC, Drabek EF, et al. Simultaneous Transcriptional Profiling of Bacteria and Their Host Cells. PLoS One. 2013;8: e80597. doi: 10.1371/journal.pone.0080597 24324615
16. Karlinsey JE, Hughes KT. Genetic transplantation: Salmonella enterica serovar Typhimurium as a host to study sigma factor and anti-sigma factor interactions in genetically intractable systems. J Bacteriol. 2006;188 : 103–114. 16352826
17. Hua L, Hefty PS, Lee YJ, Lee YM, Stephens RS, Price CW. Core of the partner switching signalling mechanism is conserved in the obligate intracellular pathogen Chlamydia trachomatis. Mol Microbiol. 2006;59 : 623–636. 16390455
18. Karimova G, Pidoux J, Ullmann A, Ladant D. A bacterial two-hybrid system based on a reconstituted signal transduction pathway. Proc Natl Acad Sci U S A. 1998;95 : 5752–5756. 9576956
19. Yang X, Kang CM, Brody MS, Price CW. Opposing pairs of serine protein kinases and phosphatases transmit signals of environmental stress to activate a bacterial transcription factor. Genes Dev. 1996;10 : 2265–2275. 8824586
20. Benson AK, Haldenwang WG. Bacillus subtilis sigma B is regulated by a binding protein (RsbW) that blocks its association with core RNA polymerase. Proc Natl Acad Sci U S A. 1993;90 : 2330–2334. 8460143
21. Wang Y, Kahane S, Cutcliffe LT, Skilton RJ, Lambden PR, Clarke IN. Development of a transformation system for Chlamydia trachomatis: restoration of glycogen biosynthesis by acquisition of a plasmid shuttle vector. PLoS Pathog. 2011;7: e1002258. doi: 10.1371/journal.ppat.1002258 21966270
22. Fagan RP, Fairweather NF. Clostridium difficile has two parallel and essential Sec secretion systems. J Biol Chem. 2011;286 : 27483–27493. doi: 10.1074/jbc.M111.263889 21659510
23. Johnson CM, Fisher DJ. Site-specific, insertional inactivation of incA in Chlamydia trachomatis using a group II intron. PLoS One. 2013;8: e83989. doi: 10.1371/journal.pone.0083989 24391860
24. Mäurer AP, Mehlitz A, Mollenkopf HJ, Meyer TF. Gene expression profiles of Chlamydophila pneumoniae during the developmental cycle and iron depletion-mediated persistence. PLoS Pathog. 2007;3: e83. 17590080
25. Thompson CC, Carabeo RA. An optimal method of iron starvation of the obligate intracellular pathogen, Chlamydia trachomatis. Front Microbiol. 2011;2 : 20. doi: 10.3389/fmicb.2011.00020 21687412
26. Tan M, Gaal T, Gourse RL, Engel JN. Mutational analysis of the Chlamydia trachomatis rRNA P1 promoter defines four regions important for transcription in vitro. J Bacteriol. 1998;180 : 2359–2366. 9573186
27. Tan M, Engel JN. Identification of sequences necessary for transcription in vitro from the Chlamydia trachomatis rRNA P1 promoter. J Bacteriol. 1996;178 : 6975–6982. 8955322
28. Douglas AL, Saxena NK, Hatch TP. Enhancement of in vitro transcription by addition of cloned, overexpressed major sigma factor of Chlamydia psittaci 6BC. J Bacteriol. 1994;176 : 4196. 8021204
29. Stephens RS, Wagar EA, Edman U. Developmental regulation of tandem promoters for the major outer membrane protein gene of Chlamydia trachomatis. J Bacteriol. 1988;170 : 744–750. 2448291
30. Yu HHY, Kibler D, Tan M. In silico prediction and functional validation of sigma28-regulated genes in Chlamydia and Escherichia coli. J Bacteriol. 2006;188 : 8206–8212. 16997971
31. Rosario CJ, Hanson BR, Tan M. The transcriptional repressor EUO regulates both subsets of Chlamydia late genes. Mol Microbiol. 2014;94 : 888–897. doi: 10.1111/mmi.12804 25250726
32. Rosario CJ, Tan M. The early gene product EUO is a transcriptional repressor that selectively regulates promoters of Chlamydia late genes. Mol Microbiol. 2012;84 : 1097–1107. doi: 10.1111/j.1365-2958.2012.08077.x 22624851
33. Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, et al. Fiji: an open-source platform for biological-image analysis. Nat Methods. 2012;9 : 676–682. doi: 10.1038/nmeth.2019 22743772
34. Alper S, Dufour A, Garsin DA, Duncan L, Losick R. Role of adenosine nucleotides in the regulation of a stress-response transcription factor in Bacillus subtilis. J Mol Biol. 1996;260 : 165–177. 8764398
35. Locke JCW, Young JW, Fontes M, Hernández Jiménez MJ, Elowitz MB. Stochastic pulse regulation in bacterial stress response. Science. 2011;334 : 366–369. doi: 10.1126/science.1208144 21979936
36. Rao X, Deighan P, Hua Z, Hu X, Wang J, Luo M, et al. A regulator from Chlamydia trachomatis modulates the activity of RNA polymerase through direct interaction with the beta subunit and the primary sigma subunit. Genes Dev. 2009;23 : 1818–1829. doi: 10.1101/gad.1784009 19651989
37. Jishage M, Ishihama A. Transcriptional organization and in vivo role of the Escherichia coli rsd gene, encoding the regulator of RNA polymerase sigma D. J Bacteriol. 1999;181 : 3768–3776. 10368152
38. Bao X, Nickels BE, Fan H. Chlamydia trachomatis protein GrgA activates transcription by contacting the nonconserved region of σ66. Proc Natl Acad Sci U S A. 2012;109 : 16870–16875. doi: 10.1073/pnas.1207300109 23027952
39. Agaisse H, Hervé A, Isabelle D. A C. trachomatis Cloning Vector and the Generation of C. trachomatis Strains Expressing Fluorescent Proteins under the Control of a C. trachomatis Promoter. PLoS One. 2013;8: e57090. doi: 10.1371/journal.pone.0057090 23441233
40. Miller. JH. A short course in bacterial genetics. Plainview, N.Y.:: Cold Spring Harbor Laboratory Press; 1992.
41. Shun M-C, Raghavendra NK, Vandegraaff N, Daigle JE, Hughes S, Kellam P, et al. LEDGF/p75 functions downstream from preintegration complex formation to effect gene-specific HIV-1 integration. Genes Dev. 2007;21 : 1767–1778. 17639082
42. Kelley LA, Sternberg MJE. Protein structure prediction on the Web: a case study using the Phyre server. Nat Protoc. 2009;4 : 363–371. doi: 10.1038/nprot.2009.2 19247286
43. Song L, Carlson JH, Whitmire WM, Kari L, Virtaneva K, Sturdevant DE, et al. Chlamydia trachomatis plasmid-encoded Pgp4 is a transcriptional regulator of virulence-associated genes. Infect Immun. 2013;81 : 636–644. doi: 10.1128/IAI.01305-12 23319558
44. Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002;3: RESEARCH0034.
Štítky
Hygiena a epidemiologie Infekční lékařství Laboratoř
Článek The Hos2 Histone Deacetylase Controls Virulence through Direct Regulation of Mating-Type GenesČlánek Transgenic Mouse Bioassay: Evidence That Rabbits Are Susceptible to a Variety of Prion IsolatesČlánek -Associated Polyomavirus Uses a Displaced Binding Site on VP1 to Engage Sialylated GlycolipidsČlánek Parsimonious Determination of the Optimal Infectious Dose of a Pathogen for Nonhuman Primate Models
Článek vyšel v časopisePLOS Pathogens
Nejčtenější tento týden
2015 Číslo 8- Jak souvisí postcovidový syndrom s poškozením mozku?
- Farmakovigilanční studie perorálních antivirotik indikovaných v léčbě COVID-19
- Měli bychom postcovidový syndrom léčit antidepresivy?
- 10 bodů k očkování proti COVID-19: stanovisko České společnosti alergologie a klinické imunologie ČLS JEP
-
Všechny články tohoto čísla
- The Long and Winding Road (Apologies to the Beatles)
- The Ebola Virus: From Basic Research to a Global Health Crisis
- Riding the R Train into the Cell
- The Two-Phase Emergence of Non Pandemic HIV-1 Group O in Cameroon
- Tumor Progression Locus 2 Promotes Induction of IFNλ, Interferon Stimulated Genes and Antigen-Specific CD8 T Cell Responses and Protects against Influenza Virus
- Type VI Secretion System Toxins Horizontally Shared between Marine Bacteria
- Incomplete Neutralization and Deviation from Sigmoidal Neutralization Curves for HIV Broadly Neutralizing Monoclonal Antibodies
- E3 Ubiquitin Ligase NEDD4 Promotes Influenza Virus Infection by Decreasing Levels of the Antiviral Protein IFITM3
- The Hos2 Histone Deacetylase Controls Virulence through Direct Regulation of Mating-Type Genes
- Hyperinvasive Meningococci Induce Intra-nuclear Cleavage of the NF-κB Protein p65/RelA by Meningococcal IgA Protease
- Active Transport of Phosphorylated Carbohydrates Promotes Intestinal Colonization and Transmission of a Bacterial Pathogen
- HTLV-1 Tax Stimulates Ubiquitin E3 Ligase, Ring Finger Protein 8, to Assemble Lysine 63-Linked Polyubiquitin Chains for TAK1 and IKK Activation
- Transgenic Mouse Bioassay: Evidence That Rabbits Are Susceptible to a Variety of Prion Isolates
- Widespread Reassortment Shapes the Evolution and Epidemiology of Bluetongue Virus following European Invasion
- Inhibiting the Recruitment of PLCγ1 to Kaposi’s Sarcoma Herpesvirus K15 Protein Reduces the Invasiveness and Angiogenesis of Infected Endothelial Cells
- Goblet Cell Derived RELM-β Recruits CD4 T Cells during Infectious Colitis to Promote Protective Intestinal Epithelial Cell Proliferation
- HLA Class-II Associated HIV Polymorphisms Predict Escape from CD4+ T Cell Responses
- An siRNA Screen Identifies the U2 snRNP Spliceosome as a Host Restriction Factor for Recombinant Adeno-associated Viruses
- Extracellular Adenosine Protects against Lung Infection by Regulating Pulmonary Neutrophil Recruitment
- : Adaptations to the Dixenous Life Cycle Analyzed by Genome Sequencing, Transcriptome Profiling and Co-infection with
- Which Way In? The RalF Arf-GEF Orchestrates Host Cell Invasion
- Intracellular Uropathogenic . Exploits Host Rab35 for Iron Acquisition and Survival within Urinary Bladder Cells
- A Non-enveloped Virus Hijacks Host Disaggregation Machinery to Translocate across the Endoplasmic Reticulum Membrane
- Supporting Role for GTPase Rab27a in Hepatitis C Virus RNA Replication through a Novel miR-122-Mediated Effect
- -Associated Polyomavirus Uses a Displaced Binding Site on VP1 to Engage Sialylated Glycolipids
- The Activation of Effector Avr3b by Plant Cyclophilin is Required for the Nudix Hydrolase Activity of Avr3b
- A Pyranose-2-Phosphate Motif Is Responsible for Both Antibiotic Import and Quorum-Sensing Regulation in
- Double-Edge Sword of Sustained ROCK Activation in Prion Diseases through Neuritogenesis Defects and Prion Accumulation
- The Rsb Phosphoregulatory Network Controls Availability of the Primary Sigma Factor in and Influences the Kinetics of Growth and Development
- Inhibits Virulence through Suppression of Pyochelin and Pyoverdine Biosynthesis
- Illuminating Targets of Bacterial Secretion
- Chemical Signals and Mechanosensing in Bacterial Responses to Their Environment
- Interdisciplinarity and Infectious Diseases: An Ebola Case Study
- Fungi That Infect Insects: Altering Host Behavior and Beyond
- Plasticity and Redundancy in Proteins Important for Invasion
- Are Human Intestinal Eukaryotes Beneficial or Commensals?
- A Novel Virus Causes Scale Drop Disease in
- STAT2 Knockout Syrian Hamsters Support Enhanced Replication and Pathogenicity of Human Adenovirus, Revealing an Important Role of Type I Interferon Response in Viral Control
- Parsimonious Determination of the Optimal Infectious Dose of a Pathogen for Nonhuman Primate Models
- Twenty-Eight Years of Poliovirus Replication in an Immunodeficient Individual: Impact on the Global Polio Eradication Initiative
- AAV-Delivered Antibody Mediates Significant Protective Effects against SIVmac239 Challenge in the Absence of Neutralizing Activity
- Interferon-γ Promotes Inflammation and Development of T-Cell Lymphoma in HTLV-1 bZIP Factor Transgenic Mice
- Transgenic Rabbits Expressing Ovine PrP Are Susceptible to Scrapie
- Mitochondrial Activity and Cyr1 Are Key Regulators of Ras1 Activation of . Virulence Pathways
- Human Non-neutralizing HIV-1 Envelope Monoclonal Antibodies Limit the Number of Founder Viruses during SHIV Mucosal Infection in Rhesus Macaques
- Serine Phosphorylation of HIV-1 Vpu and Its Binding to Tetherin Regulates Interaction with Clathrin Adaptors
- Inhibition of mTORC1 Enhances the Translation of Chikungunya Proteins the Activation of the MnK/eIF4E Pathway
- Nanoformulations of Rilpivirine for Topical Pericoital and Systemic Coitus-Independent Administration Efficiently Prevent HIV Transmission
- Arming of MAIT Cell Cytolytic Antimicrobial Activity Is Induced by IL-7 and Defective in HIV-1 Infection
- sRNA-Mediated Regulation of P-Fimbriae Phase Variation in Uropathogenic
- Evolutionary and Functional Analysis of Old World Primate TRIM5 Reveals the Ancient Emergence of Primate Lentiviruses and Convergent Evolution Targeting a Conserved Capsid Interface
- Hepcidin and Host Defense against Infectious Diseases
- Type I IFN Induction via Poly-ICLC Protects Mice against Cryptococcosis
- Mucosal B Cells Are Associated with Delayed SIV Acquisition in Vaccinated Female but Not Male Rhesus Macaques Following SIV Rectal Challenge
- PLOS Pathogens
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Human Non-neutralizing HIV-1 Envelope Monoclonal Antibodies Limit the Number of Founder Viruses during SHIV Mucosal Infection in Rhesus Macaques
- Type VI Secretion System Toxins Horizontally Shared between Marine Bacteria
- Illuminating Targets of Bacterial Secretion
- Are Human Intestinal Eukaryotes Beneficial or Commensals?
Přihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



