-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Top novinky
ReklamaFine-Mapping the Region Detects Common Variants Tagging a Rare Coding Allele: Evidence for Synthetic Association in Prostate Cancer
The HOXB13 gene has been implicated in prostate cancer (PrCa) susceptibility. We performed a high resolution fine-mapping analysis to comprehensively evaluate the association between common genetic variation across the HOXB genetic locus at 17q21 and PrCa risk. This involved genotyping 700 SNPs using a custom Illumina iSelect array (iCOGS) followed by imputation of 3195 SNPs in 20,440 PrCa cases and 21,469 controls in The PRACTICAL consortium. We identified a cluster of highly correlated common variants situated within or closely upstream of HOXB13 that were significantly associated with PrCa risk, described by rs117576373 (OR 1.30, P = 2.62×10−14). Additional genotyping, conditional regression and haplotype analyses indicated that the newly identified common variants tag a rare, partially correlated coding variant in the HOXB13 gene (G84E, rs138213197), which has been identified recently as a moderate penetrance PrCa susceptibility allele. The potential for GWAS associations detected through common SNPs to be driven by rare causal variants with higher relative risks has long been proposed; however, to our knowledge this is the first experimental evidence for this phenomenon of synthetic association contributing to cancer susceptibility.
Published in the journal: . PLoS Genet 10(2): e32767. doi:10.1371/journal.pgen.1004129
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1004129Summary
The HOXB13 gene has been implicated in prostate cancer (PrCa) susceptibility. We performed a high resolution fine-mapping analysis to comprehensively evaluate the association between common genetic variation across the HOXB genetic locus at 17q21 and PrCa risk. This involved genotyping 700 SNPs using a custom Illumina iSelect array (iCOGS) followed by imputation of 3195 SNPs in 20,440 PrCa cases and 21,469 controls in The PRACTICAL consortium. We identified a cluster of highly correlated common variants situated within or closely upstream of HOXB13 that were significantly associated with PrCa risk, described by rs117576373 (OR 1.30, P = 2.62×10−14). Additional genotyping, conditional regression and haplotype analyses indicated that the newly identified common variants tag a rare, partially correlated coding variant in the HOXB13 gene (G84E, rs138213197), which has been identified recently as a moderate penetrance PrCa susceptibility allele. The potential for GWAS associations detected through common SNPs to be driven by rare causal variants with higher relative risks has long been proposed; however, to our knowledge this is the first experimental evidence for this phenomenon of synthetic association contributing to cancer susceptibility.
Introduction
Prostate cancer (PrCa) is the most common cancer affecting men in developed countries, accounting for 25% of cancer diagnoses among males in the UK in 2010 (http://www.cancerresearchuk.org/cancer-info/cancerstats/types/prostate/incidence/). Whilst the majority of men will develop some form of prostate neoplasm during their lifetime, these are usually slow progressing and remain asymptomatic until their death; therefore only a proportion of prostate tumours require clinical intervention [1]. Currently, prostate specific antigen (PSA) is the only available biomarker for PrCa, however the specificity of this test for clinically significant disease is poor and its use for PrCa screening remains controversial; with little evidence of significant reduction in mortality and at the cost of substantial overdiagnosis and overtreatment of patients [2], [3]. Accordingly, much recent research has attempted to improve identification of individuals at greater risk of developing prostate tumours that require clinical intervention, to enable better application of treatment. Twin studies have suggested that PrCa has a substantial heritable component [4], whilst family history of PrCa among first degree relatives remains among the strongest known risk factors for the disease [5], [6]. As a result, many studies have looked for genetic variants that predispose towards the development of PrCa. Relatively few moderate penetrance risk variants for PrCa have been identified so far; however more than 70 common, low penetrance variants that individually modestly increase risk have been identified to date through GWAS studies [7], [8].
We recently reported 23 novel PrCa susceptibility SNPs identified through genotyping 20,440 PrCa cases and 21,469 controls from the PRACTICAL consortium on a custom Illumina iSelect array (iCOGS) [7]. These SNPs were all from loci that had shown some evidence for association with PrCa in our initial GWAS [9]; however the iCOGS array also contained a subset of SNPs that were included to examine possible associations with plausible PrCa susceptibility candidate genes. HOX genes are known to have crucial roles in development and previous evidence suggested their potential involvement in oncogenesis [10], including HOXB13 specifically in PrCa [11], [12]. In addition, the HOXB13 locus at chr17q21 was a region that had been previously implicated in PrCa susceptibility by linkage studies [13]–[15] and had been shown to undergo loss of heterozygosity in prostate tumours [16]–[18]. As a result, we targeted the HOXB cluster at chr17q21 to be densely genotyped on the iCOGS array. In addition, a closely situated ovarian cancer risk association around the SKAP1 gene [19] had also been targeted for fine-mapping by the Ovarian Cancer Consortium (OCAC), providing additional SNPs covering this chromosomal region.
Whilst genotyping on the iCOGS array was being performed, Ewing et al. published evidence that a rare non-synonymous coding variant in HOXB13 (G84E, rs138213197) was associated with hereditary prostate cancer (HPC) [20]. This risk variant has subsequently been confirmed to be a moderate penetrance susceptibility allele in a number of other studies and it was shown that the association was strongest with younger onset and familial PrCa [21]–[23]. Additional studies examining the geographical spread of the G84E variant have determined that it is observed almost exclusively in Caucasians and predominantly on the same haplotype background. This haplotype occurs more frequently in Nordic countries, most strikingly within the Finnish population, and suggests that rs138213197 is a founder mutation that arose relatively recently in Northern Europe [24], [25].
In this study, we show evidence that there is a cluster of novel common, low penetrance PrCa risk alleles in the HOXB region which appear to tag the rarer, moderate penetrance coding variant rs138213197. To our knowledge, this represents the first identified occurrence of a synthetic association in cancer.
Results
After QC, 700 SNPs from the interval Chr17 : 46201311–47382559 (GRCh37/hg19) encompassing the HOXB locus were analysed on the iCOGS array in 20,440 cases and 21,469 controls of European ancestry from the PRACTICAL consortium. Two panel imputation was performed for the interval Chr17 : 46200000–47400000 using a 1000 Genomes Phase 1 integrated variant set and Illumina OMNI2.5 BeadChip data for 677 PrCa cases from the UKGPCS study. This generated imputed data for 3195 SNPs with MAF≥0.01 within this region in the iCOGS sample set. These thresholds do not retain imputation information for the previously reported coding variant (G84E, rs138213197) due to its low MAF. As this SNP is a reported PrCa susceptibility variant and was in close proximity to a cluster of variants showing association with PrCa risk in our imputed data, additional genotyping was carried out by Taqman and Sequenom assays for this SNP in 5500 cases and 4923 controls from the UK and Sweden. We subsequently attempted to impute the rs138213197 variant to the entire iCOGS sample set using this additional panel, however the imputation quality remained inadequate and therefore analyses involving this variant were performed on the directly genotyped subset of samples only.
Following imputation, four SNPs in close proximity to one another and situated within or closely upstream of the HOXB13 gene remained significantly associated with PrCa at P<10−6 (Table 1); of which one, rs117576373, had been genotyped on the iCOGS array. This cluster of variants are highly correlated (r2≥0.79) and envelop the published missense coding variant rs138213197 (G84E) (Figure 1). In the subset of samples that had been genotyped for rs138213197, the correlation between rs138213197 and the cluster of variants represented by rs117576373 initially appears very modest (r2≤0.13, Figure 1), implying that the newly identified cluster of variants represented a novel association signal. However, the MAFs of these variants are substantially different (2.6–4.3% vs. 0.4% in our control set), with the rare allele of rs138213197 (T) almost exclusively co-inherited with the minor allele of rs117576373 (T) (D′ 0.98). The nature of the correlation between these variants could therefore be consistent with rs117576373 representing an additional novel, common, lower penetrance association signal at the HOXB13 region that is almost invariably present alongside the moderate penetrance rs138213197 variant but is also found by itself in a greater number of individuals. Alternatively, in spite of the low r2 between the two variants, the novel association signal could in fact be tagging the rare G84E variant, thereby resulting in detection of a synthetic association signal at a common variant that is in fact mediated by a much rarer causal variant.
Fig. 1. Results of the HOXB locus fine-mapping analysis. 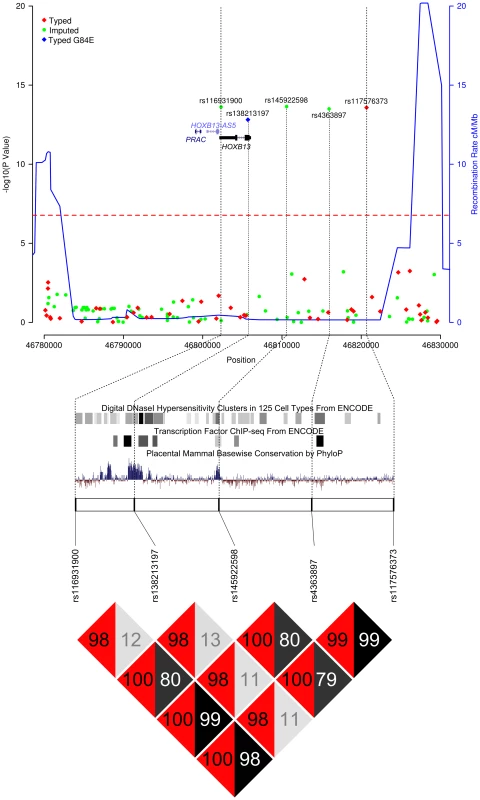
Upper Panel – Regional association plot of SNPs at the HOXB13 locus. Association data from the iCOGS dataset of 20,440 PrCa cases and 21,469 controls are shown with genotyped SNPs in red and imputed SNPs in green. The Bonferroni-adjusted level of significance is denoted by the red line. The G84E variant rs138213197 was genotyped in a smaller subset of 5500 PrCa cases and 4923 controls and is marked by the blue rectangle. Also indicated are the position of genes within this interval and the location of neighbouring recombination hotspots. Middle Panel – Intersection between the 5 SNPs significantly associated with PrCa and putative functional elements identified by the ENCODE project or regions of mammalian sequence conservation by PhyloP. Lower Panel – Pairwise Linkage Disequilibrium values for the 5 SNPs significantly associated with PrCa. r2 values are shown in grey and D′ in red. Tab. 1. PrCa association information for SNPs significant at P<10−6 in the iCOGS imputed data. 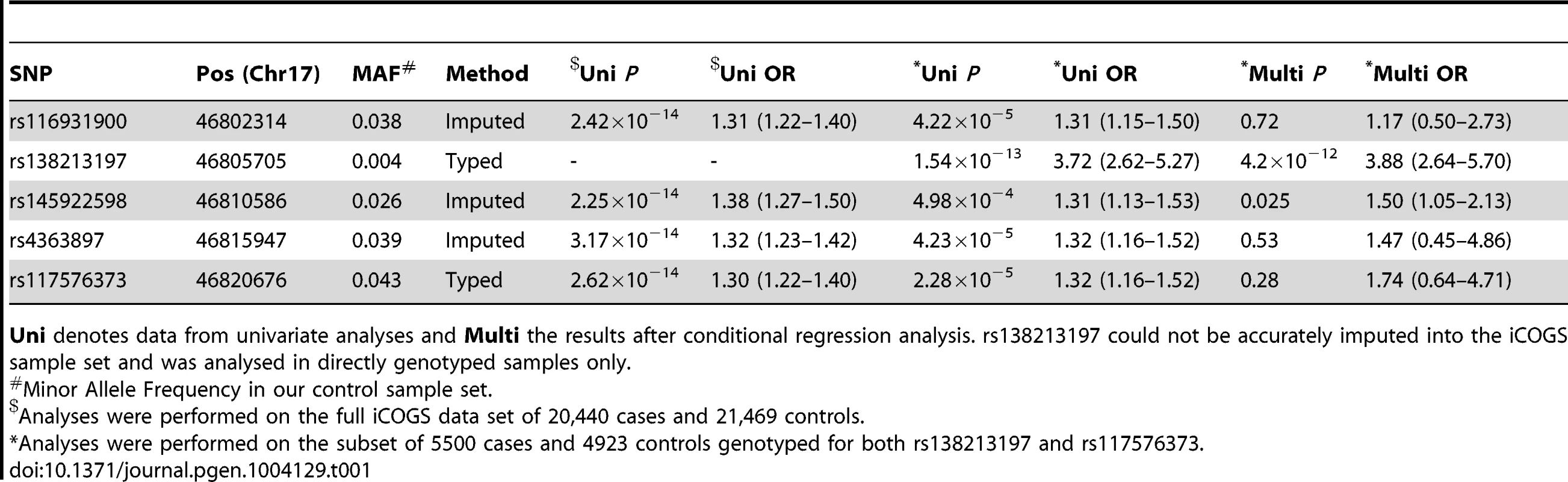
Uni denotes data from univariate analyses and Multi the results after conditional regression analysis. rs138213197 could not be accurately imputed into the iCOGS sample set and was analysed in directly genotyped samples only. To elucidate which of these scenarios explain the PrCa risk association at this locus we first performed a conditional regression analysis for rs138213197 and the cluster of newly identified variants, using the subset of cases that had been genotyped for both rs138213197 and rs117576373. rs138213197 remained highly significant in this analysis (P = 4.2×10−12, Table 1) and with an effect size substantially greater than that observed through any of the common variants and broadly similar to that previously reported in the literature (OR = 3.88, 95%C.I. 2.64–5.70). This suggests that the association with PrCa risk arises predominantly through this rare coding variant. In addition, one of the more common SNPs, rs145922598, also exhibited some evidence for association (P = 0.025). This SNP is somewhat less frequent than the other three SNPs in the newly identified cluster, however is still highly correlated with these (r2∼0.8, D′∼1) and is located in a region of high conservation and functional context (Figure 1); therefore could potentially represent a novel low penetrance association signal. To further examine whether these variants represent the same or separate association signals, in the subset of samples in which the coding variant had been directly genotyped, we conducted haplotype analyses between rs138213197 and rs117576373 (both directly genotyped) and additionally between rs138213197 and rs145922598 (imputed). This provides further confirmation that rs138213197 is most likely responsible for PrCa risk alone; since the moderately frequent rs138213197 (C, major allele)–rs117576373 (T, minor allele) and rs138213197 (C, major allele)–rs145922598 (T, minor allele) haplotypes showed no evidence for association with disease risk, and a positive association with risk was only observed in haplotypes where the rs138213197 (T, minor allele) risk allele was present (Tables 2 & 3).
Tab. 2. Haplotype analysis for rs117576373 and rs138213197 in the subset of 5500 PrCa cases and 4923 controls from the UK and Sweden for which both had been directly genotyped. 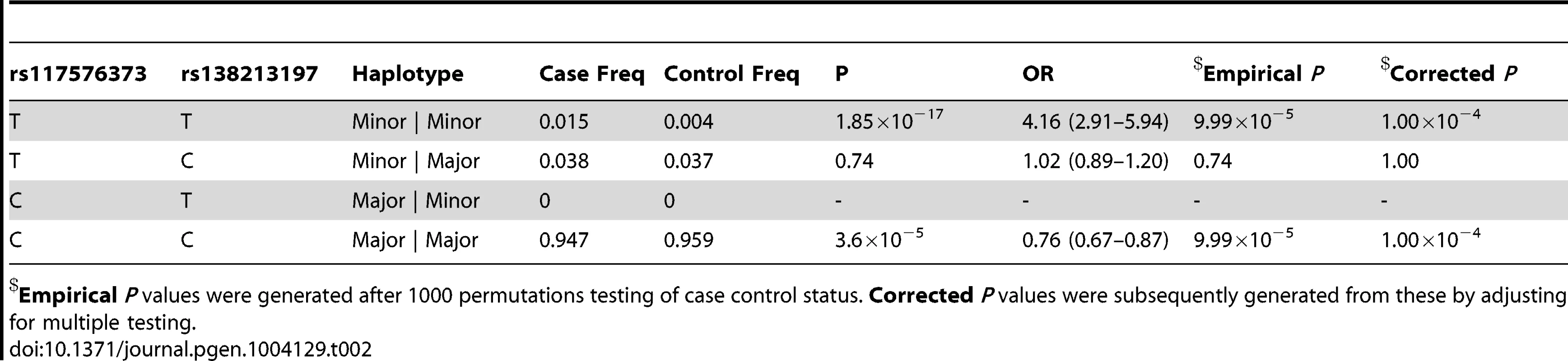
Empirical P values were generated after 1000 permutations testing of case control status. Corrected P values were subsequently generated from these by adjusting for multiple testing. Tab. 3. Haplotype analysis for rs145922598 and rs138213197 in the subset of 5500 PrCa cases and 4923 controls from the UK and Sweden genotyped for rs138213197. 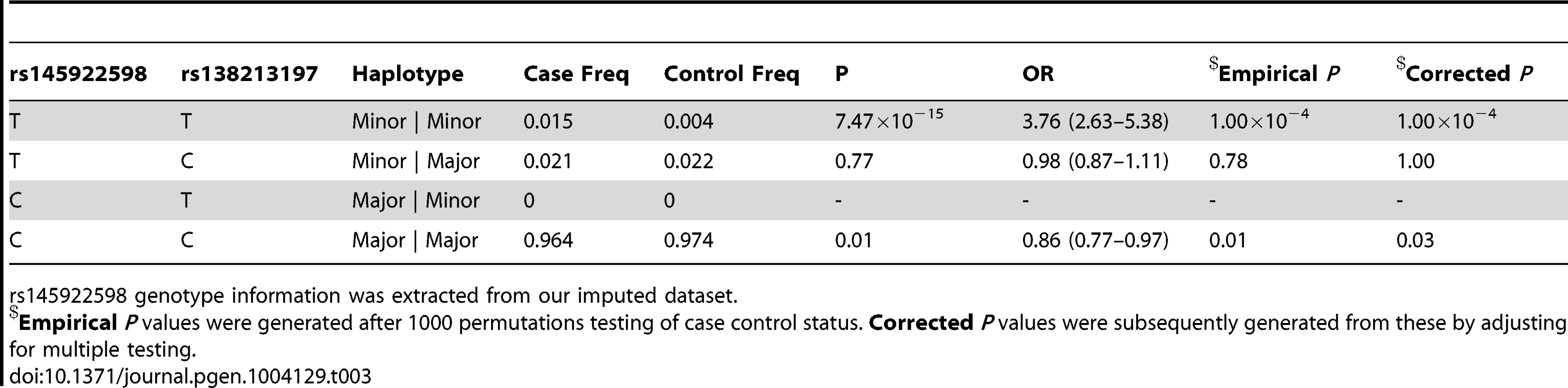
rs145922598 genotype information was extracted from our imputed dataset. In addition to this novel association signal, we also confirmed a previously reported association within our imputation interval described by rs11650494 in Caucasians [7] and rs7210100 in African Americans [26]. This signal is situated >500 kb downstream of the novel variant cluster (Supplementary Figure S1), around the ZNF652 gene. We observed no significant linkage disequilibrium between these two clusters of variants (r2 = 0, D′≈0.02) and our conditional analysis confirmed that they represent separate associations with PrCa.
Discussion
In this study, we identified a novel common PrCa association signal at the HOXB13 locus. Further investigation revealed that this signal is most likely to arise due to correlation with the previously reported rare, moderate penetrance coding variant rs138213197. Despite the fact that in this instance the rare, putative causal variant was discovered prior to that of the more common tag SNP, this PrCa susceptibility locus still serves as a useful illustration of the potential range of causal variation underpinning GWAS association signals, as well as the potential pitfalls of attempting to elucidate candidate causal variants.
rs138213197 was discovered through re-sequencing of a PrCa linkage hit at chr17q21 in hereditary prostate cancer (HPC) families; although as we show here, it could instead have been indirectly detected through the modestly correlated variant rs117576373 in a suitably powered GWAS study with sufficient marker density such as the iCOGS study. Due to the large difference in allele frequency between the tag and causal SNPs, and the inability to accurately impute the rare variant even with a priori knowledge of its existence, discovering this susceptibility locus by this route would therefore disguise the contribution of the rare variant and artificially diminish the observed relative risk; which would consequently have implicated the likely causal variant(s) to be relatively common, low penetrance and tightly correlated with the typed SNP rs117576373. However, while we cannot completely exclude that common variation may contribute to PrCa risk at this locus as data from the ENCODE project suggests some degree of potential functionality for the variants we have identified here, (in particular rs145922598, which remained marginally significant in the conditional regression, is highly conserved, overlaps a DNaseI hypersensitivity site in several cell lines including LNCaP and transcription factor binding sites for FOXA1 and FOXA2 transcription factors (Table 1, Figure 1)); the much stronger evidence for significance for the rare coding variant coupled with the results of our haplotype analyses appear to indicate that this SNP is solely responsible for the detected association signal. As such, this appears to be an example of a rare variant with a sufficiently large effect size to create a synthetic association signal detected through partially correlated yet significantly more common variants. It is also worth noting that had the rs138213197 variant not been previously identified as a PrCa susceptibility variant, it would have been unlikely to have been discovered during this imputation based fine-mapping approach since the MAF of this SNP is below conventional QC thresholds for imputation; indeed, it remains poorly imputed even using a two panel method in which a subset of samples had been directly genotyped for this variant. The potential consequence of this inability to accurately impute low frequency variants is that the search for candidate causal variants for functional follow-up would be inevitably skewed towards common variation. Furthermore, as we have observed here, where rare causal variants underpin an association signal, risk effect size estimates may consequently be significantly underestimated and the assumed proportion of carriers of the causal variant inflated. Our observations therefore provide support for the suggestion that identifying the actual causal variants behind GWAS associations could account for a proportion of the missing heritability in common diseases and that re-sequencing of GWAS loci in large numbers of cases and controls would be important for the discovery of the full spectrum of correlated variation.
The nature of the underlying genetic architecture behind GWAS signals has been the subject of much debate. Whilst few causal alleles have been unambiguously categorised, several authors have presented evidence that suggests common variants are likely to comprise the vast majority of these [27]–[29]. Conversely, computational analyses have demonstrated that rare causal variation has the potential capability to give rise to the GWAS signals detected through more common variants [30]–[32]. For PrCa, fine-mapping and functional evidence at a handful of risk loci appears to implicate common SNPs as the most likely candidate causal variants at these regions. For example, at the MSMB region at chr10q11, the common GWAS tag SNP situated in the MSMB promoter remained the most plausible candidate causal variant after fine-mapping by sequencing [33], with functional studies also demonstrating that the risk allele disrupts a transcription factor binding site, resulting in decreased expression of MSMB [34]. We have also performed fine-mapping studies by imputation for the KLK region at chr19q13 and TERT locus at chr5p15. We identified a common missense coding SNP in the KLK3 gene that was more strongly associated with PrCa than the original tag SNP and represents a candidate causal variant for this association [35], whilst four independently associated clusters of common variants were described at the TERT locus [36]; however these studies were not powered to detect any contribution by rare variants and despite refining the original associations, have not unambiguously established the causal allele(s) at these regions. This study therefore provides the first direct evidence of which we are aware for a substantial contribution of rare variation to an association signal for PrCa. This suggests that it is entirely plausible that both mechanisms may indeed give rise to GWAS associations, and need not necessarily be mutually exclusive. Logically however, the higher the MAF of the tag SNP at a susceptibility locus the greater the likelihood that the associated causal variant(s) will be common, whilst synthetic associations would become increasingly plausible at lower index SNP MAFs (in this study 2.6–4.3% MAF). Furthermore, whilst this study does provide experimental evidence for the existence of synthetic associations, no inference can be made as to how frequently they might account for the causal variant behind the numerous disease associations that GWAS have discovered. However, by capitalising upon the differences in genetic architecture between different ethnic populations in addition to the steadily increasing quantities of sequencing data that are becoming available to the research community, this may become more clear and help to guide future fine-mapping studies. In particular, as the causal alleles behind synthetic associations are rare, these associations are more likely to be limited to specific ethnic groups and therefore the absence of a multi-ethnic signal for a tag SNP of modest frequency could indicate a greater likelihood that re-sequencing the locus would identify rare causal variation.
In summary, this study provides evidence for several widely discussed concepts regarding the nature of causal variation at GWAS hits and their contribution to the heritability of common diseases. Firstly, we have shown that low frequency, moderate penetrance susceptibility variants can be detected via common tag SNPs in GWAS studies when there is little recombination between these variants. Secondly, that imputation based fine mapping alone is likely to implicate candidate causal variants as common, some of which may have plausible biological function; therefore sufficiently powered re-sequencing of loci is ultimately desirable to assess and possibly exclude the contribution of rare variants. Finally, that for GWAS associations where the tag SNP is correlated with a rare causal variant, the relative risk estimates derived from the tag SNP are likely to be considerably underestimated, which could in turn account for a proportion of the missing heritability of common diseases.
Methods
Samples
Samples for the iCOGS study were drawn from 25 studies participating in the PRACTICAL Consortium [7]. The majority of studies were population-based or hospital-based case-control studies, or nested case-control studies. All studies have the relevant IRB approval in each country in accordance with the principles embodied in the Declaration of Helsinki. After exclusion of samples that failed quality control (QC) in the iCOGS study or showed substantial non-European ancestry, genotype data for 20,440 PrCa cases and 21,469 matched controls were available.
To improve imputation performance, Illumina OMNI2.5 SNP array data were available for 677 UK PrCa cases from the UKGPCS study (www.icr.ac.uk/ukgpcs); 262 of these cases were also genotyped on the iCOGS array. The rare coding variant rs138213197 was also genotyped in 2476 cases and 2198 controls from the UK (UKGPCS study), and 3024 cases and 2725 controls from Sweden (CAPS and STHM1 study).
Genotyping
Detailed information relating to the custom iCOGS Illumina Infinium array can be found in Eeles et al., 2013 [7]. With respect to the HOXB locus, 747 SNPs spanning the interval chr17 : 46201311–47382559 were genotyped on the iCOGS array, submitted by a combination of the PRACTICAL and OCAC consortia (Supplementary Figure S1).
To boost imputation performance, additional genotyping of 677 PrCa cases from the UK was conducted using the Illumina (San Diego, CA, USA) OMNI2.5 BeadChip according to the manufacturer's instructions. Further genotyping of the rs138213197 variant was carried out by Taqman assay (Applied Biosystems Inc., Foster City, CA, USA) for 2476 cases and 2198 controls from the UK and by MassARRAY iPLEX (Sequenom Inc., San Diego, CA, USA) for 3024 cases and 2725 controls from Sweden.
Imputation
Imputation was performed on 20,440 case and 21,469 control samples across the 700 iCOGS SNPs from the HOXB13 interval which passed pre-imputation QC metrics [37] (Supplementary Figure S2). IMPUTE v2.3.0 [38], [39] was used to impute the interval Chr17 : 46200000–47400000 (GRCh37/hg19). Two panel imputation [40] was performed using OMNI2.5 BeadChip data for 677 PrCa cases from the UKGPCS study (Panel 1) and a 1000 Genomes Phase 1 integrated variant set “version 3” (SNPs and in/dels) from 5th March 2012 (Panel 0) (http://mathgen.stats.ox.ac.uk/impute/data_download_1000G_phase1_integrated.html). Imputation concordance was examined using “leave one out” internal concordance check. For single panel imputation (Panel 0 only) concordance was 96.5% at SNPs r2≥0.5 and 98.0% at r2≥0.9, which rose to 98.9% at r2≥0.5 and 99.8% at r2≥0.9 respectively for two panel (Panel 0+1) imputation. SNPs with info <0.5, MAF<0.01 were excluded during QC filtering.
Statistical Analysis
Association tests were performed on genotypes in the MaCH dosage format (0–2) converted from the IMPUTE genotype posterior probabilities using GenABEL [41]. Associations between each SNP and PrCa risk were analysed using a per-allele trend test, adjusted for study and six principal components derived from analysis of the whole iCOGS dataset [7]. Odds ratios (OR) and 95% confidence limits were estimated using unconditional logistic regression. Tests of homogeneity of the ORs across strata were assessed using a likelihood ratio test. SNPs significant at P<10−6 were considered for further analysis. The independence of these associations was assessed by performing a conditional logistic regression analysis. For further assessment of the relationship between rs138213197 and rs117576373, haplotype analyses were performed with Plink 1.07 (http://pngu.mgh.harvard.edu/purcell/plink/) on the subset of samples where both SNPs had been directly genotyped. Haplotypes were first imputed using the Expectation-Maximisation algorithm in order to then perform a case-control association, using 10,000 permutations of the phenotype labels [42].
Supporting Information
Zdroje
1. RuijterE, van de KaaC, MillerG, RuiterD, DebruyneF, et al. (1999) Molecular genetics and epidemiology of prostate carcinoma. Endocr Rev 20 : 22–45.
2. DjulbegovicM, BeythRJ, NeubergerMM, StoffsTL, ViewegJ, et al. (2010) Screening for prostate cancer: systematic review and meta-analysis of randomised controlled trials. BMJ 341: c4543.
3. VickersAJ, RoobolMJ, LiljaH (2012) Screening for prostate cancer: early detection or overdetection? Annu Rev Med 63 : 161–170.
4. LichtensteinP, HolmNV, VerkasaloPK, IliadouA, KaprioJ, et al. (2000) Environmental and heritable factors in the causation of cancer–analyses of cohorts of twins from Sweden, Denmark, and Finland. N Engl J Med 343 : 78–85.
5. HemminkiK, CzeneK (2002) Age specific and attributable risks of familial prostate carcinoma from the family-cancer database. Cancer 95 : 1346–1353.
6. ZeegersMP, JellemaA, OstrerH (2003) Empiric risk of prostate carcinoma for relatives of patients with prostate carcinoma: a meta-analysis. Cancer 97 : 1894–1903.
7. EelesRA, OlamaAA, BenllochS, SaundersEJ, LeongamornlertDA, et al. (2013) Identification of 23 new prostate cancer susceptibility loci using the iCOGS custom genotyping array. Nat Genet 45 : 385–382, 385-391, 391e381-382.
8. GohCL, SchumacherFR, EastonD, MuirK, HendersonB, et al. (2012) Genetic variants associated with predisposition to prostate cancer and potential clinical implications. J Intern Med 271 : 353–365.
9. EelesRA, Kote-JaraiZ, GilesGG, OlamaAA, GuyM, et al. (2008) Multiple newly identified loci associated with prostate cancer susceptibility. Nat Genet 40 : 316–321.
10. ShahN, SukumarS (2010) The Hox genes and their roles in oncogenesis. Nat Rev Cancer 10 : 361–371.
11. KimSD, ParkRY, KimYR, KimIJ, KangTW, et al. (2010) HOXB13 is co-localized with androgen receptor to suppress androgen-stimulated prostate-specific antigen expression. Anat Cell Biol 43 : 284–293.
12. NorrisJD, ChangCY, WittmannBM, KunderRS, CuiH, et al. (2009) The homeodomain protein HOXB13 regulates the cellular response to androgens. Mol Cell 36 : 405–416.
13. GillandersEM, XuJ, ChangBL, LangeEM, WiklundF, et al. (2004) Combined genome-wide scan for prostate cancer susceptibility genes. J Natl Cancer Inst 96 : 1240–1247.
14. LangeEM, GillandersEM, DavisCC, BrownWM, CampbellJK, et al. (2003) Genome-wide scan for prostate cancer susceptibility genes using families from the University of Michigan prostate cancer genetics project finds evidence for linkage on chromosome 17 near BRCA1. Prostate 57 : 326–334.
15. XuJ, DimitrovL, ChangBL, AdamsTS, TurnerAR, et al. (2005) A combined genomewide linkage scan of 1,233 families for prostate cancer-susceptibility genes conducted by the international consortium for prostate cancer genetics. Am J Hum Genet 77 : 219–229.
16. ChengL, BostwickDG, LiG, WangQ, HuN, et al. (1999) Allelic imbalance in the clonal evolution of prostate carcinoma. Cancer 85 : 2017–2022.
17. GaoX, ZacharekA, SalkowskiA, GrignonDJ, SakrW, et al. (1995) Loss of heterozygosity of the BRCA1 and other loci on chromosome 17q in human prostate cancer. Cancer Res 55 : 1002–1005.
18. UchidaT, WangC, SatoT, GaoJ, TakashimaR, et al. (1999) BRCA1 gene mutation and loss of heterozygosity on chromosome 17q21 in primary prostate cancer. Int J Cancer 84 : 19–23.
19. GoodeEL, Chenevix-TrenchG, SongH, RamusSJ, NotaridouM, et al. (2010) A genome-wide association study identifies susceptibility loci for ovarian cancer at 2q31 and 8q24. Nat Genet 42 : 874–879.
20. EwingCM, RayAM, LangeEM, ZuhlkeKA, RobbinsCM, et al. (2012) Germline mutations in HOXB13 and prostate-cancer risk. N Engl J Med 366 : 141–149.
21. KarlssonR, AlyM, ClementsM, ZhengL, AdolfssonJ, et al. (2012) A Population-based Assessment of Germline HOXB13 G84E Mutation and Prostate Cancer Risk. Eur Urol 65 : 169–76.
22. ShangZ, ZhuS, ZhangH, LiL, NiuY (2013) Germline homeobox B13 (HOXB13) G84E mutation and prostate cancer risk in European descendants: a meta-analysis of 24,213 cases and 73, 631 controls. Eur Urol 64 : 173–176.
23. WitteJS, MeffordJ, PlummerSJ, LiuJ, ChengI, et al. (2013) HOXB13 mutation and prostate cancer: studies of siblings and aggressive disease. Cancer Epidemiol Biomarkers Prev 22 : 675–680.
24. XuJ, LangeEM, LuL, ZhengSL, WangZ, et al. (2013) HOXB13 is a susceptibility gene for prostate cancer: results from the International Consortium for Prostate Cancer Genetics (ICPCG). Hum Genet 132 : 5–14.
25. ChenZ, GreenwoodC, IsaacsWB, FoulkesWD, SunJ, et al. (2013) The G84E mutation of HOXB13 is associated with increased risk for prostate cancer: results from the REDUCE trial. Carcinogenesis 34 : 1260–1264.
26. HaimanCA, ChenGK, BlotWJ, StromSS, BerndtSI, et al. (2011) Genome-wide association study of prostate cancer in men of African ancestry identifies a susceptibility locus at 17q21. Nat Genet 43 : 570–573.
27. HuntKA, MistryV, BockettNA, AhmadT, BanM, et al. (2013) Negligible impact of rare autoimmune-locus coding-region variants on missing heritability. Nature 498 : 232–235.
28. MarigortaUM, NavarroA (2013) High trans-ethnic replicability of GWAS results implies common causal variants. PLoS Genet 9: e1003566.
29. WhiffinN, DobbinsSE, HoskingFJ, PallesC, TenesaA, et al. (2013) Deciphering the genetic architecture of low-penetrance susceptibility to colorectal cancer. Hum Mol Genet 22 : 507–82.
30. ChangD, KeinanA (2012) Predicting signatures of “synthetic associations” and “natural associations” from empirical patterns of human genetic variation. PLoS Comput Biol 8: e1002600.
31. DicksonSP, WangK, KrantzI, HakonarsonH, GoldsteinDB (2010) Rare variants create synthetic genome-wide associations. PLoS Biol 8: e1000294.
32. SunX, NamkungJ, ZhuX, ElstonRC (2011) Capability of common SNPs to tag rare variants. BMC Proc 5 Suppl 9: S88.
33. Kote-JaraiZ, LeongamornlertD, TymrakiewiczM, FieldH, GuyM, et al. (2010) Mutation analysis of the MSMB gene in familial prostate cancer. Br J Cancer 102 : 414–418.
34. LouH, YeagerM, LiH, BosquetJG, HayesRB, et al. (2009) Fine mapping and functional analysis of a common variant in MSMB on chromosome 10q11.2 associated with prostate cancer susceptibility. Proc Natl Acad Sci U S A 106 : 7933–7938.
35. Kote-JaraiZ, Amin Al OlamaA, LeongamornlertD, TymrakiewiczM, SaundersE, et al. (2011) Identification of a novel prostate cancer susceptibility variant in the KLK3 gene transcript. Hum Genet 129 : 687–694.
36. Kote-JaraiZ, SaundersEJ, LeongamornlertDA, TymrakiewiczM, DadaevT, et al. (2013) Fine-mapping identifies multiple prostate cancer risk loci at 5p15, one of which associates with TERT expression. Hum Mol Genet 22 : 2520–2528.
37. AndersonCA, PetterssonFH, ClarkeGM, CardonLR, MorrisAP, et al. (2010) Data quality control in genetic case-control association studies. Nat Protoc 5 : 1564–1573.
38. HowieB, MarchiniJ, StephensM (2011) Genotype imputation with thousands of genomes. G3 (Bethesda) 1 : 457–470.
39. HowieBN, DonnellyP, MarchiniJ (2009) A flexible and accurate genotype imputation method for the next generation of genome-wide association studies. PLoS Genet 5: e1000529.
40. SampsonJN, JacobsK, WangZ, YeagerM, ChanockS, et al. (2012) A two-platform design for next generation genome-wide association studies. Genet Epidemiol 36 : 400–408.
41. AulchenkoYS, RipkeS, IsaacsA, van DuijnCM (2007) GenABEL: an R library for genome-wide association analysis. Bioinformatics 23 : 1294–1296.
42. PurcellS, NealeB, Todd-BrownK, ThomasL, FerreiraMA, et al. (2007) PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81 : 559–575.
Štítky
Genetika Reprodukční medicína
Článek Natural Polymorphisms in Influence Negative Selection and CD4∶CD8 Lineage Commitment in the RatČlánek MicroRNAs Located in the Hox Gene Clusters Are Implicated in Huntington's Disease PathogenesisČlánek Comparative RNAi Screens in and Reveal the Impact of Developmental System Drift on Gene FunctionČlánek Mutation of SLC35D3 Causes Metabolic Syndrome by Impairing Dopamine Signaling in Striatal D1 Neurons
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2014 Číslo 2
-
Všechny články tohoto čísla
- Fifteen Years Later: Hard and Soft Selection Sweeps Confirm a Large Population Number for HIV In Vivo
- The Same but Different: Worms Reveal the Pervasiveness of Developmental System Drift
- Serine Carboxypeptidase SCPEP1 and Cathepsin A Play Complementary Roles in Regulation of Vasoconstriction via Inactivation of Endothelin-1
- Coherent Functional Modules Improve Transcription Factor Target Identification, Cooperativity Prediction, and Disease Association
- A Long-Chain Flavodoxin Protects from Oxidative Stress and Host Bacterial Clearance
- Mammalian E-type Cyclins Control Chromosome Pairing, Telomere Stability and CDK2 Localization in Male Meiosis
- Influenza Virus Drug Resistance: A Time-Sampled Population Genetics Perspective
- Transcriptome-Wide Analyses of 5′-Ends in RNase J Mutants of a Gram-Positive Pathogen Reveal a Role in RNA Maturation, Regulation and Degradation
- Selective Disruption of Aurora C Kinase Reveals Distinct Functions from Aurora B Kinase during Meiosis in Mouse Oocytes
- X Chromosome Control of Meiotic Chromosome Synapsis in Mouse Inter-Subspecific Hybrids
- A Cohesin-Independent Role for NIPBL at Promoters Provides Insights in CdLS
- Extreme Population Differences in the Human Zinc Transporter ZIP4 (SLC39A4) Are Explained by Positive Selection in Sub-Saharan Africa
- Classic Selective Sweeps Revealed by Massive Sequencing in Cattle
- Genomic Networks of Hybrid Sterility
- Natural Polymorphisms in Influence Negative Selection and CD4∶CD8 Lineage Commitment in the Rat
- Oxidative Stress Is Not a Major Contributor to Somatic Mitochondrial DNA Mutations
- Molecular Identification of Collagen 17a1 as a Major Genetic Modifier of Laminin Gamma 2 Mutation-Induced Junctional Epidermolysis Bullosa in Mice
- Uncoupling of Molecular Maturation from Peripheral Target Innervation in Nociceptors Expressing a Chimeric TrkA/TrkC Receptor
- MicroRNAs Located in the Hox Gene Clusters Are Implicated in Huntington's Disease Pathogenesis
- Loss of Trabid, a New Negative Regulator of the Immune-Deficiency Pathway at the Level of TAK1, Reduces Life Span
- Targeted Ablation of Nesprin 1 and Nesprin 2 from Murine Myocardium Results in Cardiomyopathy, Altered Nuclear Morphology and Inhibition of the Biomechanical Gene Response
- Identification of Novel Genetic Loci Associated with Thyroid Peroxidase Antibodies and Clinical Thyroid Disease
- CEP-1, the p53 Homolog, Mediates Opposing Longevity Outcomes in Mitochondrial Electron Transport Chain Mutants
- Transcriptomics and Functional Genomics of ROS-Induced Cell Death Regulation by
- Quantitative Genome-Wide Genetic Interaction Screens Reveal Global Epistatic Relationships of Protein Complexes in
- Cascades of Genetic Instability Resulting from Compromised Break-Induced Replication
- Serine- and Threonine/Valine-Dependent Activation of PDK and Tor Orthologs Converge on Sch9 to Promote Aging
- Zfp322a Regulates Mouse ES Cell Pluripotency and Enhances Reprogramming Efficiency
- Insertional Mutagenesis and Deep Profiling Reveals Gene Hierarchies and a -Dependent Bottleneck in Lymphomagenesis
- DAAM Is Required for Thin Filament Formation and Sarcomerogenesis during Muscle Development in Drosophila
- Plasma Cholesterol–Induced Lesion Networks Activated before Regression of Early, Mature, and Advanced Atherosclerosis
- High-Resolution Profiling of Stationary-Phase Survival Reveals Yeast Longevity Factors and Their Genetic Interactions
- Comparative RNAi Screens in and Reveal the Impact of Developmental System Drift on Gene Function
- Accurate and Robust Genomic Prediction of Celiac Disease Using Statistical Learning
- Sex-Specific Embryonic Gene Expression in Species with Newly Evolved Sex Chromosomes
- Chromosome X-Wide Association Study Identifies Loci for Fasting Insulin and Height and Evidence for Incomplete Dosage Compensation
- Negative Feedback and Transcriptional Overshooting in a Regulatory Network for Horizontal Gene Transfer
- DNA Sequence Explains Seemingly Disordered Methylation Levels in Partially Methylated Domains of Mammalian Genomes
- Insights into the Genomic Landscape: Comparative Genomics Reveals Variations in Ploidy and Nutrient Utilisation Potential amongst Wine Isolates
- Molecular Evidence for the Inverse Comorbidity between Central Nervous System Disorders and Cancers Detected by Transcriptomic Meta-analyses
- The Centriolar Satellite Protein AZI1 Interacts with BBS4 and Regulates Ciliary Trafficking of the BBSome
- Fine-Mapping the Region Detects Common Variants Tagging a Rare Coding Allele: Evidence for Synthetic Association in Prostate Cancer
- Transmission Distortion Affecting Human Noncrossover but Not Crossover Recombination: A Hidden Source of Meiotic Drive
- A Variant in the Neuropeptide Receptor is a Major Determinant of Growth and Physiology
- Mutation of SLC35D3 Causes Metabolic Syndrome by Impairing Dopamine Signaling in Striatal D1 Neurons
- NSUN4 Is a Dual Function Mitochondrial Protein Required for Both Methylation of 12S rRNA and Coordination of Mitoribosomal Assembly
- MicroRNA-133 Inhibits Behavioral Aggregation by Controlling Dopamine Synthesis in Locusts
- Convergence of Light and ABA Signaling on the Promoter
- Arf4 Is Required for Mammalian Development but Dispensable for Ciliary Assembly
- Distinct Requirements for Cranial Ectoderm and Mesenchyme-Derived Wnts in Specification and Differentiation of Osteoblast and Dermal Progenitors
- Chk2 and P53 Regulate the Transmission of Healed Chromosomes in the Male Germline
- Ddc2 Mediates Mec1 Activation through a Ddc1- or Dpb11-Independent Mechanism
- Mapping the Fitness Landscape of Gene Expression Uncovers the Cause of Antagonism and Sign Epistasis between Adaptive Mutations
- Euchromatic Transposon Insertions Trigger Production of Novel Pi- and Endo-siRNAs at the Target Sites in the Germline
- miR-100 Induces Epithelial-Mesenchymal Transition but Suppresses Tumorigenesis, Migration and Invasion
- Canine Hereditary Ataxia in Old English Sheepdogs and Gordon Setters Is Associated with a Defect in the Autophagy Gene Encoding
- Within-Host Spatiotemporal Dynamics of Plant Virus Infection at the Cellular Level
- Analysis of Meiosis in SUN1 Deficient Mice Reveals a Distinct Role of SUN2 in Mammalian Meiotic LINC Complex Formation and Function
- Genome-Wide Association Study of Metabolic Traits Reveals Novel Gene-Metabolite-Disease Links
- Mechanistically Distinct Mouse Models for -Associated Retinopathy
- DAF-16/FoxO Directly Regulates an Atypical AMP-Activated Protein Kinase Gamma Isoform to Mediate the Effects of Insulin/IGF-1 Signaling on Aging in
- Chromosome I Controls Chromosome II Replication in
- Integrated Genomic Characterization Reveals Novel, Therapeutically Relevant Drug Targets in FGFR and EGFR Pathways in Sporadic Intrahepatic Cholangiocarcinoma
- The Iodotyrosine Deiodinase Ortholog SUP-18 Functions through a Conserved Channel SC-Box to Regulate the Muscle Two-Pore Domain Potassium Channel SUP-9
- The Genome of Highlights a Fish Pathogen Adapted to Fluctuating Environments
- Distinct DNA Binding Sites Contribute to the TCF Transcriptional Switch in and
- The Streamlined Genome of spp. Relative to Human Pathogenic Kinetoplastids Reveals a Parasite Tailored for Plants
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Genome-Wide Association Study of Metabolic Traits Reveals Novel Gene-Metabolite-Disease Links
- A Cohesin-Independent Role for NIPBL at Promoters Provides Insights in CdLS
- Classic Selective Sweeps Revealed by Massive Sequencing in Cattle
- Arf4 Is Required for Mammalian Development but Dispensable for Ciliary Assembly
Přihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



