-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Top novinky
ReklamaAdaptation to High Ethanol Reveals Complex Evolutionary Pathways
Organisms can evolve resistance to specific stress factors, which allows them to thrive in environments where non-adapted organisms fail to grow. However, the molecular mechanisms that underlie adaptation to complex stress factors that interfere with basic cellular processes are poorly understood. In this study, we reveal how yeast populations adapt to high ethanol concentrations, an ecologically and industrially relevant stress that is still poorly understood. We exposed six independent populations of genetically identical yeast cells to gradually increasing ethanol levels, and we monitored the changes in their DNA sequence over a two-year period. Together with novel computational analyses, we could identify the mutational dynamics and molecular mechanisms underlying increased ethanol resistance. Our results show how adaptation to high ethanol is complex and can be reached through different mutational pathways. Together, our study offers a detailed picture of how populations adapt to a complex continuous stress and identifies several mutations that increase ethanol resistance, which opens new routes to obtain superior biofuel yeast strains.
Published in the journal: . PLoS Genet 11(11): e32767. doi:10.1371/journal.pgen.1005635
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1005635Summary
Organisms can evolve resistance to specific stress factors, which allows them to thrive in environments where non-adapted organisms fail to grow. However, the molecular mechanisms that underlie adaptation to complex stress factors that interfere with basic cellular processes are poorly understood. In this study, we reveal how yeast populations adapt to high ethanol concentrations, an ecologically and industrially relevant stress that is still poorly understood. We exposed six independent populations of genetically identical yeast cells to gradually increasing ethanol levels, and we monitored the changes in their DNA sequence over a two-year period. Together with novel computational analyses, we could identify the mutational dynamics and molecular mechanisms underlying increased ethanol resistance. Our results show how adaptation to high ethanol is complex and can be reached through different mutational pathways. Together, our study offers a detailed picture of how populations adapt to a complex continuous stress and identifies several mutations that increase ethanol resistance, which opens new routes to obtain superior biofuel yeast strains.
Introduction
The ability to survive and proliferate in high levels of ethanol is an ecologically important and industrially relevant trait of yeast cells. The ethanol produced by yeast cells slows down growth of competing microbes, but at higher concentrations, it causes stress for the yeast cells themselves. Different yeast strains show significant differences in their ability to grow in the presence of ethanol, with the more ethanol-tolerant ones likely having a fitness advantage over non-tolerant strains [1–3]. Moreover, ethanol tolerance is a key trait of industrial yeasts that often encounter very high ethanol concentrations, for example during beer and wine making and industrial bio-ethanol production.
Because of its industrial importance, there is great interest in fully understanding the genetic underpinnings of ethanol tolerance in microbes. Different experimental approaches have been used, including screening of (deletion) mutants for increased ethanol tolerance, transcriptome analysis of ethanol stressed cells and QTL analyses aimed at identifying mutations that cause differences in ethanol tolerance between different yeast strains [4–10]. Together, these studies have linked multiple different genetic loci to ethanol tolerance and identified hundreds of genes, involved in a multitude of cellular processes [11–14]. While it becomes increasingly clear that ethanol is a complex stress that acts on several different processes including increasing fluidity and permeability of cellular membranes, changing activity and solubility of membrane-bound and cytosolic proteins and interfering with the proton motive force (for review, see [15–17], the exact molecular mechanisms and genetic architecture underlying ethanol tolerance are still largely unknown.
Experimental evolution to study adaptation to increased ethanol levels could provide more insight into the molecular mechanisms underlying ethanol tolerance since such experiments could reveal different mutational paths that make a sensitive strain more tolerant. Only a handful of studies have looked at adaptation to ethanol in originally non-ethanol tolerant microbes exposed to gradually increasing levels of ethanol [6,18–22]. These have mostly focused on the physiological adaptations found in the evolved cells and have not performed an extensive analysis of the mechanisms and genetic changes underlying this adaptation. Hence, a comprehensive analysis of the type and number of mutations a non-ethanol tolerant strain can (or needs to) acquire to become more ethanol tolerant is still lacking.
Experimental evolution has proven to be a valuable tool to investigate the different mechanisms and pathways important for cells to adapt to specific selective conditions. Seminal papers have increased our understanding of the molecular basis of adaptation to specific stresses, such as heat stress, nutrient limitation and antibiotic treatment [23–28]. Recent advances in DNA sequencing technologies allow affordable and fast sequencing of complete genomes of clones and populations. While sequencing clones yields information on individual lineages within the experiment, population data provides information on the heterogeneity of adaptation. Additionally, sequencing samples isolated at different time points during the evolution experiment makes it possible to capture evolution in action. This has provided valuable information on the rate and types of mutations underlying adaptation, the genetic basis of ‘novel’ phenotypes and the existence of parallel pathways to establish comparable phenotypic outcomes [29–32].
For example, a common strategy observed in populations evolving under nutrient limitation is the amplification of genetic regions encoding transporters responsible for the uptake of the limiting nutrient [24,33]. Other studies using multiple replicate populations have discovered a high degree of parallelism in the adaptive solutions found by different populations. However, in other cases, the evolutionary pathways can be more complex. For example, clonal interference, the competition between lineages carrying different beneficial mutations, is a commonly observed phenomenon in evolution of asexually propagating populations that can increase complexity of mutational dynamics as well as impede the spread of beneficial mutations in a population. Clonal interference is typically expected to be prevalent in large populations that are adapting to a complex stress, where multiple adaptive mutations can occur [32,34,35].
To unravel the molecular mechanisms of adaptation to a specific condition, most studies have used isogenic replicate populations, with all cells having the same initial genome size. Genome size can significantly change during evolution; with both small-scale changes (chromosomal deletions and amplifications) and large-scale changes (increase or decrease in ploidy). Moreover, ploidy shifts have been reported in the evolutionary history of many organisms, including Saccharomyces cerevisiae and as a response to selective pressure [36–38]. Conversely, genome size has also been reported to affect evolution rate: polyploidy has been suggested to increase adaptability [39,40]. Multiplying the amount of DNA increases the genetic material available for evolution to tinker with and can alter gene expression [41–43]. These polyploid genomes can be unstable, resulting in loss of chromosomes and thus aneuploid cells [44–46]. Although studies have looked at adaptation of lineages of different ploidy, none have followed the mutational dynamics in these evolving populations over time in detail [36,47–49].
In this study, we use experimental evolution to dissect the adaptive mechanisms underlying ethanol tolerance in yeast. Six isogenic Saccharomyces cerevisiae populations of different ploidy (haploid, diploid and tetraploid) were asexually propagated in a turbidostat over a two-year period, with ethanol levels gradually increasing during the experiment. This step-wise increase in exogenous ethanol levels resulted in a constant selective pressure for our cells. Whole-genome sequencing of evolved populations and isolated, ethanol-tolerant clones at different times during the experiment allowed us to paint a detailed picture of the mutational dynamics in the different populations. Several common themes in the type of adaptations and evolutionary mechanisms emerge, with all lines showing extensive clonal interference as well as copy number variations. Additionally, the haploid and tetraploid lines showed rapid convergence towards a diploid state. Despite these common themes, we find multiple lineage-specific adaptations, with little overlap in the mutated genes between the different populations. By applying a novel computational pipeline to identify affected pathways, we were able to reveal overlap between the functional modules affected in the different adapted populations, with both novel and previously established pathways and genes contributing to ethanol tolerance. Importantly, introduction of specific mutated alleles present in adapted populations into the ancestral strain significantly increased its ethanol tolerance, demonstrating the adaptive nature of these mutations and the potential of using experimental evolution to unravel and improve a complex phenotype.
Results
Evolved cells are more ethanol tolerant
To study the mutational dynamics underlying increased ethanol tolerance, six prototrophic, isogenic S. cerevisiae strains of different ploidy (two haploid, two diploid and two tetraploid lines—with each line of the same initial ploidy started from the same preculture (VK111, VK145 and VK202 respectively, see also S4 Table) were subjected to increasing levels of ethanol. Specifically, ethanol levels were gradually increased from 6% (v/v) to 12% over a two-year period in a continuous turbidostat with glucose (4% (w/v)) as a carbon source. Samples were taken at regular time intervals and subjected to whole-genome sequencing analysis. Chloramphenical was added to the medium of our long-term evolution experiment to prevent bacterial growth. Chloramphenicol does not affect yeast growth (S1 Fig). In addition to sequencing each of the 6 evolving populations, we also sequenced the genomes of three clones isolated from each of the population samples, resulting in a total of 34 population samples and 102 clonal samples that were sequenced. Fig 1 depicts the experimental set-up as well as the time points (number of generations) for which whole-genome sequencing was performed.
Fig. 1. Experimental setup. 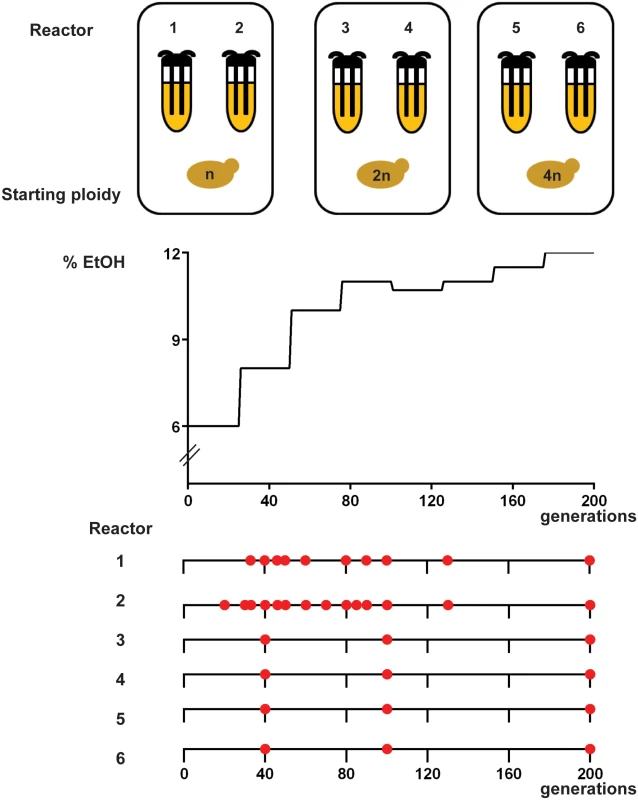
(A) Experimental evolution of prototrophic, isogenic populations of different ploidy (haploid (VK111), diploid (VK145) and tetraploid (VK202)) for increased ethanol tolerance was performed in a turbidostat. Every 25 generations, the ethanol concentration in the media was increased in a stepwise manner (starting at 6% (v/v) and reaching 12% at 200 generations). Increasing the ethanol concentration from 10% to 11% dramatically reduced growth rate of evolving cells. Therefore, instead of increasing ethanol levels, we first reduced the ethanol level to 10.7% after 100 generations. (B) Red circles represent sampling points (indicated as number of generations) for which whole-genome sequencing was performed. For each circle, heterogeneous populations as well as three evolved, ethanol tolerant clones were sequenced. Sequencing of the population sample of reactor 4 at 200 generations failed, so this data is omitted from the manuscript. For generation 80 of reactor 1, only population data is available. We determined the relative fitness of the evolved populations, isolated at 40 and 200 generations, and generally observed increases in fitness in high (9% v/v) EtOH (Fig 2 & Materials and Methods). In general, clones taken from the same population at the same time point had similar fitness; with some notable exceptions suggesting considerable heterogeneity within populations (S2 Fig). These fitness measurements were performed in 9% ethanol because the ancestral reference strains did not grow at higher ethanol levels. It is important to note that the actual increase in fitness of our evolved strains in higher ethanol levels (above 9% (v/v)) is much larger than what can be appreciated from Fig 2. Spotting of our evolved lineages on agar plates with increasing levels of ethanol indicates that these adapted strains can grow on concentrations up to 11% (v/v) EtOH, with the haploid control ancestral strain showing almost no growth under these conditions (i.e. an infinite increase in fitness compared to the ancestral haploid strain) (S3 Fig). Ancestral diploid and tetraploid strains show growth on 11% EtOH, although they are still outperformed by their evolved clones under these conditions.
Fig. 2. Evolved populations are more ethanol tolerant. 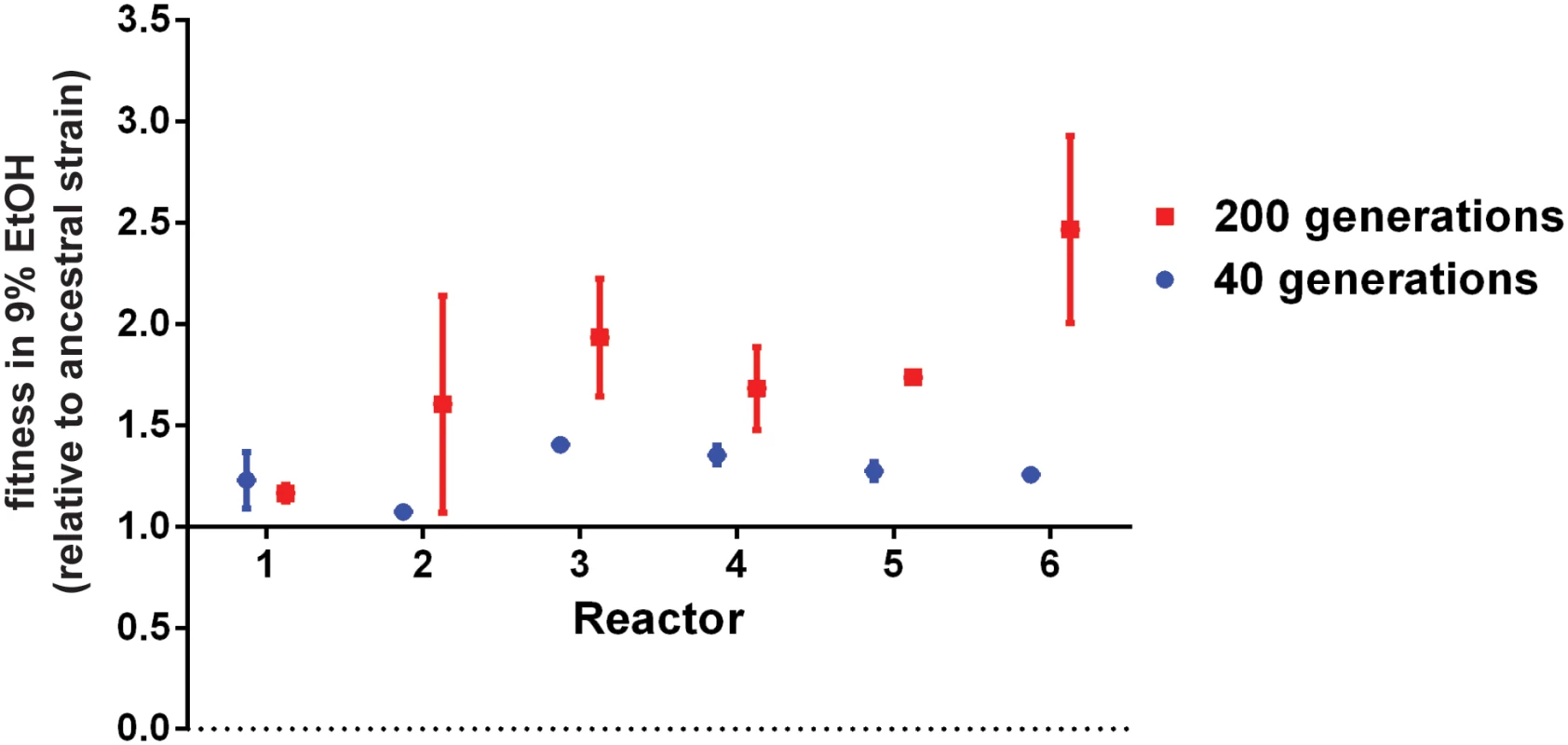
Evolved populations from different reactors show increased fitness in EtOH. Fitness was determined for population samples of each reactor after 40 generations (blue) and 200 generations (red). Fitness is expressed relative to the ancestral strain of each reactor (haploid for reactor 1–2; diploid for reactor 3–4 and tetraploid for reactor 5–6). Data represent the average of three independent measurements, error bars represent standard deviation. Populations with mutator phenotypes showed higher standard deviations. This might be due to the large supply of mutations generated by these mutators, which could affect mutation-selection balance. Diploidization of haploid cells is a fast way of increasing ethanol tolerance
Propidium iodide staining and flow cytometry analysis of evolved populations showed that diploid cells appeared relatively quickly in the originally haploid populations (Fig 3; PI staining profiles of ancestral strains can be found in S4 Fig; ploidy of sequenced clones can be found in S1 File). This was observed independently in both reactors started from the same isogenic haploid population. It should be noted that around generation 40 in reactor 1, some diploids are already present in the originally haploid population. By generation 60, haploid cells took over the population again, possibly by acquiring a mutation that made them more fit than these new diploids. In both reactors, diploid cells have taken over the entire population by 130 generations. These diploid variants are still of mating type α, the same as the initial haploid ancestral strain, indicating that they did not arise through mating type switching and subsequent mating. Our results indicate that the diploids in our turbidostat are likely the result of failed cytokinesis. The relatively rapid evolutionary sweep of the new diploid variants points to a significant selective advantage. Competition experiments confirmed that the fitness of a diploid cell is indeed significantly higher than that of an isogenic haploid strain in 9% EtOH (p = 0.0063; unpaired t-test), whereas there is no significant fitness difference in the absence of ethanol (p = 0.469; unpaired t-test) (S5 Fig). Interestingly, our tetraploid starting populations also converged to a diploid state relatively fast during the evolution experiment. The parallel diploidization observed in two reactors with haploid ancestral cells and two reactors with tetraploid ancestral cells might suggest that becoming diploid is one of the main and/or one of the more easily accessible routes leading to increased ethanol tolerance.
Fig. 3. Haploid lineages diploidized during adaptation to EtOH. 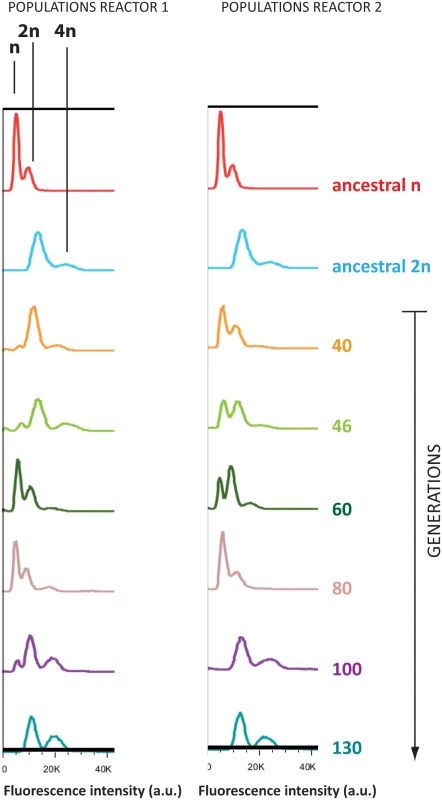
Flow cytometry analysis of DNA content (stained by propidium iodide) of evolved populations, compared to ancestral haploid (red) and diploid (blue). For the clonal ancestral samples, two peaks are observed, corresponding to the G1 and G2 phase of the cell cycle. Evolved populations sometimes display three peaks, indicative of both haploid and diploid subpopulations. Whole-genome sequencing of evolved populations and clones reveals a complex pattern of adaptation
To identify the mutational pathways during adaptation to ethanol, we performed whole-genome sequencing of populations of evolving cells throughout the two-year experiment. Each population sample was sequenced to 500 fold coverage on average, and this for multiple time points (34 samples for all reactors combined, see Fig 1). In addition to this whole-population sequencing strategy, we also sequenced for each time point the genomes of three adapted clones to 80 fold coverage on average, resulting in 102 clonal samples sequenced in total. Details on the sequencing and analysis pipelines can be found in Materials and Methods.
Whole genome sequencing of adapted clones reveals mutators and extensive aneuploidies
After 2 years, yielding around 200 generations, evolved clones (excluding clones from reactor 2 and 6, which acquired a mutator phenotype, see below) contained on average 23 SNPs compared to the ancestral strain (S1 File). This number is higher than what would be expected based on measured rates of spontaneous mutations [50], and could reflect an increased mutation rate under the stressful conditions imposed by the high ethanol levels in our set-up. Across all reactors and clones sequenced, we identified a total of 8932 different sites mutated. The largest fraction are SNPs (6424 out of 8932; 72%); Indels are found mostly in non-coding regions (1830 out of 2508; 73%), whereas SNPs are mostly found inside genes (4971 out of 6424; 77%). Most of these coding SNPs are non-synonymous (3672 out of 4971; 74%). A full list of all mutations (SNPs and Indels) in all sequenced clones for the different reactors and time points can be found in the S1 File.
In two reactors (reactor 2 and reactor 6), we noticed a marked increase in the number of mutations found in individual clones (see Fig 4, S1 and S2 Files). Specifically, clones from reactor 2 contain a significantly higher number of indels (about 929 on average per genome for clones isolated after 200 generations, compared to 21 on average per genome for clones isolated after 200 generations in other reactors), whereas clones from reactor 6 display an increase in the number of SNPs (about 1765 per genome for clones isolated after 200 generations, compared to an average of 31 per clone for other reactors).This high number of mutations points to a so-called “mutator” phenotype typically present in cells with lower DNA replication fidelity and/or DNA repair. Interestingly, such mutators are frequently observed during evolution experiments, probably because mutators are more likely to acquire (combinations of) adaptive mutations faster [23,51–54].
Fig. 4. Mutations present in evolved clones isolated from all reactors at 200 generations. 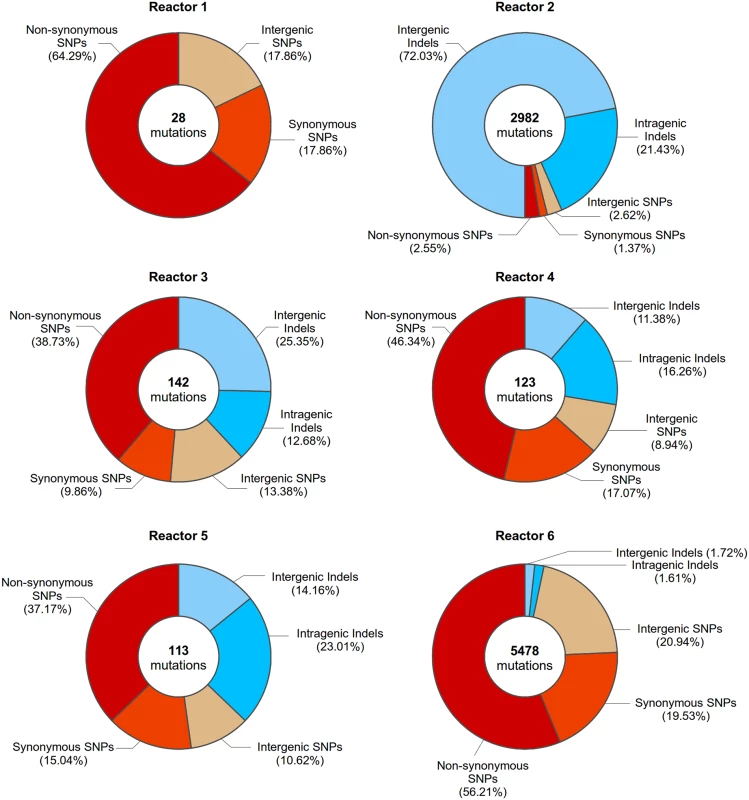
Circle diagrams depict the number and types of mutations identified in evolved clones at 200 generations of reactor 1, reactor 2, reactor 3, reactor 4, reactor 5 and reactor 6. Data for individual clones isolated at different time points can be found in S1 and S2 Files. Interestingly, the increased mutation rate in reactor 2 is first observed around generation 70–80, coinciding with the appearance of a 21 bp insertion in the MSH2 gene – a key player in DNA mismatch repair. Mutations in MSH2 have been previously reported to confer a mutator phenotype [55,56], and mutations in MutS, the ortholog of MSH2 in E. coli, were identified in a long term evolution experiment and also result in a mutator phenotype [23]. Indeed, deletion of this gene, as well as re-creating this insertion in an otherwise wild type background drastically increases mutation rate (S6 Fig). The MSH2 mutation eventually reaches a frequency of 100% at the end of the evolution experiment (200 generations). We currently do not know the exact cause of the elevated mutation rate observed in reactor 6.
Apart from the convergence towards diploidy (see above), we also detected extensive copy number variation (CNVs) in our evolved clones, comprising both duplicated and deleted chromosomal regions (see Fig 5 and S3 File). Other studies have observed similar copy number variation during adaptation to stress, including heat stress and specific nutrient limitations [24,28,33]. Interestingly, acquisition of an extra copy of chromosome III appeared to be a common feature for most of our evolved clones (S3 File). Some (parts of) other chromosomes, including chromosome XII and chromosome IV, are also duplicated in several of our evolved clones. These frequent occurrences indicate that these specific aneuploidies could be adaptive under ethanol conditions, although the exact mechanistic basis remains to be elucidated and further experiments are needed to validate the (potential) adaptive role of these observed aneuploidies.
Fig. 5. Copy number variation in evolved clones of reactor 2 and 3 isolated at 200 generations. 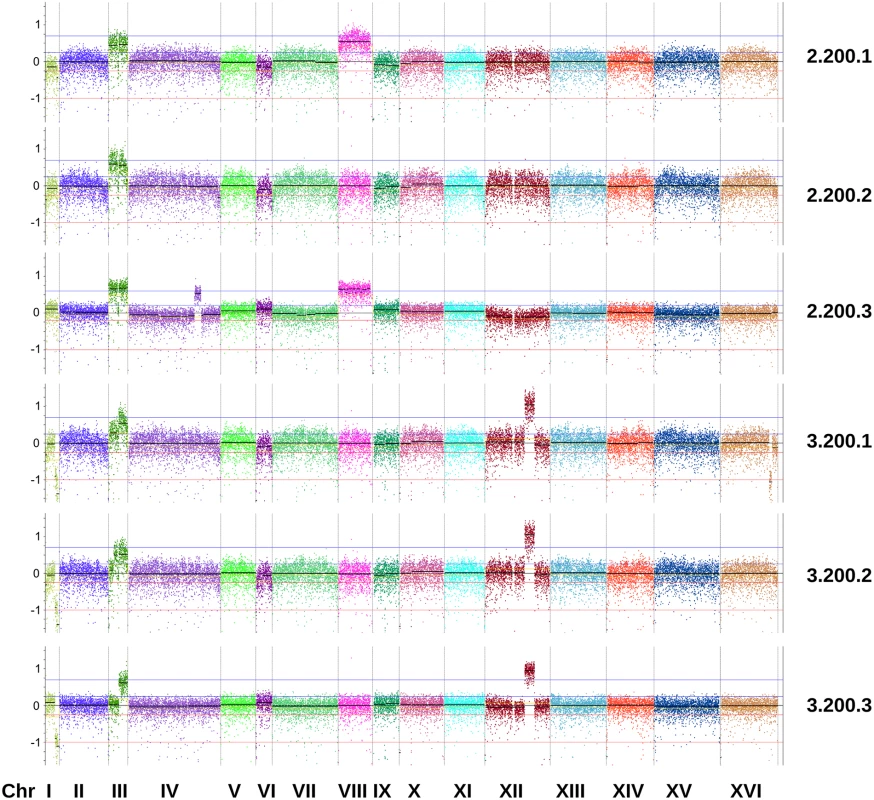
Genome view of yeast chromosomes and CNV patterns from a sliding window analysis. The y-axis represents log2 ratios of the coverage observed across 500bp genomic windows to the coverage expected in a diploid genome without CNVs. Area of the plot located between the red lines (from -0.23 to -1) marks putative CNV loss events, whereas region between the blue lines (from 0.23 to 0.58) marks putative CNV gain events. Data for other reactors is shown in S3 File. It should be noted that the amplified region of chromosome XII observed in some of our clones does not correspond to the ribosomal DNA genes. Whole-genome sequencing of evolved populations reveals extensive clonal interference
While the sequencing of clones yielded valuable information on individual lineages within the evolving populations; sequencing of evolving populations yielded more information on the complex mutational paths and dynamics between sub-populations within each evolving population. A list of SNPs and Indels identified in the evolved populations, together with their allele frequency in the population at each sampling point, can be found in S4 File. We observe distinct pattern of mutations appearing and disappearing over time in each of the six reactors. Some of these mutations remain in the population, eventually reaching high levels or even complete fixation (i.e. presence in 100% of all cells in the population). Other mutations only persist for a short time, until lineages carrying these mutations are outcompeted by others, so-called clonal interference. In total, we identified 1637 mutations across all populations and time points. 117 of these mutations are no longer present in the final time points sequenced, and 101 mutations drop more than 10% in frequency after reaching their maximum frequency, indicative of clonal interference. Interestingly, we also identified some overlap between the mutations found in different independently evolving populations (i.e. in different reactors). Specifically, we find 20 genes that are mutated twice in different generations and populations, 3 genes mutated 3 times, 2 genes mutated 4 times, 2 genes mutated 5 times and 1 gene 6 times (see also S1 Table). This significantly differs from what would be expected by chance (see Materials and Methods and S1 Table for p-values, exact binomial test). Repeatedly hit genes are, amongst others, involved in stress response, cell cycle and heme biosynthesis.
The higher number of sequenced samples from reactors 1 and 2 allowed us to further analyze these population sequences and group mutations based on correlations in the changes in their respective frequencies (based on the pipeline developed by [32]; see also Materials and Methods). This yields a more detailed picture of the different co-evolving sub-populations present in these reactors, which is depicted in the Muller diagrams of Fig 6 and S7 Fig, see also S2 Table for haplotype frequencies.
Fig. 6. Dynamics and linkage of mutations in evolved populations of reactor 2. 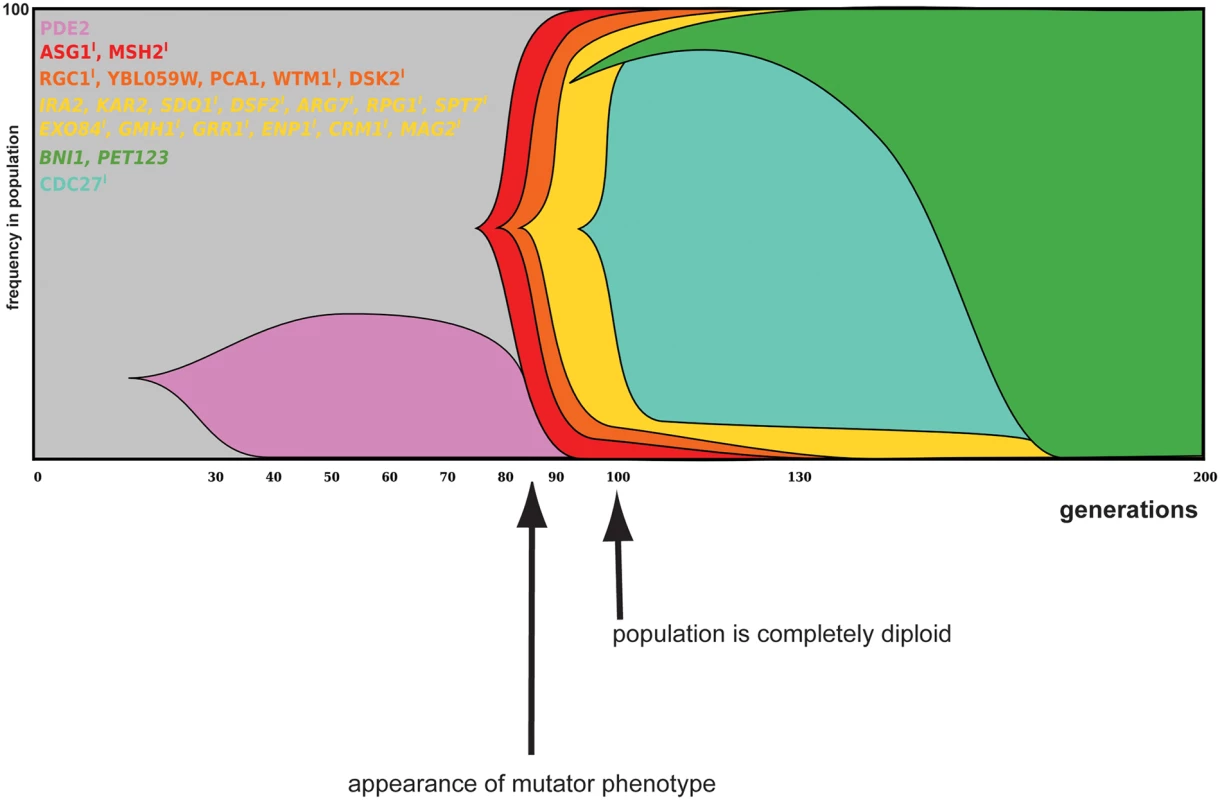
Mutations (reaching a frequency of least 20% in the evolved population samples) and corresponding frequencies were identified from population sequencing data. Muller diagram represents the hierarchical clustering of these mutations, with each color block representing a specific group of linked mutations. Indels are designated with I, whereas heterozygous mutations are in italics. Mutations present as heterozygous mutations in all clones of a specific time point and present at a frequency of 50% in the population, are depicted as a frequency of 100% in the population, since it is expected that all cells in the population contain this mutation. After 80 generations, a mutator phenotype appeared in this reactor (indicated by arrow under graph), which coincides with the rise in frequency of an indel in the mismatch repair gene MSH2. Frequencies of haplotypes can be found in S2 Table. Dynamics and linkage for reactor 1 is shown in S7 Fig. In both reactors, selective sweeps mostly consists of groups of mutations that move through the population together. While these reactors were inoculated with the same strain, the type and dynamics of mutations observed during adaptation appear very different. However, both evolving populations of reactor 1 and reactor 2 are characterized by strong clonal interference. In reactor 1, 4 different subpopulations are present around generation 90, each carrying different mutations. By generation 130, these lineages have been outcompeted by another lineage that almost completely dominates the population by 200 generations (S7 Fig). In reactor 2, a lineage carrying a mutation in PDE2 (encoding a high-affinity cAMP phosphodiesterase) is driven to extinction by a subpopulation carrying indels in ASG1 and MSH2. ASG1 is a transcriptional regulator involved in the stress response and has been found mutated in evolved populations from different reactors (including reactor 1, see also Fig 6, S7 Fig and S4 File). Another example of clonal interference is observed in the later generations of this reactor: a subpopulation carrying a mutation in CDC27 (encoding a subunit of the anaphase promoting complex/cyclosome) is driven to extinction by a subpopulation carrying mutations in BNI1 (important for nucleation of actin filaments) and PET123 (encoding a mitochondrial ribosomal protein).
Diverse pathways involved in adaptation to ethanol
The results discussed above revealed extensive variability in the type and number of mutations present in each evolving population. While this could suggest the presence of several, different mutational pathways (and thus lack of parallel evolution), mutations in different genes might affect identical or similar pathways, implying that the physiological adaptation to high ethanol might in fact be more similar than what is immediately apparent from the individual mutations. It should also be noted that some of the mutations identified in our evolved lineages might represent adaptations to the device used, or other aspects of the selection regime, rather than ethanol per se.
To gain insight into the affected biological pathways and investigate the possible similarities in adaptation to increased ethanol, different computational approaches were used (see also S1 Text). First, a term-enrichment analysis was performed (for enriched clusters, see Table 1 and S5 File). For this analyses, we excluded mutations obtained from the populations with a mutator phenotype (reactor 2 and 6) because of their low signal to noise ratio. These enrichment methods have been used as one of the standard functional analysis tools and gave us a first insight into potential adaptive pathways present in our evolved lineages.
Tab. 1. Top functional clusters identified amongst genes hit by mutations in the evolution experiment. 
An enrichment score cutoff value of 1.3, equivalent to a p-value of 0.05 for term enrichment was used [57]. In a second step, we used a sub-network-based selection method [58,59] (see Materials and Methods) first developed for E. coli expression data. Here, we adapted and extended this method to select the subnetwork from the global yeast interaction network that best connected the mutated genes in the most parsimonious way. This method also identifies the intermediary genes involved in signaling mechanisms, which are not necessarily mutated in our evolved lineages but mediate the cellular response. We excluded mutations obtained from the populations with a mutator phenotype (reactor 2 and 6) because of their low signal to noise ratio. This analysis identifies genes frequently mutated in the different populations (DSK2, ASG1), as well as genes that are closely connected on the interaction graph (HEM3, HEM12,…). This latter set reflects parallelism at the pathway level in the different reactors. 25% of genes from inside our identified networks have been previously linked to ethanol tolerance (broadly defined as fermentation/growth capacity under conditions with EtOH, as reported in literature), whereas such as a connection was found for only 9% of genes outside of the enriched networks. From Fig 7, it is clear that sometimes multiple mutations occurring in the same pathway originated in the same reactor rather than across different reactors. This could indicate not only a level of parallelism between the different reactors but also between the sub-populations in the same reactor. To confirm this, we also applied the sub-network selection method on the mutations obtained for each of the populations separately. This confirms that within single populations the same pathways seem to be affected as those that are consistently affected between the populations (including cell cycle, DNA repair and protoporphyrinogen metabolism, see also below). All resulting sub-networks as well as the enriched genes (including p-values) can be found in S6 File, Fig 7 and S8–S13 Figs.
Fig. 7. Adaptive pathways are involved in cell cycle, DNA repair and protoporphyrinogen metabolism. 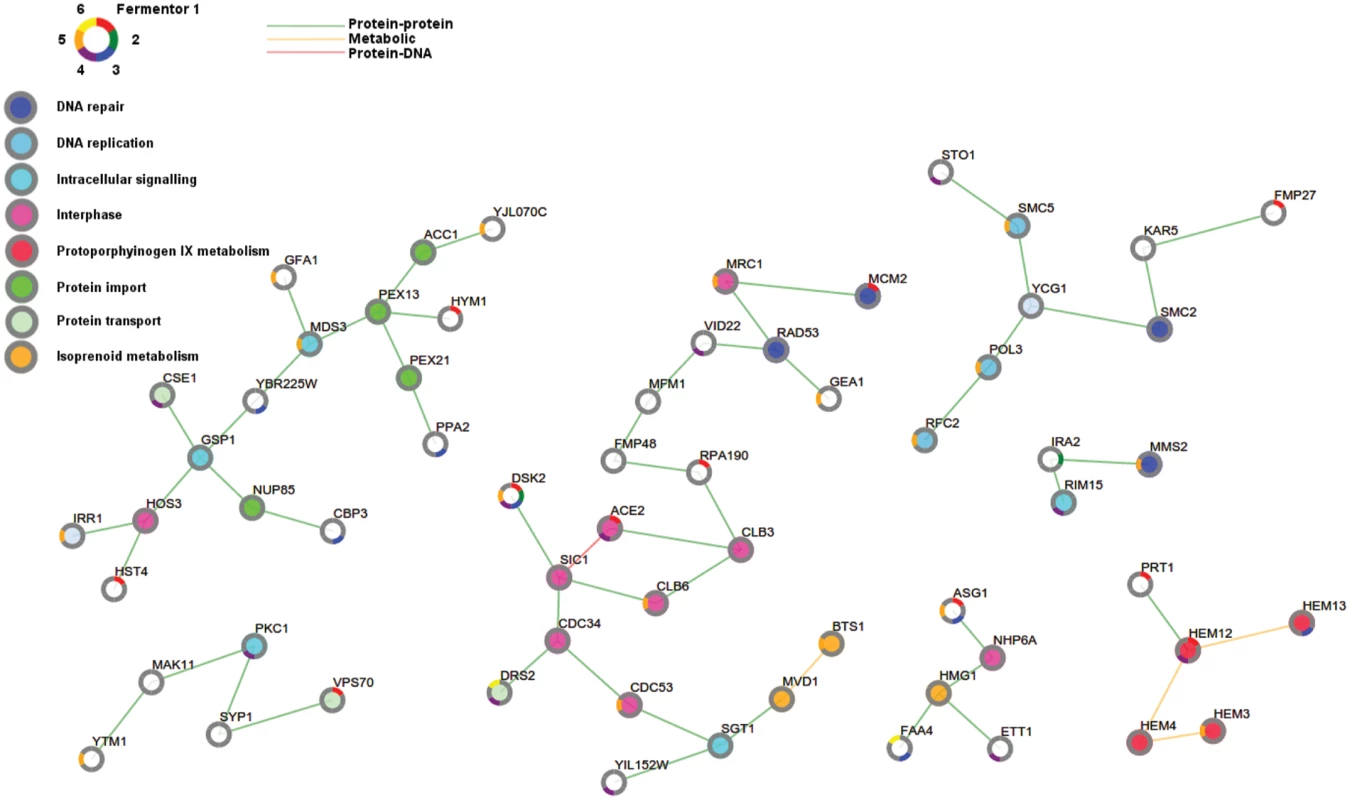
Shown is the sub-network that prioritizes putative adaptive mutations by applying PheNetic on all selected mutations, excluding those originating from the populations with a mutator phenotype i.e. reactor 2 and 6. The nodes in the network correspond to genes and/or their associated gene products. Node borders are colored according to the reactors containing the populations in which these genes were mutated. Nodes are colored according to gene function, for each gene the most enriched term is visualized (grey indicates no enrichment). Cell cycle related processes have been subdivided into DNA replication and interphase. The edge colors indicate different interaction types. Orange lines represent metabolic interactions, green lines represent protein-protein interactions, red lines represent protein-DNA interactions. Sub-networks extracted by separately analyzing the mutated genes observed in each of the different populations (reactors) are shown in S8–S13 Figs. To increase mechanistic insight, we performed several additional analyses, such as interactome analyses of genes containing non-synonymous mutations in our different population samples as well as analyses of the effect of these mutations on protein functional domains (see also S1 Text, S3 Table and S7 File for more details). Together, these analyses helped us identify the different pathways that were affected in our evolved lineages. Pathways identified as affected across different reactors by these different analyses are discussed in more detail in the next paragraphs.
Cell cycle and DNA replication
Our analyses suggest that the cell cycle and DNA replication are affected in our evolved lineages (Fig 7). Ethanol has been previously reported to delay cell cycle progression, probably through depolarization of the actin cytoskeleton [60]. This could be relevant for adaptation in the sense that slower growth could protect cells from stress, perhaps by inducing genes involved in the general stress response [61–63].
Respiration
PheNetic analysis shows that protoporphyrinogen metabolism is affected in our evolved populations (Fig 7). Since this is important for heme biosynthesis, this could indicate that respiration is affected downstream of mutations. Non-synonymous SNPs in functional domains or modified sites are expected to have more effect on the cell than mutations outside of these regions. We find such mutations in proteins involved in regulation and in enzymes of heme biosynthesis and proteins that need heme groups for proper functioning, that are themselves involved in respiration (S3 Table and S7 File). These analyses point to a role for changes in respiration in adaptation to ethanol.
Together, our results show evidence for parallel evolution, with the same processes (cell cycle and DNA replication) affected by different mutations, and also highlight the plethora of processes involved in increased ethanol tolerance in our adapted lineages.
Several high-frequency mutations increase ethanol tolerance of non-evolved strain
The high number of mutations (both SNPs and Indels) precluded an exhaustive analysis of all mutations present in our tolerant clones and populations and their effect on ethanol tolerance. One commonly used approach to investigate putative beneficial mutations is backcrossing of evolved clones to their ancestor, which does not contain any mutation. However, since each of the evolved populations proved unable to form any spores, this strategy was not accessible to us. Hence, we focused on SNPs reaching high frequencies in our 200 generation population samples. In total, nine SNPs were selected for further study (Table 2). Several of the mutated genes belong to or are linked to the processes affected across and/or within specific reactors (heme metabolism, protein transport and cell cycle, see also Table 2).
Tab. 2. SNPs in evolved lineages selected for introduction in non-ethanol tolerant strain. 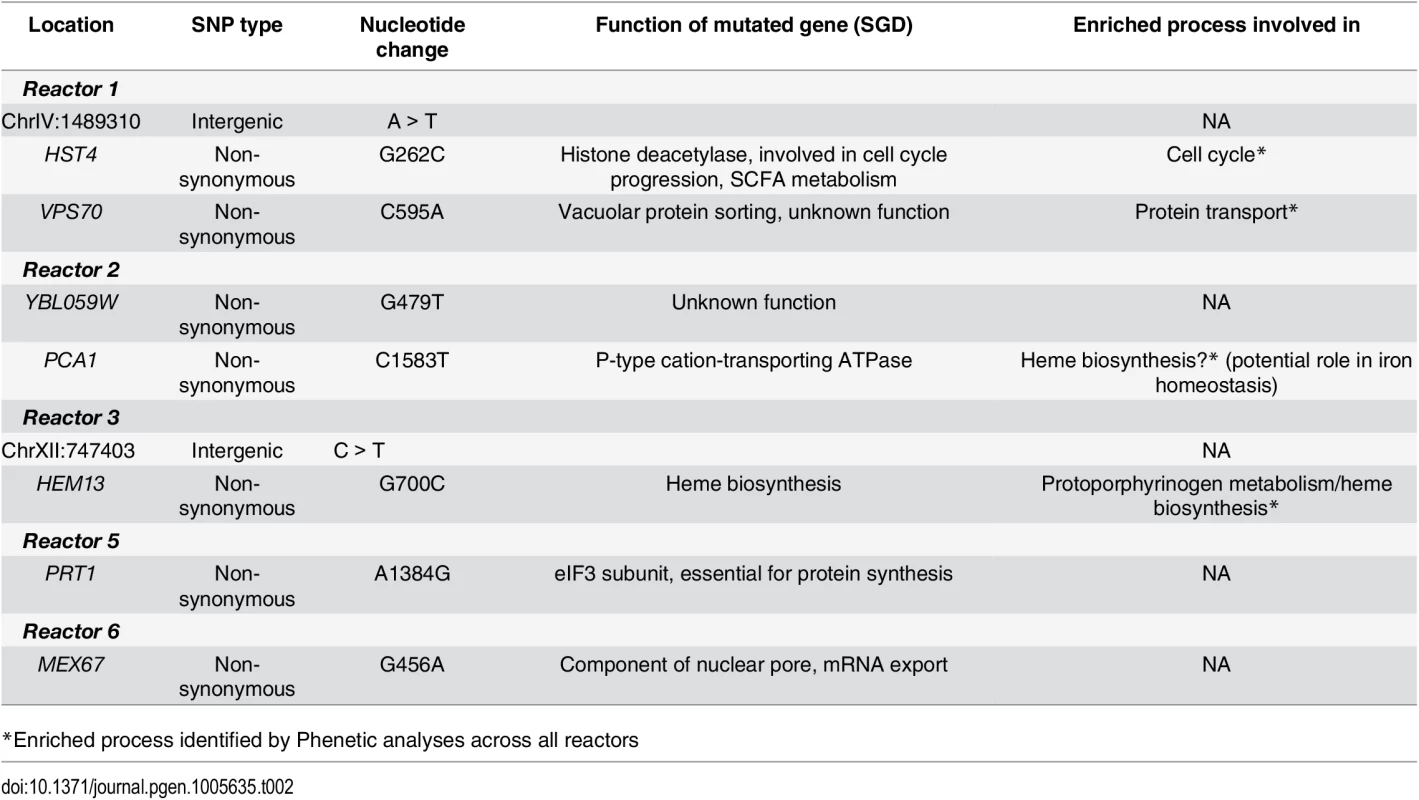
SNPs reaching high frequencies in the different evolved lineages were introduced in a haploid, non-ethanol tolerant strain. Gene functions were obtained from SGD, http://www.yeastgenome.org. These nine SNPs were subsequently introduced into the ancestral haploid strain. The effect of these mutations was assessed by high-throughput competition experiments [64], in 0, 4, 6 and 8% (v/v) EtOH conditions, with glucose as a carbon source (Fig 8, S14 and S15 Figs and S6 Table). Several mutants show a clear increase in fitness, with the fitness effect often depending on the concentration of ethanol in the medium. Most mutants show slightly increased fitness in medium without added ethanol, with further increases in relative fitness with increasing exogeneous ethanol concentrations. Mutants in PRT1 and MEX67 are less fit than the WT in non-ethanol conditions, but show increased fitness in higher ethanol levels. MEX67 is a poly (A) RNA binding protein involved in nuclear mRNA export. PRT1 encodes the eIF3b subunit of the eukaryotic translation initiation factor 3 (eIF3). The increased ethanol tolerance associated with these mutations hints at translation processes as targets of ethanol. Interestingly, a recent study in E. coli showed that ethanol negatively impacts transcription as well as translation [6]. However, if and how mutations in MEX67 and PRT1 might mitigate this effect and contribute to ethanol tolerance remains unknown.
Fig. 8. Single mutations present in evolved populations can increase ethanol tolerance of a non-adapted strain. 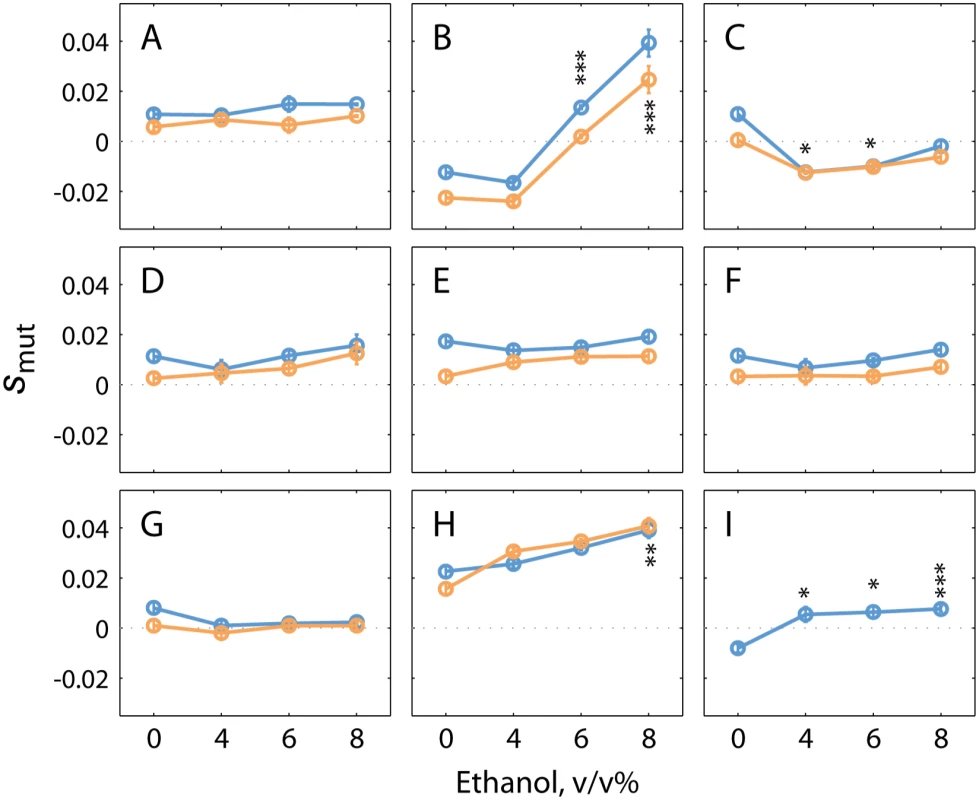
Plots show the average selection coefficient (smut) as a function of ethanol concentration for (A) pca1C1583T, (B) prt1A1384G, (C) ybl059wG479T, (D) intergenic ChrIV A1489310T, (E) hem13G700C, (F) intergenic ChrXII C747403T, (G) hst4G262C, (H) vps70C595A, and (I) mex67G456A. Superscripts denote the exact nucleotide change in each of the mutants tested. YECitrine-tagged mutants were competed with the mCherry-tagged parental strain (orange dots); dye-swap experiments were carried out by competing the mCherry-tagged mutants with the YECitrine parental strain (blue dots), except for (I). Error bars show the S.E.M. from three experimental replicates. Asterisk show P-values from the one-way ANOVA tests of the mean differences in 4–8% ethanol compared to fitness in 0% ethanol: * p < 0.05; ** p < 0.01; ***p < 0.005. P values can be found in S7 Table. Of all mutations introduced into the ancestral strain, vps70C595A provided the largest fitness increase. VPS70 is putatively involved in sorting of vacuolar carboxypeptidase Y to the vacuole [65]. A mutation in VPS70 has been recently identified by members of our team as a determinant of ethanol tolerance in a Brazilian bioethanol strain [66]. Interestingly, the VPS70 mutation in our evolved ethanol-tolerant lineages alters the same amino acid as the mutation present in the industrial ethanol tolerant strain (Goovaerts A. and Thevelein J.M., personal communication). The fact that one of the mutations identified in our evolved lineages was also found in an industrially used bio-ethanol strain underscores the potential of our approach to find biologically relevant mutations for increased ethanol tolerance. Other members of the VPS family have been previously implicated in ethanol tolerance as well, but also here the exact molecular mechanism through which they could increase ethanol tolerance is still unclear [10,12,67]. To our knowledge, none of the other genes investigated in this study have been previously implicated in ethanol tolerance nor have these mutations been found in natural or industrial yeasts so far. This implies that these mutations could be prime candidates to improve the ethanol tolerance and production of existing industrial yeasts [68].
Discussion
Many fundamental questions on the dynamics and genetics of adaptation to a complex and severe stress such as ethanol are still unanswered. Which processes contribute to ethanol tolerance? Which type and number of mutations are needed and/or sufficient, and how do they penetrate and fix within populations? To what extent are these mutations and the pathways they affect predictable? To address these questions, we performed high-coverage whole-genome sequencing of clones and populations isolated throughout a two-year evolution experiment and characterized their adaptation to increasing ethanol levels. This allowed us to paint a detailed picture of the mutational dynamics in our evolving lineages.
Our study demonstrates how many different evolutionary mechanisms all come together to provide adaptation to a severe and complex stress. Specifically, we find that adaptation to high ethanol levels involves changes in ploidy, copy number variation and the appearance of mutator phenotypes, with evolving populations showing strong clonal interference. Although these mechanisms have been observed in other studies (see, for example, [24,27,32,69–71], different mechanisms are often observed and/or studied separately. More importantly, in traditional evolution studies, populations are exposed to a fixed concentration of antibiotic or limiting nutrient. Under these conditions, selection pressure is reduced or even eliminated when cells become resistant. In our experimental set-up, ethanol levels were stepwise increased over time so that cells were evolving under constant selection. High concentrations of ethanol also actively kill non-tolerant cells, so that cells not adapted to the higher ethanol concentrations die and/or are washed out of the turbidostat. Taken together, our study combines aspects of traditional evolution studies with principles used in morbidostat experiments [27]. This set-up is likely a better representation of what happens when a population gradually penetrates and adapts to a new niche. Under such severe and continuous stress conditions (near-morbidostats), many cells fail to survive. This also implies that the traditional way of measuring evolutionary time by counting the number of generations may become problematic as cell death and mutations contributing to survival instead of replication become increasingly important. This phenomenon can also result in extremely quick sweeps, because a mutation cannot only penetrate a population because it increases the replication rate, but also because it protects against death. Moreover, as cell replication slows down, the number of mutations that is not associated with DNA replication but rather with DNA damage and repair likely increases. It seems plausible that these mechanisms contribute to the very quick genetic sweeps that we observe in some reactors, most notably the quick sweep of diploidized cells in reactors 1 and 2. This could also explain why the number of mutations that we observe in our two-year experiment might seem high for only 200 generations when compared to experiments where cells are growing at higher rates and where 200 generations are often reached within only one or two months of evolution. While it is difficult to think of a better universal measure of evolutionary time, it is important to realize that in some cases, a combination of physical time and generations may be a more appropriate way to assess evolution.
We find extensive variation in genome size in our evolved cells; with aneuploidies in a large number of our evolved clones as well as convergence toward a diploid state in our initially haploid and tetraploid populations. Becoming diploid thus appears to be a frequently used strategy by cells of different ploidy which effectively increases ethanol tolerance. Convergence to diploidy has been observed in other laboratory evolution experiments [25,36,72]. In contrast to these studies, we could demonstrate a clear fitness advantage of our diploid strains in ethanol. Although further research is needed to fully understand this fitness advantage, changes in gene expression and/or cell size could be important factors contributing to the increased fitness of diploid cells [41,73,74]. Unfortunately, this convergent evolution prevented us from performing a detailed analysis on the difference in adaptive strategies employed by haploids, diploids and tetraploids to increase ethanol tolerance.
Aneuploidy and copy-number variation are increasingly recognized as common themes in rapid adaptation [24,28,69,70]. It is believed that changes in the copy number of chromosomes or chromosomal fragments provide a relatively easily accessible way to change expression levels of specific key genes [75,76], and these CNVs can provide large, usually condition-specific, fitness effects [77]. However, such large-scale changes in the genome likely also have some unwanted, detrimental side effects, such as imbalance between gene products and genome destabilization [78–80]. Evolving populations are believed to gradually replace adaptive CNVs with more specific mutations that show fewer pleiotropic effects [28]. Notably, several of our evolved clones, isolated from different reactors, carry an extra copy of chromosome III and/or ChrXII; pointing towards a potential adaptive benefit of this specific aneuploidy. Interestingly, we find that clones isolated at later time points have a specific, smaller region of chromosome XII amplified (see also S3 File); indicating a more refined solution. GO enrichment and network analyses of the repeatedly amplified region of ChrXII (position 657500–818000) hints at cell wall formation as one of the key processes affected by these amplifications. Previous studies have indeed shown that cell wall stability is a key factor involved in ethanol tolerance [18].
Apart from diploidization of our evolving lineages, another example of parallelism at the phenotypic level is the appearance of a mutator phenotype in two of our six evolving populations. The sweep of the MSH2 mutation in reactor 2 is likely caused by a so-called hitchhiking event, with the high mutation rates in the MSH2 mutant leading to the appearance of one or several beneficial mutations that drive the selective sweep.
Parallelism at the genotypic level is less clear: we find few mutations and mutated genes shared between the different evolved lineages. Interestingly, nine (ACE2, POL3, PUF4, GFA1, UTH1, JID1, RIM15, ATG11 and VPS74) out of the 28 genes (32%) that were found hit in multiple reactors have been previously implicated in ethanol tolerance. Applying different types of network and enrichment analyses revealed functional modules affected in several of the adapted populations. These pathways include response to stress, intracellular signal transduction, cell cycle and pathways related to membrane composition and organization (such as isoprenoid metabolism, glycerophospholipid catabolism and fatty-acyl-coA metabolism). For some of these pathways, further work is needed to clarify their exact involvement in ethanol tolerance.
To investigate the phenotypic effect of mutations present in our evolved lineages, we performed high-throughput fitness measurements in different ethanol concentrations. While our evolved lineages contained multiple mutations, single mutations reaching high frequency in the evolved populations could already significantly increase ethanol tolerance when introduced into the ancestral, non-evolved haploid strain. Moreover, several of these single mutations selected for further study also showed a (modest) fitness benefit in conditions with no ethanol, with greater increases as ethanol levels rise. The number of mutations identified in this study was too large to investigate the fitness effect of each individual mutation. However, our strategy to select mutations that reached fixation in the evolved populations clearly proved successful, identifying as many as 4 mutations (out of 9 tested) that confer a fitness advantage in high ethanol environments. Mutations in genes linked to processes identified as affected across our different reactors—such as protein transport (VPS70)–indeed increased fitness in EtOH. This underscores the potential of PheNetic, a sub-network based selection method for identifying adaptive mutations. Five of the mutations tested did not significantly increase fitness, although they reached high frequency in our adapted populations. This is indicative of hitch-hiking with other, beneficial mutations; or possible epistatic interactions with other mutations.
Why then would not all feral yeasts show high ethanol tolerance, if it appears so easy to attain? Firstly, it seems plausible that not all yeasts are confronted with selection for high ethanol tolerance. Furthermore, it is important to note that we have not tested the fitness of the mutants under many different conditions that mimic the natural habitats of yeasts. It seems likely that some of the mutations identified in this study would result in lower fitness in other environments [81,82]. Moreover, we have not investigated the effect of combined mutations. While it is possible that combining different mutations could increase ethanol tolerance even further, it also seems likely that some mutations and/or specific combinations of mutations could lead to reduced fitness in different low and/or high ethanol environments. Indeed, while our clones have increased fitness in EtOH, we observe that fitness of several of our evolved clones (containing multiple other mutations apart from the ones investigated in this study) decreased in medium without exogenous EtOH (see S16 Fig). These results are indicative of antagonistic pleiotropy: the specific mutations present in our evolved clones increase fitness in one condition (high ethanol, which was selected for), whereas they reduce fitness in other environments.
Ethanol resistance is an important trait for the survival of feral yeasts in nature because the ethanol produced inhibits growth of competing microorganisms, while it serves as a carbon source in later stages of growth, when all fermentable sugars are depleted (the so-called make-accumulate-consume strategy [83–85]). Our results suggest that adaptation to high ethanol is complex and can be reached through different mutational pathways. Apart from yielding insight into the evolutionary mechanisms leading to such complex and ecologically important phenotypes, our study is also of considerable industrial importance. Several of the mutations identified in this study may be useful to increase the ethanol tolerance of industrial strains used for the production of alcoholic beverages or biofuels.
Materials and Methods
Strains used in this study
Starting strains for the evolution experiment are all derived from the haploid prototrophic S288c strain FY5 [86]. To prevent clumping of cells during the evolution experiment, the flocculation genes FLO1, FLO10 and FLO11 were deleted in this strain using deletion cassettes based on pUG6, conferring resistance to G-418 disulfate [87]. Markers were removed through the Cre/LoxP technique using pSH65 [88]. Mating type switching of this strain was then performed, using plasmid pSB283, to create isogenic diploid and tetraploid strains. Fluorescent versions of strains (YECitrine or mCherry tagged) were constructed by integrating fluorescent markers at an intergenic, neutral region of chromosome II. A full list of strains used, with their complete genotype, can be found in S4 Table. Primers used for strain construction and verification can be found in S5 Table.
Long-term selection
Populations were founded in 400 mL ethanol containing media. Media contained 10 g/L yeast extract, 20g/L bactopeptone, 4% (w/v) glucose, 0.001% (v/v) Rhodorsil, Antifoam Silicone 46R, chloramphenicol (50 μg/mL) and increasing concentrations of ethanol. Populations were maintained at an average population size of 1010 cells. After 25 generations, the level of EtOH in the media was increased each time (starting at 6% (v/v) and reaching 12% at 200 generations).
Turbidostat cultures were maintained using Sixfors reactors (Infors) at 30°C, pH was kept constant at 5.0 with continuous mixing at 250 rpm in aerobic conditions. At regular times, a population sample was obtained from each of the cultures for further analyses and stored in glycerol at -80°C. For DNA extraction purposes, a population cell pellet was also frozen down at -80°C.
Fitness determination
Fitness for all evolved strains was determined in rich medium (YP, 2% (w/v) glucose) with 9% (v/v) ethanol, by competing strains against a YECitrine labeled ancestral strain. Cultures were pre-grown in YPD 6% ethanol. After 12h, wells of a 96 deep well plate were inoculated with equal numbers of labeled reference and unlabeled strains (~ 106 cells of each) and allowed to grow for around 10 generations. Outer wells only contained medium and acted as a buffer to prevent ethanol evaporation. Additionally, plates were closed with an adhesive seal and plastic lid, and parafilm was used to prevent ethanol evaporation. Cultures were regularly transferred to new medium to prevent nutrient depletion. The ratio of the two competitors was quantified at the initial and final time points by flow cytometry. Data analysis was done in FlowJo version 10. Measurements were corrected for the small percentage of labeled, non-fluorescent cells that occurred even when the reference strain was cultured separately as well as for the cost of YFP expression in the labeled reference strain. For each fitness measurement, three independent replicates were performed. The selective advantage, s, of each strain was calculated as s = (ln(Uf/Rf)-ln (Ui/Ri))/T where U and R are the numbers of unlabeled and reference strain respectively, the subscripts refer to final and initial populations and T is the number of generations that reference cells have proliferated during the competition. The fitness of the unlabeled WT strain was designated 1, fitness of the evolved strains as 1+s.
Determination of cell ploidy
DNA content of evolved populations and evolved clones was determined by staining cells with propidiumiodide (PI) and analyzing 50 000 cells by flow cytometry on a BD Influx. The ancestral haploid and diploid strains used in the evolution experiment were used for calibration.
Whole genome sequencing
For evolved populations, genomic DNA was directly extracted from pellets that were frozen at the time of sample taking.
Evolved clones were selected from the different population samples by streaking glycerol stocks from the corresponding population samples on YPD plates. Swabs from each population were subsequently grown in YPD 6% EtOH and dilutions were plated on YPD plates with different ethanol concentrations (ranging from 8% to 10%; with a 0.5% stepwise increase in ethanol concentrations). From these plates, ethanol tolerant clones were selected and genomic DNA of these clones was extracted.
Genomic DNA was prepared using the Qiagen genomic tip kit. Final DNA concentrations were measured using Qubit. Paired-end sequencing libraries with a mean insert size of 500 bp were prepared and libraries were run on an Illumina HiSeq2000 (EMBL GeneCore facility, Heidelberg). Average sequencing coverage for clone and population sample is 80X and 500X respectively.
Variant calling, filtration and annotation
Adaptors removal and reads quality control were done by Trim Galore! (http://www.bioinformatics.babraham.ac.uk/projects/trim_galore/) with options (-q 30—length 50). The clean reads were then mapped reference S. cerevisiae genome (S288C, version genebank64) using Burrows Wheeler Alignment allowing maximum 50bp gaps [89]. To identify mutations we employed the BROAD Institute Genome Analysis Toolkit (GATK, version 3.1) [90]. We began by performing local realignment of reads around Indels, in order to eliminate false positives due to misalignment of reads, which was followed by a base recalibration step. We then performed SNP and Indel calling according to the GATK best practice recommendations. Population samples and single clone datasets were analyzed independently.
For population data, Indel calling was performed using the UnifiedGenotyper tool, and the identified variants were filtered based on the recommended criteria (QD < 2.0, FS > 200.0, ReadPosRankSum < -20 and InbreedingCoef < -0.8). For the SNPs, we used the UnifiedGenotyper to perform multi-sample SNP calling on all the population samples together. The resulting multi-sample callset was then used as a prior to call SNPs in all individual samples. The called SNPs were then subject to filtering (QD < 2.0, FS > 60, ReadPosRankSum < -8.0, MQ < 40, MQRankSum < -12.5). Furthermore, we filtered out SNPs that were called inside regions containing Indels. We also generated a list of all known repetitive regions and low complexity regions using the program RepeatMasker (http://www.repeatmasker.org/cgi-bin/WEBRepeatMasker), which were masked out from the assemblies. Finally, variants present in the ancestral strain were filtered out from all samples.
A similar approach was used to analyze the clonal samples. Ploidy level of each isolated clone was determined by PI staining and variant calling was performed with UnifiedGenotyper for haploid clones or HaplotypeCaller for diploid clones. Subsequently, SNPs and Indels were filtered based on the GATK best practice recommendations as mentioned above. The inbreeding coefficient filter was excluded in the clonal sample filtering for Indels, as it is a population-level metric. Annotation and effect prediction of all identified variants was performed using snpEff [91].
Final processing of the data was performed using a R script, which involved filtering out variants called within the sub-telomeric regions (15 kbps from the chromosome ends) and—for population samples—variants whose frequency did not change by more than 10% during the experiment. We used the infrastructure of the VSC—Flemish Supercomputer Center for these analyses. For improved performance, CPU multithreading (-nct) capabilities of GATK were utilized, providing up to 20 cores and maximum memory capacity of 64 GB per process.
Identification of Copy Number Variations (CNV)
CNVs in the samples were identified using Nexus Copy Number software, version 7.5 (http://www.biodiscovery.com/software/nexus-copy-number). Reads were accumulated in 500-base bins along all chromosomes, rejecting bins with less than 10 reads. Log2 ratios of copy numbers were then estimated from the read-depth counts in these intervals. We used the following calling parameters: minimum number of probes per segment of 5, significance threshold of p-value = 1 × 10^-9, percentage of removed outliers of 5% and the limits for copy gain and loss of+0.25 and -0.25, respectively.
Statistical analyses of genes hit multiple times
The p-values probabilities of genes being hit by a mutation a specific number of times were calculated using the binomial distribution. The number of draws was set to the total number of mutations found inside coding regions (i.e. 817); the number of successes was set to the number of times a specific gene was hit by a mutation (2 to 6); the probability of success was set to the specific ratio of the length of each gene hit multiple times (in nucleotides) over the entire coding content of the S. cerevisiae genome (9080922 nucleotides).
Haplotype reconstruction
Haplotype reconstruction was based on the approach described by [32]. Prior to the actual reconstruction, variants identified in the sequencing data were subject to further processing by excluding variants that were multi-allelic, variants that exhibited mixed zygosity in the isolated clones (i.e. homozygous in one clone and heterozygous in another clone) and variants that didn't reach the frequency of 0.2 at any point during the experiment. Haplotypes (or 'mutational cohorts') present in the remaining variants were next reconstructed using a Matlab script (kindly provided by the Desai lab) on a local machine running Matlab 2013a. Briefly, variants present in our dataset were clustered into haplotypes based on the Euclidean distance between their frequencies at specific time-points of the experiment. Afterward, frequencies of individual variants assigned to a specific haplotype were averaged to obtain the frequency of the haplotype itself. Frequencies of the identified haplotypes at specific time points (see S2 Table) were then used as source data to draw their approximate Muller diagram representations with Inkscape 0.48.4.
Enrichment and network analysis
Functionally meaningful terms enriched in the list of genes hit in our evolution experiment were identified using DAVID Tools [57,92]. An enrichment score cutoff value of 1.3 was used, equivalent to a p-value of 0.05 for term enrichment, as recommended by the authors [57].
Phenetic analyses
Intergenic mutations were discarded for the analysis. The interaction network used as input for PheNetic was composed of interactomics data obtained from KEGG [93] for metabolic interactions, String for protein-protein interactions [94] and Yeastract for protein-DNA interactions [95]. The total interaction network contains 6592 genes and 135266 interactions. This interaction network was converted to a probabilistic network using the distribution of the out-degrees of the terminal nodes of the network edges. By doing so, edges connecting nodes with a low out-degree will receive a high probability while edges connecting nodes with a high out-degree receive a low probability. Using lists of mutated genes as input, PheNetic will now infer that sub-network of the probabilistic network that best connects the mutated genes in the list over the probabilistic network. As the probabilistic network penalizes hub nodes, the inferred sub-network will therefore preferentially connect the mutated genes through the least connected parts of the network. This results in selecting the most specific parts of the network that can be associated with the mutated genes.
PheNetic was used to connect the mutated genes over the interaction network [58] with the following parameters: 100-best paths with a maximum path length of 4 were sampled between the different mutations in combination with a search tree cutoff of 0.01. As the size of the selected sub-network by PheNetic is dependent on both a cost parameter and the number of mutated genes in the input, different costs were used for the sub-network inference from different sizes of mutated gene lists. For the sub-network inference between all the mutated genes from the non-mutator reactors a cost of 0.25 was used, for the non-mutator reactors (1,3,4,5) a cost of 0.05 was used as they all have a similar amount of mutated genes, and for the mutator reactors (2 and 6) a cost of 0.5 was used. For more details, see S1 Text.
Network visualization and enrichment
The resulting networks were visualized using Cytoscape and a functional enrichment using the biological process terms of Gene Ontology in combination with the annotation of SGD of the sub-networks was performed using the Bingo plugin, version 2.44 [96]. The resulting enrichment results are listed in S6 File.
Construction of mutant strains
Selected mutations identified from the whole-genome sequencing data were introduced into the ancestral genetic background using the following protocol. First, a selectable marker conferring resistance to hygromycine was introduced near the genomic location of interest through homologous recombination. Part of this locus was then amplified together with the selectable marker using a forward primer containing the desired mutation. The resulting PCR product was then transformed into the ancestral strain; and presence of the mutation was verified by Sanger sequencing. Primers used can be found in S5 Table.
High-throughput competitive fitness measurements
YECitrine - or mCherry-tagged site-directed mutant strains were competed with the parental mCherry or YECitrine tagged strains, respectively, as described [64]. In brief, saturated cultures of mutant and parental strains were mixed in equivalent volumes and inoculated onto 150 μl of YNB-low fluorescent medium in 96-well microtiter plates (Corning 3585). Micro-cultures grew without shaking and were serial-diluted every 24 hrs for approximately 28 generations (7 days) in a fully automated robotic system (Tecan Freedom EVO200) that integrates a plate carrousel (Liconic STX110), a plate reader (Tecan Infinite M1000), a 96-channel pipetting head, an orbital plate shaker, and a robotic manipulator arm. The equipment was maintained in an environmental room at constant temperature (30°C) and relative humidity (70%). Fluorescence signal (mCherry: Ex 587 nm/5 nm and Em 610 nm/5 nm; YECitrine: Ex 514 nm/5 nm and Em 529 nm/5 nm) and absorbance at 600 nm were monitored every hour during the entire experiment. The YECitrine - or mCherry-tagged parental strains were competed to each other for normalization and monitored individually to determine background fluorescence signal. Fluorescence and absorbance output data was analyzed in Matlab as described [64] to obtain an average selection coefficient, smut, with its S.E.M. from three experimental replicates.
Accession numbers
All sequencing data are available from the NCBI Sequence Read Archive: Bioproject, accession number PRJNA292495.
Supporting Information
Zdroje
1. Steensels J, Verstrepen KJ (2014) Taming wild yeast: potential of conventional and nonconventional yeasts in industrial fermentations. Annu Rev Microbiol 68 : 61–80. doi: 10.1146/annurev-micro-091213-113025 24773331
2. Snoek T, Picca Nicolino M, Van den Bremt S, Mertens S, Saels V, et al. (2015) Large-scale robot-assisted genome shuffling yields industrial Saccharomyces cerevisiae yeasts with increased ethanol tolerance. Biotechnology for biofuels in press.
3. Mukherjee V, Steensels J, Lievens B, Van de Voorde I, Verplaetse A, et al. (2014) Phenotypic evaluation of natural and industrial Saccharomyces yeasts for different traits desirable in industrial bioethanol production. Appl Microbiol Biotechnol 98 : 9483–9498. doi: 10.1007/s00253-014-6090-z 25267160
4. Alexandre H, Ansanay-Galeote V, Dequin S, Blondin B (2001) Global gene expression during short-term ethanol stress in Saccharomyces cerevisiae. FEBS Lett 498 : 98–103. 11389906
5. Fujita K, Matsuyama A, Kobayashi Y, Iwahashi H (2006) The genome-wide screening of yeast deletion mutants to identify the genes required for tolerance to ethanol and other alcohols. FEMS Yeast Res 6 : 744–750. 16879425
6. Haft RJ, Keating DH, Schwaegler T, Schwalbach MS, Vinokur J, et al. (2014) Correcting direct effects of ethanol on translation and transcription machinery confers ethanol tolerance in bacteria. Proc Natl Acad Sci U S A 111: E2576–2585. doi: 10.1073/pnas.1401853111 24927582
7. Horinouchi T, Tamaoka K, Furusawa C, Ono N, Suzuki S, et al. (2010) Transcriptome analysis of parallel-evolved Escherichia coli strains under ethanol stress. BMC Genomics 11 : 579. doi: 10.1186/1471-2164-11-579 20955615
8. Lewis JA, Broman AT, Will J, Gasch AP (2014) Genetic architecture of ethanol-responsive transcriptome variation in Saccharomyces cerevisiae strains. Genetics 198 : 369–382. doi: 10.1534/genetics.114.167429 24970865
9. Lewis JA, Elkon IM, McGee MA, Higbee AJ, Gasch AP (2010) Exploiting natural variation in Saccharomyces cerevisiae to identify genes for increased ethanol resistance. Genetics 186 : 1197–1205. doi: 10.1534/genetics.110.121871 20855568
10. van Voorst F, Houghton-Larsen J, Jonson L, Kielland-Brandt MC, Brandt A (2006) Genome-wide identification of genes required for growth of Saccharomyces cerevisiae under ethanol stress. Yeast 23 : 351–359. 16598687
11. Ehrenreich IM, Torabi N, Jia Y, Kent J, Martis S, et al. (2010) Dissection of genetically complex traits with extremely large pools of yeast segregants. Nature 464 : 1039–1042. doi: 10.1038/nature08923 20393561
12. Hu XH, Wang MH, Tan T, Li JR, Yang H, et al. (2007) Genetic dissection of ethanol tolerance in the budding yeast Saccharomyces cerevisiae. Genetics 175 : 1479–1487. 17194785
13. Swinnen S, Schaerlaekens K, Pais T, Claesen J, Hubmann G, et al. (2012) Identification of novel causative genes determining the complex trait of high ethanol tolerance in yeast using pooled-segregant whole-genome sequence analysis. Genome Res 22 : 975–984. doi: 10.1101/gr.131698.111 22399573
14. Wohlbach DJ, Rovinskiy N, Lewis JA, Sardi M, Schackwitz WS, et al. (2014) Comparative genomics of Saccharomyces cerevisiae natural isolates for bioenergy production. Genome Biol Evol 6 : 2557–2566. 25364804
15. D'Amore T, Panchal CJ, Russell I, Stewart GG (1990) A study of ethanol tolerance in yeast. Crit Rev Biotechnol 9 : 287–304. 2178780
16. Ding J, Huang X, Zhang L, Zhao N, Yang D, et al. (2009) Tolerance and stress response to ethanol in the yeast Saccharomyces cerevisiae. Appl Microbiol Biotechnol 85 : 253–263. doi: 10.1007/s00253-009-2223-1 19756577
17. Ma M, Liu ZL (2010) Mechanisms of ethanol tolerance in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 87 : 829–845. doi: 10.1007/s00253-010-2594-3 20464391
18. Avrahami-Moyal L, Engelberg D, Wenger JW, Sherlock G, Braun S (2012) Turbidostat culture of Saccharomyces cerevisiae W303-1A under selective pressure elicited by ethanol selects for mutations in SSD1 and UTH1. FEMS Yeast Res 12 : 521–533. doi: 10.1111/j.1567-1364.2012.00803.x 22443114
19. Brown SW, Oliver SG (1982) Isolation of ethanol-tolerant mutants of yeast by continuous selection. Eur J Appl Microbiol Biotechnol 16 : 119–122.
20. Goodarzi H, Bennett BD, Amini S, Reaves ML, Hottes AK, et al. (2010) Regulatory and metabolic rewiring during laboratory evolution of ethanol tolerance in E. coli. Mol Syst Biol 6 : 378. doi: 10.1038/msb.2010.33 20531407
21. Stanley D, Fraser S, Chambers PJ, Rogers P, Stanley GA (2010) Generation and characterisation of stable ethanol-tolerant mutants of Saccharomyces cerevisiae. J Ind Microbiol Biotechnol 37 : 139–149. doi: 10.1007/s10295-009-0655-3 19902282
22. Cakar ZP, Seker UO, Tamerler C, Sonderegger M, Sauer U (2005) Evolutionary engineering of multiple-stress resistant Saccharomyces cerevisiae. FEMS Yeast Res 5 : 569–578. 15780656
23. Barrick JE, Yu DS, Yoon SH, Jeong H, Oh TK, et al. (2009) Genome evolution and adaptation in a long-term experiment with Escherichia coli. Nature 461 : 1243–1247. doi: 10.1038/nature08480 19838166
24. Gresham D, Desai MM, Tucker CM, Jenq HT, Pai DA, et al. (2008) The repertoire and dynamics of evolutionary adaptations to controlled nutrient-limited environments in yeast. PLoS Genet 4: e1000303. doi: 10.1371/journal.pgen.1000303 19079573
25. Hong J, Gresham D (2014) Molecular specificity, convergence and constraint shape adaptive evolution in nutrient-poor environments. PLoS Genet 10: e1004041. doi: 10.1371/journal.pgen.1004041 24415948
26. Oz T, Guvenek A, Yildiz S, Karaboga E, Tamer YT, et al. (2014) Strength of selection pressure is an important parameter contributing to the complexity of antibiotic resistance evolution. Mol Biol Evol 31 : 2387–2401. doi: 10.1093/molbev/msu191 24962091
27. Toprak E, Veres A, Michel JB, Chait R, Hartl DL, et al. (2011) Evolutionary paths to antibiotic resistance under dynamically sustained drug selection. Nat Genet 44 : 101–105. doi: 10.1038/ng.1034 22179135
28. Yona AH, Manor YS, Herbst RH, Romano GH, Mitchell A, et al. (2012) Chromosomal duplication is a transient evolutionary solution to stress. Proc Natl Acad Sci U S A 109 : 21010–21015. doi: 10.1073/pnas.1211150109 23197825
29. Barrick JE, Lenski RE (2009) Genome-wide mutational diversity in an evolving population of Escherichia coli. Cold Spring Harb Symp Quant Biol 74 : 119–129. doi: 10.1101/sqb.2009.74.018 19776167
30. Herron MD, Doebeli M (2013) Parallel evolutionary dynamics of adaptive diversification in Escherichia coli. PLoS Biol 11: e1001490. doi: 10.1371/journal.pbio.1001490 23431270
31. Kvitek DJ, Sherlock G (2013) Whole genome, whole population sequencing reveals that loss of signaling networks is the major adaptive strategy in a constant environment. PLoS Genet 9: e1003972. doi: 10.1371/journal.pgen.1003972 24278038
32. Lang GI, Rice DP, Hickman MJ, Sodergren E, Weinstock GM, et al. (2013) Pervasive genetic hitchhiking and clonal interference in forty evolving yeast populations. Nature 500 : 571–574. doi: 10.1038/nature12344 23873039
33. Payen C, Di Rienzi SC, Ong GT, Pogachar JL, Sanchez JC, et al. (2014) The dynamics of diverse segmental amplifications in populations of Saccharomyces cerevisiae adapting to strong selection. G3 (Bethesda) 4 : 399–409.
34. Gerrish PJ, Lenski RE (1998) The fate of competing beneficial mutations in an asexual population. Genetica 102–103 : 127–144. 9720276
35. Kao KC, Sherlock G (2008) Molecular characterization of clonal interference during adaptive evolution in asexual populations of Saccharomyces cerevisiae. Nat Genet 40 : 1499–1504. doi: 10.1038/ng.280 19029899
36. Gerstein AC, Chun HJ, Grant A, Otto SP (2006) Genomic convergence toward diploidy in Saccharomyces cerevisiae. PLoS Genet 2: e145. 17002497
37. Kellis M, Birren BW, Lander ES (2004) Proof and evolutionary analysis of ancient genome duplication in the yeast Saccharomyces cerevisiae. Nature 428 : 617–624. 15004568
38. Wolfe KH, Shields DC (1997) Molecular evidence for an ancient duplication of the entire yeast genome. Nature 387 : 708–713. 9192896
39. Fawcett JA, Maere S, Van de Peer Y (2009) Plants with double genomes might have had a better chance to survive the Cretaceous-Tertiary extinction event. Proc Natl Acad Sci U S A 106 : 5737–5742. doi: 10.1073/pnas.0900906106 19325131
40. Selmecki AM, Maruvka YE, Richmond PA, Guillet M, Shoresh N, et al. (2015) Polyploidy can drive rapid adaptation in yeast. Nature 519 : 349–352. doi: 10.1038/nature14187 25731168
41. Galitski T, Saldanha AJ, Styles CA, Lander ES, Fink GR (1999) Ploidy regulation of gene expression. Science 285 : 251–254. 10398601
42. Ohno S (1970) Evolution by gene duplication. Berlin: Springer-Verlag.
43. Semon M, Wolfe KH (2007) Consequences of genome duplication. Curr Opin Genet Dev 17 : 505–512. 18006297
44. Andalis AA, Storchova Z, Styles C, Galitski T, Pellman D, et al. (2004) Defects arising from whole-genome duplications in Saccharomyces cerevisiae. Genetics 167 : 1109–1121. 15280227
45. Song K, Lu P, Tang K, Osborn TC (1995) Rapid genome change in synthetic polyploids of Brassica and its implications for polyploid evolution. Proc Natl Acad Sci U S A 92 : 7719–7723. 7644483
46. Storchova Z, Breneman A, Cande J, Dunn J, Burbank K, et al. (2006) Genome-wide genetic analysis of polyploidy in yeast. Nature 443 : 541–547. 17024086
47. Anderson JB, Sirjusingh C, Ricker N (2004) Haploidy, diploidy and evolution of antifungal drug resistance in Saccharomyces cerevisiae. Genetics 168 : 1915–1923. 15371350
48. Gerstein AC, Cleathero LA, Mandegar MA, Otto SP (2011) Haploids adapt faster than diploids across a range of environments. J Evol Biol 24 : 531–540. doi: 10.1111/j.1420-9101.2010.02188.x 21159002
49. Zeyl C, Vanderford T, Carter M (2003) An evolutionary advantage of haploidy in large yeast populations. Science 299 : 555–558. 12543972
50. Lynch M, Sung W, Morris K, Coffey N, Landry CR, et al. (2008) A genome-wide view of the spectrum of spontaneous mutations in yeast. Proc Natl Acad Sci U S A 105 : 9272–9277. doi: 10.1073/pnas.0803466105 18583475
51. Giraud A, Matic I, Tenaillon O, Clara A, Radman M, et al. (2001) Costs and benefits of high mutation rates: adaptive evolution of bacteria in the mouse gut. Science 291 : 2606–2608. 11283373
52. McDonald MJ, Hsieh YY, Yu YH, Chang SL, Leu JY (2012) The evolution of low mutation rates in experimental mutator populations of Saccharomyces cerevisiae. Curr Biol 22 : 1235–1240. doi: 10.1016/j.cub.2012.04.056 22727704
53. Pal C, Macia MD, Oliver A, Schachar I, Buckling A (2007) Coevolution with viruses drives the evolution of bacterial mutation rates. Nature 450 : 1079–1081. 18059461
54. Sniegowski PD, Gerrish PJ, Lenski RE (1997) Evolution of high mutation rates in experimental populations of E. coli. Nature 387 : 703–705. 9192894
55. Drotschmann K, Clark AB, Tran HT, Resnick MA, Gordenin DA, et al. (1999) Mutator phenotypes of yeast strains heterozygous for mutations in the MSH2 gene. Proc Natl Acad Sci U S A 96 : 2970–2975. 10077621
56. Lang GI, Parsons L, Gammie AE (2013) Mutation rates, spectra, and genome-wide distribution of spontaneous mutations in mismatch repair deficient yeast. G3 (Bethesda) 3 : 1453–1465.
57. Huang da W, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4 : 44–57. doi: 10.1038/nprot.2008.211 19131956
58. De Maeyer D, Renkens J, Cloots L, De Raedt L, Marchal K (2013) PheNetic: network-based interpretation of unstructured gene lists in E. coli. Mol Biosyst 9 : 1594–1603. doi: 10.1039/c3mb25551d 23591551
59. De Maeyer D, Weytjens B, Renkens J, De Raedt L, Marchal K (2015) PheNetic: network-based interpretation of molecular profiling data. Nucleic Acids Res.
60. Kubota S, Takeo I, Kume K, Kanai M, Shitamukai A, et al. (2004) Effect of ethanol on cell growth of budding yeast: genes that are important for cell growth in the presence of ethanol. Biosci Biotechnol Biochem 68 : 968–972. 15118337
61. Gasch AP, Spellman PT, Kao CM, Carmel-Harel O, Eisen MB, et al. (2000) Genomic expression programs in the response of yeast cells to environmental changes. Mol Biol Cell 11 : 4241–4257. 11102521
62. Lu C, Brauer MJ, Botstein D (2009) Slow growth induces heat-shock resistance in normal and respiratory-deficient yeast. Mol Biol Cell 20 : 891–903. doi: 10.1091/mbc.E08-08-0852 19056679
63. Brauer MJ, Huttenhower C, Airoldi EM, Rosenstein R, Matese JC, et al. (2008) Coordination of growth rate, cell cycle, stress response, and metabolic activity in yeast. Mol Biol Cell 19 : 352–367. 17959824
64. DeLuna A, Vetsigian K, Shoresh N, Hegreness M, Colon-Gonzalez M, et al. (2008) Exposing the fitness contribution of duplicated genes. Nat Genet 40 : 676–681. doi: 10.1038/ng.123 18408719
65. Bonangelino CJ, Chavez EM, Bonifacino JS (2002) Genomic screen for vacuolar protein sorting genes in Saccharomyces cerevisiae. Mol Biol Cell 13 : 2486–2501. 12134085
66. Duitama J, Sanchez-Rodriguez A, Goovaerts A, Pulido-Tamayo S, Hubmann G, et al. (2014) Improved linkage analysis of Quantitative Trait Loci using bulk segregants unveils a novel determinant of high ethanol tolerance in yeast. BMC Genomics 15 : 207. doi: 10.1186/1471-2164-15-207 24640961
67. Teixeira MC, Raposo LR, Mira NP, Lourenco AB, Sa-Correia I (2009) Genome-wide identification of Saccharomyces cerevisiae genes required for maximal tolerance to ethanol. Appl Environ Microbiol 75 : 5761–5772. doi: 10.1128/AEM.00845-09 19633105
68. Steensels J, Snoek T, Meersman E, Nicolino MP, Voordeckers K, et al. (2014) Improving industrial yeast strains: exploiting natural and artificial diversity. FEMS Microbiol Rev 38 : 947–995. doi: 10.1111/1574-6976.12073 24724938
69. Dunham MJ, Badrane H, Ferea T, Adams J, Brown PO, et al. (2002) Characteristic genome rearrangements in experimental evolution of Saccharomyces cerevisiae. Proc Natl Acad Sci U S A 99 : 16144–16149. 12446845
70. Rancati G, Pavelka N, Fleharty B, Noll A, Trimble R, et al. (2008) Aneuploidy underlies rapid adaptive evolution of yeast cells deprived of a conserved cytokinesis motor. Cell 135 : 879–893. doi: 10.1016/j.cell.2008.09.039 19041751
71. Wildenberg GA, Murray AW (2014) Evolving a 24-hr oscillator in budding yeast. Elife 3.
72. Gerstein AC, Otto SP (2012) Cryptic fitness advantage: diploids invade haploid populations despite lacking any apparent advantage as measured by standard fitness assays. PLoS One 6: e26599.
73. Wu CY, Rolfe PA, Gifford DK, Fink GR (2010) Control of transcription by cell size. PLoS Biol 8: e1000523. doi: 10.1371/journal.pbio.1000523 21072241
74. Zorgo E, Chwialkowska K, Gjuvsland AB, Garre E, Sunnerhagen P, et al. (2013) Ancient evolutionary trade-offs between yeast ploidy states. PLoS Genet 9: e1003388. doi: 10.1371/journal.pgen.1003388 23555297
75. Pavelka N, Rancati G, Zhu J, Bradford WD, Saraf A, et al. (2010) Aneuploidy confers quantitative proteome changes and phenotypic variation in budding yeast. Nature 468 : 321–325. doi: 10.1038/nature09529 20962780
76. Torres EM, Sokolsky T, Tucker CM, Chan LY, Boselli M, et al. (2007) Effects of aneuploidy on cellular physiology and cell division in haploid yeast. Science 317 : 916–924. 17702937
77. Sunshine AB, Payen C, Ong GT, Liachko I, Tan KM, et al. (2015) The fitness consequences of aneuploidy are driven by condition-dependent gene effects. PLoS Biol 13: e1002155. doi: 10.1371/journal.pbio.1002155 26011532
78. Pavelka N, Rancati G, Li R (2010) Dr Jekyll and Mr Hyde: role of aneuploidy in cellular adaptation and cancer. Curr Opin Cell Biol 22 : 809–815. doi: 10.1016/j.ceb.2010.06.003 20655187
79. Sheltzer JM, Amon A (2011) The aneuploidy paradox: costs and benefits of an incorrect karyotype. Trends Genet 27 : 446–453. doi: 10.1016/j.tig.2011.07.003 21872963
80. Sheltzer JM, Blank HM, Pfau SJ, Tange Y, George BM, et al. (2011) Aneuploidy drives genomic instability in yeast. Science 333 : 1026–1030. doi: 10.1126/science.1206412 21852501
81. Koschwanez JH, Foster KR, Murray AW (2011) Sucrose utilization in budding yeast as a model for the origin of undifferentiated multicellularity. PLoS Biol 9: e1001122. doi: 10.1371/journal.pbio.1001122 21857801
82. Wenger JW, Piotrowski J, Nagarajan S, Chiotti K, Sherlock G, et al. (2011) Hunger artists: yeast adapted to carbon limitation show trade-offs under carbon sufficiency. PLoS Genet 7: e1002202. doi: 10.1371/journal.pgen.1002202 21829391
83. Ihmels J, Bergmann S, Gerami-Nejad M, Yanai I, McClellan M, et al. (2005) Rewiring of the yeast transcriptional network through the evolution of motif usage. Science 309 : 938–940. 16081737
84. Rozpedowska E, Hellborg L, Ishchuk OP, Orhan F, Galafassi S, et al. (2011) Parallel evolution of the make-accumulate-consume strategy in Saccharomyces and Dekkera yeasts. Nat Commun 2 : 302. doi: 10.1038/ncomms1305 21556056
85. Thomson JM, Gaucher EA, Burgan MF, De Kee DW, Li T, et al. (2005) Resurrecting ancestral alcohol dehydrogenases from yeast. Nat Genet 37 : 630–635. 15864308
86. Brachmann CB, Davies A, Cost GJ, Caputo E, Li J, et al. (1998) Designer deletion strains derived from Saccharomyces cerevisiae S288C: a useful set of strains and plasmids for PCR-mediated gene disruption and other applications. Yeast 14 : 115–132. 9483801
87. Guldener U, Heck S, Fielder T, Beinhauer J, Hegemann JH (1996) A new efficient gene disruption cassette for repeated use in budding yeast. Nucleic Acids Res 24 : 2519–2524. 8692690
88. Gueldener U, Heinisch J, Koehler GJ, Voss D, Hegemann JH (2002) A second set of loxP marker cassettes for Cre-mediated multiple gene knockouts in budding yeast. Nucleic Acids Res 30: e23. 11884642
89. Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25 : 1754–1760. doi: 10.1093/bioinformatics/btp324 19451168
90. DePristo MA, Banks E, Poplin R, Garimella KV, Maguire JR, et al. (2011) A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat Genet 43 : 491–498. doi: 10.1038/ng.806 21478889
91. Cingolani P, Platts A, Wang le L, Coon M, Nguyen T, et al. (2012) A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly (Austin) 6 : 80–92.
92. Huang da W, Sherman BT, Lempicki RA (2009) Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res 37 : 1–13. doi: 10.1093/nar/gkn923 19033363
93. Kanehisa M, Goto S (2000) KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res 28 : 27–30. 10592173
94. Jensen LJ, Kuhn M, Stark M, Chaffron S, Creevey C, et al. (2009) STRING 8—a global view on proteins and their functional interactions in 630 organisms. Nucleic Acids Res 37: D412–416. doi: 10.1093/nar/gkn760 18940858
95. Teixeira MC, Monteiro P, Jain P, Tenreiro S, Fernandes AR, et al. (2006) The YEASTRACT database: a tool for the analysis of transcription regulatory associations in Saccharomyces cerevisiae. Nucleic Acids Res 34: D446–451. 16381908
96. Maere S, Heymans K, Kuiper M (2005) BiNGO: a Cytoscape plugin to assess overrepresentation of gene ontology categories in biological networks. Bioinformatics 21 : 3448–3449. 15972284
Štítky
Genetika Reprodukční medicína
Článek A Hereditary Enteropathy Caused by Mutations in the Gene, Encoding a Prostaglandin TransporterČlánek Exocyst-Dependent Membrane Addition Is Required for Anaphase Cell Elongation and Cytokinesis inČlánek Spindle-F Is the Central Mediator of Ik2 Kinase-Dependent Dendrite Pruning in Sensory Neurons
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2015 Číslo 11
-
Všechny články tohoto čísla
- Agricultural Genomics: Commercial Applications Bring Increased Basic Research Power
- Ernst Rüdin’s Unpublished 1922-1925 Study “Inheritance of Manic-Depressive Insanity”: Genetic Research Findings Subordinated to Eugenic Ideology
- Convergent Evolution During Local Adaptation to Patchy Landscapes
- The Locus Controls Age at Maturity in Wild and Domesticated Atlantic Salmon ( L.) Males
- A Hereditary Enteropathy Caused by Mutations in the Gene, Encoding a Prostaglandin Transporter
- Absence of Maternal Methylation in Biparental Hydatidiform Moles from Women with Maternal-Effect Mutations Reveals Widespread Placenta-Specific Imprinting
- Calibrating the Human Mutation Rate via Ancestral Recombination Density in Diploid Genomes
- Anaplastic Lymphoma Kinase Acts in the Mushroom Body to Negatively Regulate Sleep
- Connecting Replication and Repair: YoaA, a Helicase-Related Protein, Promotes Azidothymidine Tolerance through Association with Chi, an Accessory Clamp Loader Protein
- UFBP1, a Key Component of the Ufm1 Conjugation System, Is Essential for Ufmylation-Mediated Regulation of Erythroid Development
- Mosaic and Intronic Mutations in Explain the Majority of TSC Patients with No Mutation Identified by Conventional Testing
- Members of the Epistasis Group Contribute to Mitochondrial Homologous Recombination and Double-Strand Break Repair in
- QTL Mapping of Sex Determination Loci Supports an Ancient Pathway in Ants and Honey Bees
- Genetic Interactions Implicating Postreplicative Repair in Okazaki Fragment Processing
- Genomics of Cancer and a New Era for Cancer Prevention
- Adaptation to High Ethanol Reveals Complex Evolutionary Pathways
- Dynamics of Transcription Factor Binding Site Evolution
- Exocyst-Dependent Membrane Addition Is Required for Anaphase Cell Elongation and Cytokinesis in
- Enhancer Runaway and the Evolution of Diploid Gene Expression
- Cattle Sex-Specific Recombination and Genetic Control from a Large Pedigree Analysis
- Drosophila Mutants Model Cornelia de Lange Syndrome in Growth and Behavior
- Pleiotropic Effects of Immune Responses Explain Variation in the Prevalence of Fibroproliferative Diseases
- Leaderless Transcripts and Small Proteins Are Common Features of the Mycobacterial Translational Landscape
- Tissue-Specific Effects of Reduced β-catenin Expression on Mutation-Instigated Tumorigenesis in Mouse Colon and Ovarian Epithelium
- Genus-Wide Comparative Genomics of Delineates Its Phylogeny, Physiology, and Niche Adaptation on Human Skin
- Mapping of Craniofacial Traits in Outbred Mice Identifies Major Developmental Genes Involved in Shape Determination
- Conserved Genetic Interactions between Ciliopathy Complexes Cooperatively Support Ciliogenesis and Ciliary Signaling
- Metabolomic Quantitative Trait Loci (mQTL) Mapping Implicates the Ubiquitin Proteasome System in Cardiovascular Disease Pathogenesis
- DNA Repair Cofactors ATMIN and NBS1 Are Required to Suppress T Cell Activation
- Spindle-F Is the Central Mediator of Ik2 Kinase-Dependent Dendrite Pruning in Sensory Neurons
- Ernst Rüdin and the State of Science
- ABCs of Insect Resistance to Bt
- Epigenetic Control of O-Antigen Chain Length: A Tradeoff between Virulence and Bacteriophage Resistance
- Encodes Dual Oxidase, Which Acts with Heme Peroxidase Curly Su to Shape the Adult Wing
- The Fanconi Anemia Pathway Protects Genome Integrity from R-loops
- Controls Quantitative Variation in Maize Kernel Row Number
- Genome-Wide Association Study of Golden Retrievers Identifies Germ-Line Risk Factors Predisposing to Mast Cell Tumours
- Insect Resistance to Toxin Cry2Ab Is Conferred by Mutations in an ABC Transporter Subfamily A Protein
- A Cytosine Methytransferase Modulates the Cell Envelope Stress Response in the Cholera Pathogen
- Conserved piRNA Expression from a Distinct Set of piRNA Cluster Loci in Eutherian Mammals
- The Multi-allelic Genetic Architecture of a Variance-Heterogeneity Locus for Molybdenum Concentration in Leaves Acts as a Source of Unexplained Additive Genetic Variance
- The lncRNA Controls Cryptococcal Morphological Transition
- Sae2 Function at DNA Double-Strand Breaks Is Bypassed by Dampening Tel1 or Rad53 Activity
- A Tandem Duplicate of Anti-Müllerian Hormone with a Missense SNP on the Y Chromosome Is Essential for Male Sex Determination in Nile Tilapia,
- Ectodysplasin/NF-κB Promotes Mammary Cell Fate via Wnt/β-catenin Pathway
- The QTL within the Complex Involved in the Control of Tuberculosis Infection in Mice Is the Classical Class II Gene
- Identifying Loci Contributing to Natural Variation in Xenobiotic Resistance in
- Variation in Rural African Gut Microbiota Is Strongly Correlated with Colonization by and Subsistence
- A Flexible, Efficient Binomial Mixed Model for Identifying Differential DNA Methylation in Bisulfite Sequencing Data
- Competition between Heterochromatic Loci Allows the Abundance of the Silencing Protein, Sir4, to Regulate Assembly of Heterochromatin
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- UFBP1, a Key Component of the Ufm1 Conjugation System, Is Essential for Ufmylation-Mediated Regulation of Erythroid Development
- Metabolomic Quantitative Trait Loci (mQTL) Mapping Implicates the Ubiquitin Proteasome System in Cardiovascular Disease Pathogenesis
- Genus-Wide Comparative Genomics of Delineates Its Phylogeny, Physiology, and Niche Adaptation on Human Skin
- Encodes Dual Oxidase, Which Acts with Heme Peroxidase Curly Su to Shape the Adult Wing
Přihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



