-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariérní portál
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaRisk of Advanced Neoplasia in First-Degree Relatives with Colorectal Cancer: A Large Multicenter Cross-Sectional Study
Quintero and colleagues show that relatives of colorectal cancer patients have a higher risk of themselves developing the disease, as well as neoplasias. Screening guidelines should be accordingly adjusted.
Published in the journal: . PLoS Med 13(5): e32767. doi:10.1371/journal.pmed.1002008
Category: Research Article
doi: https://doi.org/10.1371/journal.pmed.1002008Summary
Quintero and colleagues show that relatives of colorectal cancer patients have a higher risk of themselves developing the disease, as well as neoplasias. Screening guidelines should be accordingly adjusted.
Introduction
Clinical guidelines recommend more intensive surveillance in first-degree relatives (FDR) of patients with colorectal cancer than in average-risk individuals, as their risk of developing colorectal cancer is 2 - to 4-fold higher [1–4]. Screening in this population is recommended from the age of 40 or 10 y before the youngest case in the immediate family, since the disease tends to develop about 10 y earlier in FDR than in the general population [5]. Colonoscopy every 5 [3,6–8] or 10 [3] y, depending on the number of relatives affected and age at cancer diagnosis, is the predominant screening strategy for these individuals, as it is the most effective procedure to detect and remove advanced adenomas. However, this recommendation is not supported by prospective studies.
Low-penetrance genetic alterations may favor earlier development of advanced adenomas or accelerate the transition from adenoma to carcinoma, increasing the risk of colorectal cancer in this population [9]. If so, this should be reflected in a higher prevalence and an earlier onset of advanced adenomas in FDRs of patients with colorectal cancer than in average-risk individuals (i.e., asymptomatic individuals over 50 y). However, the results of several studies are inconclusive. Some show an increased prevalence of advanced adenoma but are questionable due to small sample size [10] or a retrospective design [11,12], based on registries that could introduce a selection bias because they included patients with hereditary colorectal cancer syndromes, indication of colonoscopy was not specifically ascertained, and information on colonoscopy quality was not documented. On the other hand, few prospective studies that have specifically addressed this issue were underpowered to stratify for familial risk or did not include appropriate average-risk individuals [13–17]. Indeed, there is little evidence supporting the notion that the natural history and prognosis of non-syndromic familial colorectal cancer and sporadic colorectal cancer may differ. Overall, evidence only favors screening at a younger age in familial-risk individuals [5]. In fact, there are no large-scale studies comparing the prevalence of advanced adenomas in asymptomatic familial - and average-risk individuals stratified by number of affected relatives, age and gender. Therefore, clarification on this issue in different familial-risk groups is crucial to guide future screening recommendations for this population.
The current study aimed to compare the neoplastic findings on first screening colonoscopy and the predicted probability of advanced adenoma or cancer according to age and gender between asymptomatic FDR of patients with colorectal cancer and average-risk individuals.
Methods
Analysis Plan
The current study was planned in March 2011 as a post-hoc analysis of three prospective studies [18–20]: one study that assessed the perfomance of fecal immunochemical testing in asymptomatic FDR of patients with colorectal cancer [18], one randomized control trial (RCT) that assessed the efficacy of fecal immunochemical testing and colonoscopy as screening methods in asymptomatic FDR of patients with colorectal cancer [19], and one RCT that assessed the efficacy of fecal immunochemical testing and colonoscopy as screening methods in asymptomatic average-risk individuals [20]. Patient recruitment was completed by December 2011 and all colonoscopies were finished by March 2012. The database audit was carried out by EQ and FB and queries were solved between June 2013 and February 2014. Finally, data were analyzed between March and September 2014.
Study Design and Setting
The study includes prospectively collected cross-sectional data from asymptomatic whites undergoing their first lifetime screening colonoscopy between January 2006 and March 2012 (Fig 1), attended at six Spanish tertiary hospitals with specific high-risk colorectal cancer clinics responsible for the management of patients with hereditary colorectal cancer syndromes and familial colorectal cancer. In the two studies including asymptomatic familial-risk individuals the recruitment period was from January 2006 to December 2010 [19] and from January 2010 to December 2011 [18], respectively. In the RCT including asymptomatic average-risk individuals the recruitment period was initiated in June 2009, and finished in June 2011.
Fig. 1. Study enrollment and outcomes. 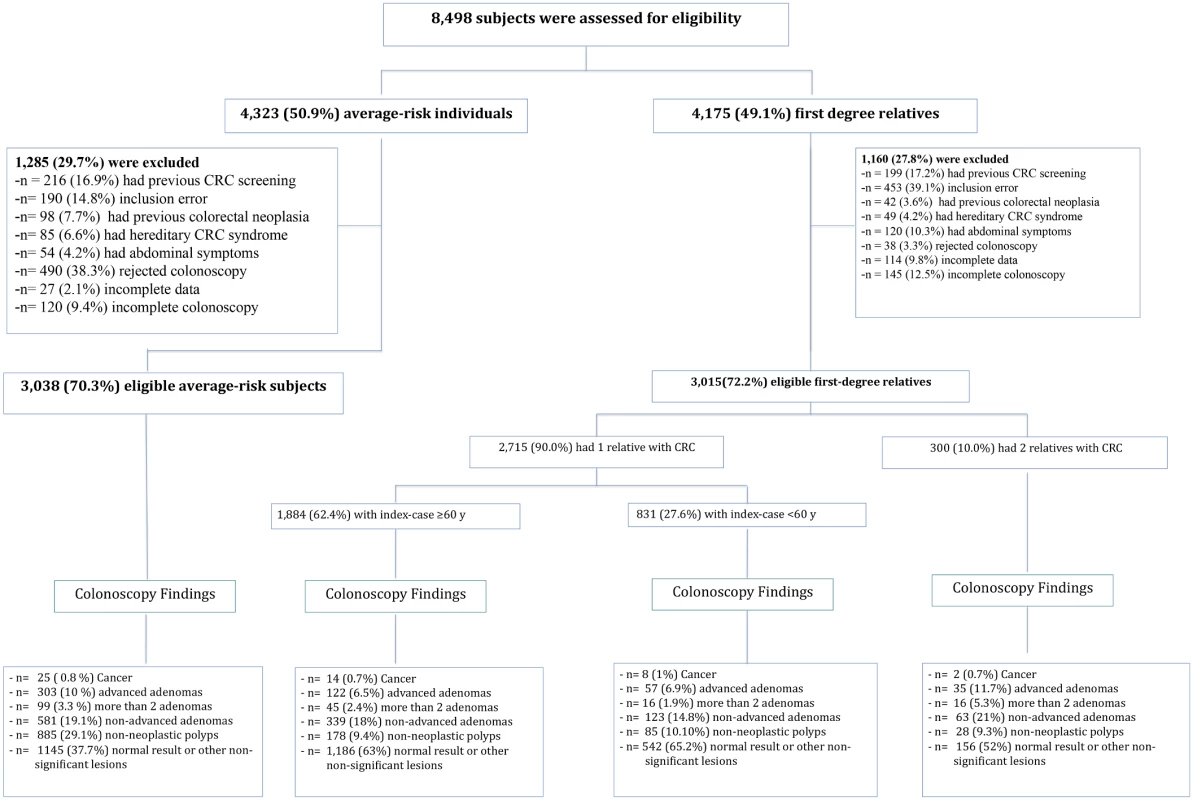
Asymptomatic average-risk individuals and familial-risk individuals undergoing their first lifetime screening colonoscopy were prospectively included in the six participating centers. All had high-quality colonoscopies (good or excellent bowel cleansing and cecal intubation). Study Sample
An asymptomatic familial-risk group comprising 4,175 asymptomatic FDR of patients with non-syndromic colorectal cancer was created by combining data from multiple sources: 2,322 (55.6%) of the individuals were consecutively included in two prospective trials [18,19] designed to analyze the efficacy of fecal immunochemical testing to detect advanced neoplasia, and the remaining FDR (n = 1,853; 44.4%) were recruited from those attending their respective colorectal cancer high-risk outpatient clinics due to family history of colorectal cancer, to complete sample size requirements. These individuals were mainly referred either by primary care physicians or colleagues from the oncology and surgery departments following local referring protocols. Screening recommendations for individuals with family history of colorectal cancer were based on the Spanish Clinical Practice Guideline for colorectal cancer screening [6]. In summary, colonoscopy every 5 y from age 40 (or 10 y before the youngest case in the immediate family) is recommended for individuals with one FDR with colorectal cancer diagnosed before the age of 60 or two or more FDR with colorectal cancer (regardless of age), and colonoscopy every 10 y from age 40 (or 10 y before the youngest case in the immediate family) for individuals with one FDR diagnosed over the age of 60. Baseline and outcome data from FDR included at colorectal cancer high-risk outpatient clinics were prospectively collected in an anonymized database, according to the Spanish personal data protection law.
A second group of asymptomatic individuals with average risk of colorectal cancer, served as a control group, and included 4,323 asymptomatic average-risk individuals (adults 50 to 69 y of age with no family history of colorectal cancer) assigned to the colonoscopy arm in the COLONPREV trial [20], a RCT designed to compare the efficacy of colonoscopy and biennial fecal immunochemical testing for reducing colorectal cancer-related mortality in the average-risk population. The study protocol is available online (www.nejm.org/doi/suppl/10.1056/NEJMoa1108895/suppl_file/nejmoa1108895_appendix.pdf) [20].
FDR of patients with colorectal cancer was determined during an interview by a gastroenterologist and using a questionnaire on demographic data as well as their own medical and family history of cancer at each participant center. Recruitment of individuals in the average-risk cohort has been previously described and included a personal interview about personal and family history of neoplasia performed at the local colorectal cancer screening office [20].
Inclusion criteria for the familial-risk group were the following: age 40–69 or 10 y younger than the youngest case in the family; complete colonoscopy (good or excellent bowel cleansing and cecal intubation), and colorectal cancer confirmed by written medical report. In the average-risk cohort, inclusion criteria were the following: age 50–69 y and no family history of colorectal neoplasia [20].
Exclusion criteria for both groups included the following: personal history of colorectal neoplasia, inflammatory bowel disease, familial history of hereditary colorectal cancer, abdominal symptoms needing further investigation, previous colorectal cancer screening with any technique, severe comorbidity, or refusal to participate.
Ethics Statement
The Clinical Research Ethics Committee of Hospital Clinic de Barcelona approved the study. The Ethical Committee of Hospital Universitario de Canarias [19] and the Galician Clinical Research Ethics Committee [18] approved the two studies involving familial-risk individuals The ethics committees of the 15 tertiary hospitals participating in the RCT involving average-risk individuals approved the study protocol. All participants belonging to the referred clinical trials [18–20] provided written informed consent and all authors had access to the study data and reviewed and approved the final manuscript. Familial-risk individuals included at the colorectal cancer high-risk outpatient clinics did not provide informed consent as they were waived by the IRB of Hospital Clinic de Barcelona.
Data Collection
In the familial-risk cohort, age, sex, number of relatives with colorectal cancer, kinship, and index-case age at diagnosis of colorectal cancer were recorded.
In all centers, colonoscopy quality was ensured following the guidelines of the Spanish Gastroenterological Association and the Spanish Society for Digestive Endoscopy [21]. All endoscopists involved in the study had personal experience of more than 200 colonoscopies per year and findings were documented in a standardized report form [20]. The quality of bowel preparation for each colonic segment was categorized as excellent or good versus poor or bad, as previously described [22]. Cases not meeting these requirements or with unexplored cecum were re-scheduled for colonoscopy.
At colonoscopy, the number and size of polyps were recorded. Polyps were classified according to proximal or distal location with respect to the splenic flexure. Adenomas ≥10 mm in size, with tubulovillous architecture, high-grade dysplasia or intramucosal carcinoma were classified as advanced adenomas. The presence of malignant cells observed beyond the muscularis mucosa was considered evidence of invasive cancer. Advanced neoplasia was defined as three or more non-advanced adenomas, advanced adenoma or invasive cancer. Patients were classified according to the most advanced lesion.
Sample Size and Data Analysis
The risk of advanced adenoma and advanced neoplasia according to familial - or average-risk was the main study outcome measure. As the familial-risk population comprised individuals with different neoplastic risk, we estimated the sample size needed to yield sufficient statistical power for significant results with respect to the main outcome measure in the least numerous subgroup, i.e., FDR with two FDR diagnosed with colorectal cancer at any age. Considering a ratio of 10 : 1 for one versus two relatives affected, an advanced adenoma detection rate of 7.8% in individuals with one FDR and 14.7% in those with two FDR with colorectal cancer, with an alpha error of 0.05 (two-sided) and a beta error of 0.10, the number of individuals having two FDR with colorectal cancer required was 235. Individuals having one family member with colorectal cancer diagnosed at <60 or ≥60 y were analyzed separately, as an age threshold of 60 y in the index-case is considered a colorectal cancer risk factor for their relatives [1,6,12].
Between-group differences in the risk of neoplastic lesions with respect to both overall or colon side-specific colorectal neoplasia detection rates were analyzed by multinomial logistic regression analysis when considering the 4-level categorical variables (normal colonoscopy, non-advanced adenoma, advanced adenoma, and cancer). In these analyses, the most severe colonoscopic finding was represented as an independent category. Binary logistic regression analysis was used when considering another colonoscopic findings included in the previous 4-level categorical variable. All logistic regression analyses were adjusted for age, gender, and center and reported as odds ratios (OR) with 95% confidence intervals (CI) [23].
A logistic regression model was developed to predict the probability of advanced neoplasia according to age, gender and familial groups. The observed frequencies and probabilities predicted from the regression equation were graphically represented stratifying individuals in 10-y subsets (20 to 70 for the affected familial groups, 50 to 70 for the control group) and by gender.
The analysis of resources, based on the number of colonoscopies needed to detect one advanced neoplasia, was performed by inverse marginal probability estimated by binary logistic regression analysis [24], adjusted for age and center. All analyses were performed using SPSS version 15.0 (SPSS Inc., Chicago, IL, United States) and STATA version 13.1 (Stata Corp, TX, US) software. All statistical tests were two-sided, and p-values <0.05 were considered statistically significant. STROBE guidelines were followed (S1 Text).
Results
Study Population
Overall, 8,498 individuals were assessed for eligibility, of whom 4,175 (49.1%) were assigned to the familial-risk group and 4,323 (50.9%) to the average-risk group. Of these, 1,160 (27.8%) individuals having FDR with colorectal cancer and 1,285 (29.7%) average-risk individuals were excluded (Fig 1) for a total of 3,015 individuals with familial-risk and 3,038 individuals with average-risk included in the analyses.
Demographic data of the familial-risk cohort are shown in Table 1.
Tab. 1. Demographic data. 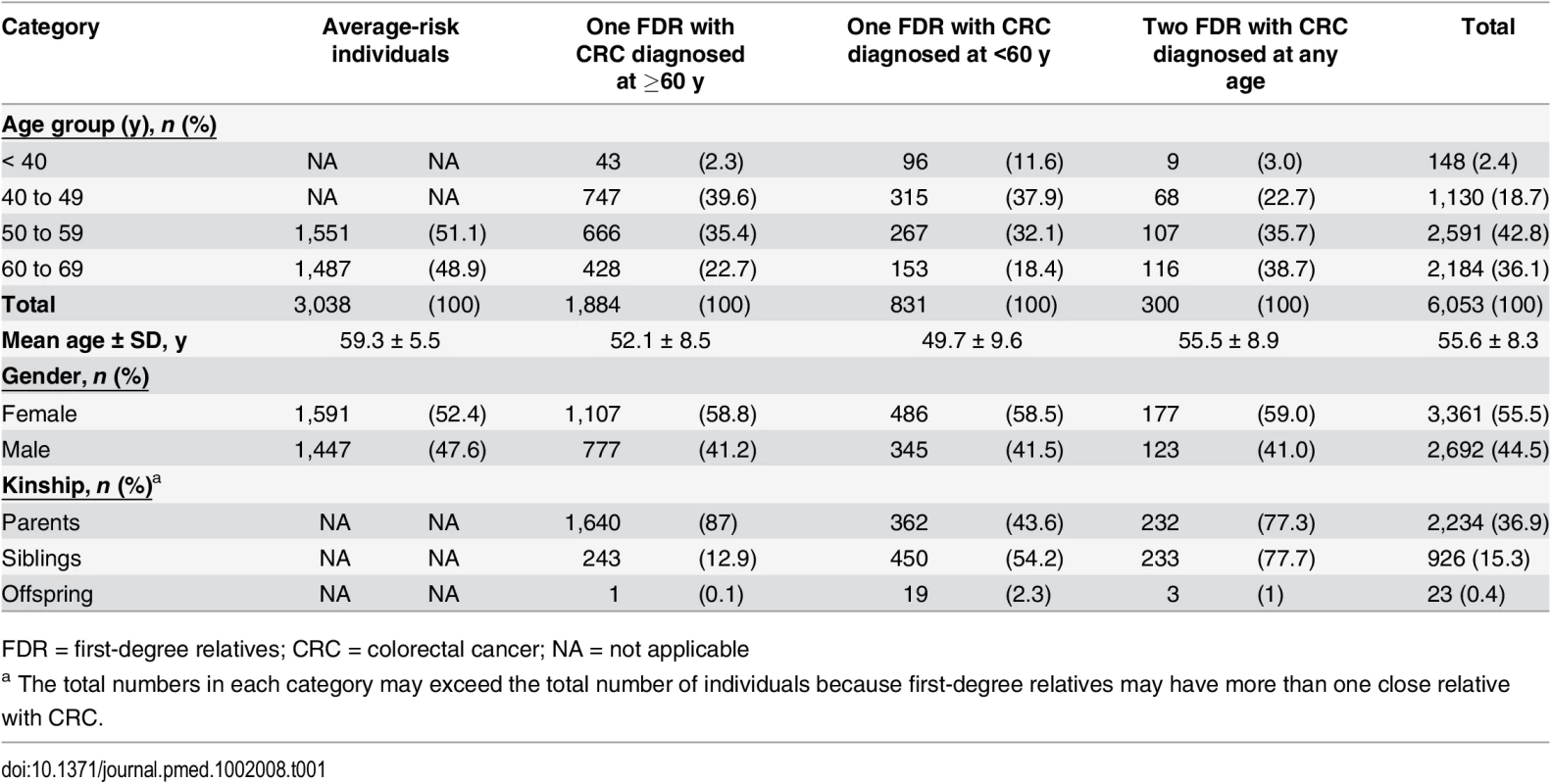
FDR = first-degree relatives; CRC = colorectal cancer; NA = not applicable The number of index-cases was 2,474 (median age 66.2 y). Of these, 1,399 (56.5%) were male and 786 (31.7%) were aged <60 y at diagnosis of colorectal cancer. Siblings with colorectal cancer predominated among individuals having two FDR affected compared to individuals having only one FDR with colorectal cancer (77.7% versus 25.5%, p < 0.001) (Table 1). Compared to average risk individuals, mean age of the familial-risk individuals was lower (51.8 ± 9.0 versus 59.3 ± 5.5 y, p < 0.001), as was the proportion of men (41.3% versus 47.6%, p < 0.001), as shown in Table 1.
Risk of Advanced Neoplasia
Colonoscopy findings are shown in Fig 1. Compared to average-risk individuals, in individuals having two FDR with colorectal cancer, we observed a higher prevalence and risk of non-advanced adenoma (OR 1.40; 95% CI 1.02 to 1.92, p = 0.03), advanced adenoma (OR 2.13; 95% CI 1.42 to 3.19, p < 0.001), three or more non-advanced adenomas (OR 2.16, 95% CI 1.21 to 3.84, p = 0.01), and advanced neoplasia (OR 1.90; 95% CI 1.36 to 2.66, p < 0.001) after adjusting for age, sex, and center (Table 2).
Tab. 2. Risk of colorectal neoplasia in familial- and average-risk groups. 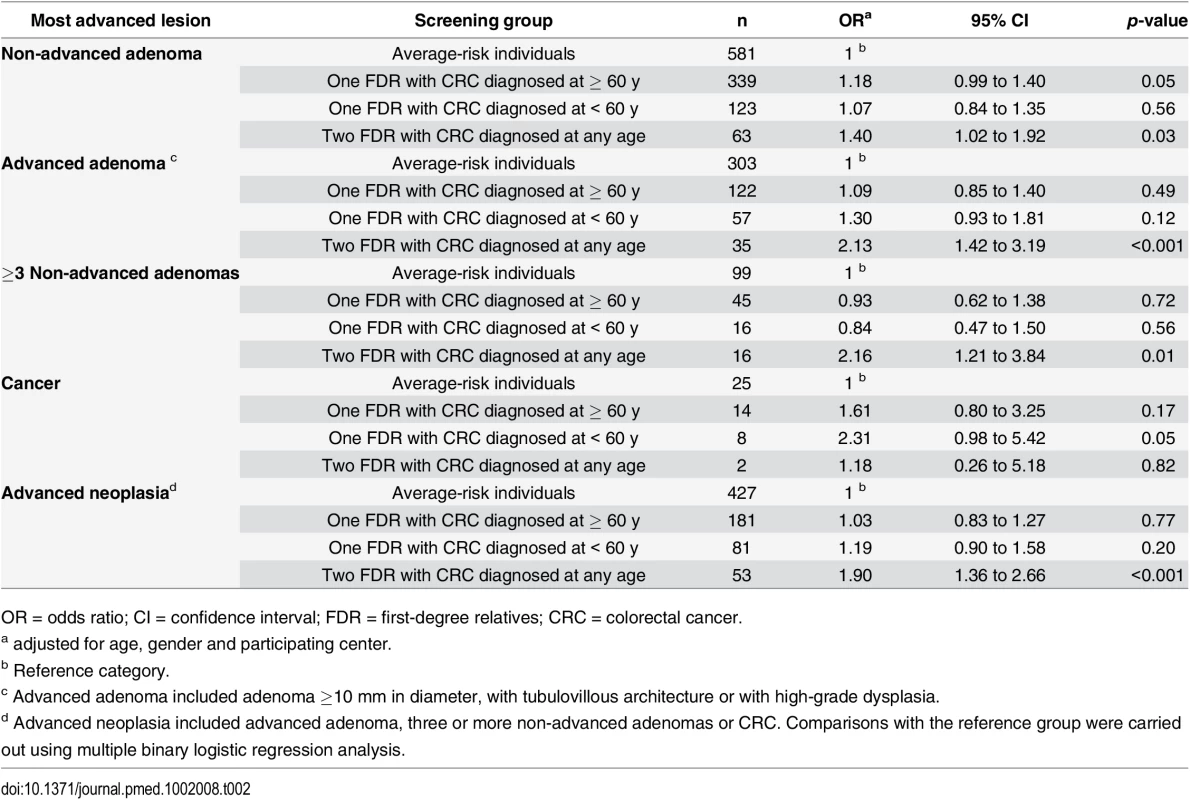
OR = odds ratio; CI = confidence interval; FDR = first-degree relatives; CRC = colorectal cancer. The probability of advanced neoplasia increased with age in both cohorts, as expected (Fig 2).
Fig. 2. Predicted probabilities of advanced colorectal neoplasia according to age and gender. 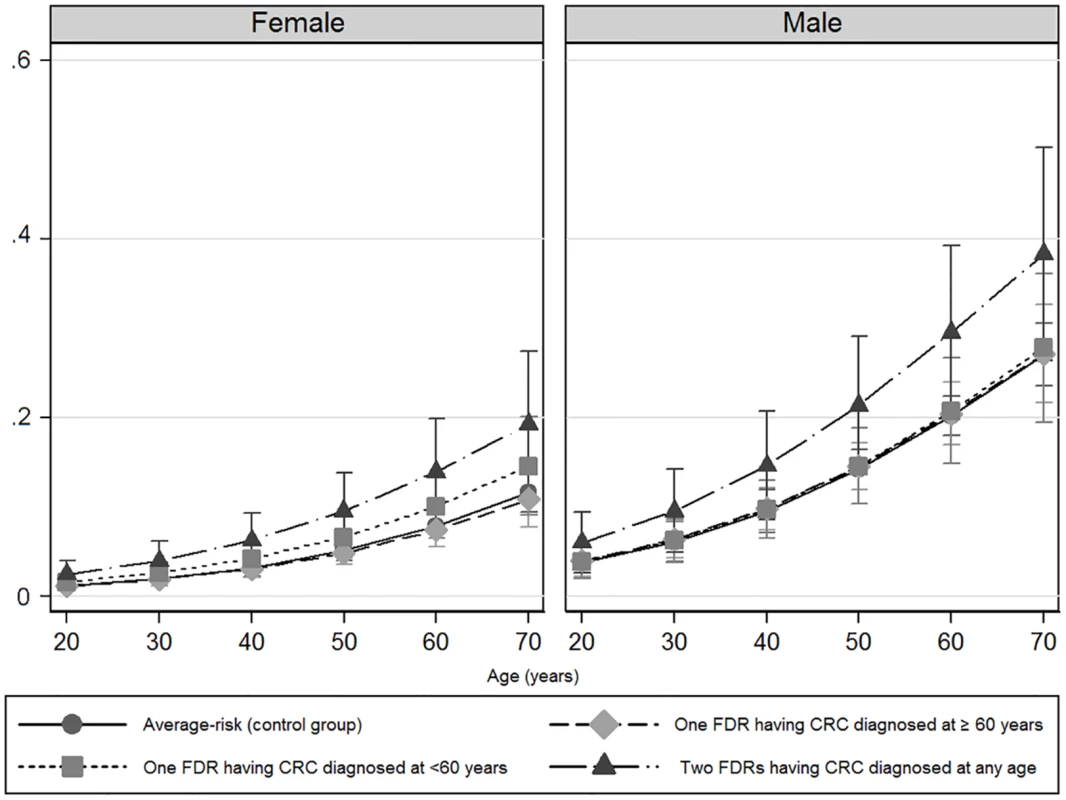
Women (left) and men (right). A logistic regression model was developed to predict the probability of advanced neoplasia according to age. The observed frequencies and probabilities predicted from the regression equation were graphically represented, stratifying individuals in 10-y subsets (from 20 to 70 y for the affected familial groups, and from 50 to 70 y for the control group). Vertical lines mark the corresponding 95% confidence intervals. The prevalence and risk of advanced neoplasia was markedly greater in men than in women in all groups (Table 3) and at each age interval (Fig 2).
Tab. 3. Risk of colorectal neoplasia according to gender in familial- and average-risk groups. 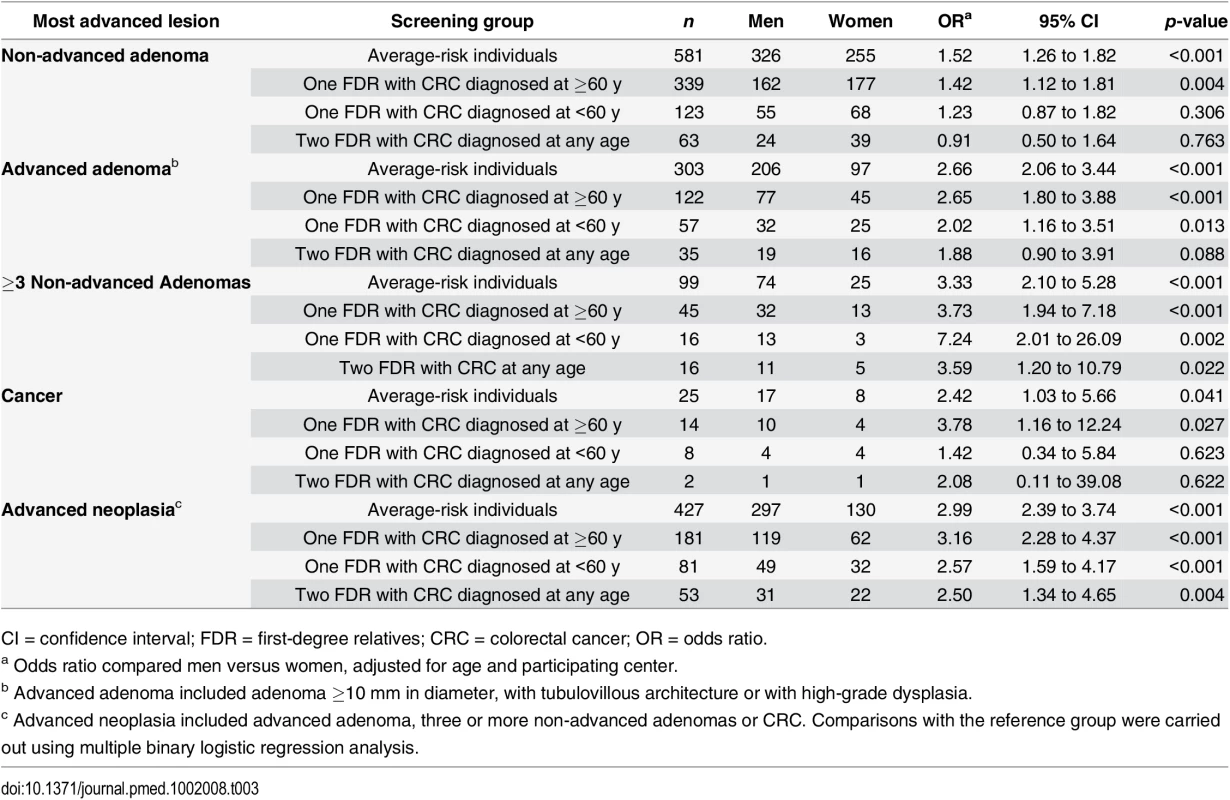
CI = confidence interval; FDR = first-degree relatives; CRC = colorectal cancer; OR = odds ratio. Men and women having one FDR with colorectal cancer consistently showed a similar probability of advanced neoplasia than average-risk men and women, respectively (Fig 2). Interestingly, after the age of 50 y, advanced neoplasia was over 2-fold higher (OR 2.50; 95% CI 1.36–2.66, p < 0.001) and developed at least 10 y earlier in men compared to women in the subgroup of individuals having two FDR with colorectal cancer, (Fig 2 and Table 4).
Tab. 4. Advanced adenomasa and advanced neoplasiab found at colonoscopy according to age and gender. 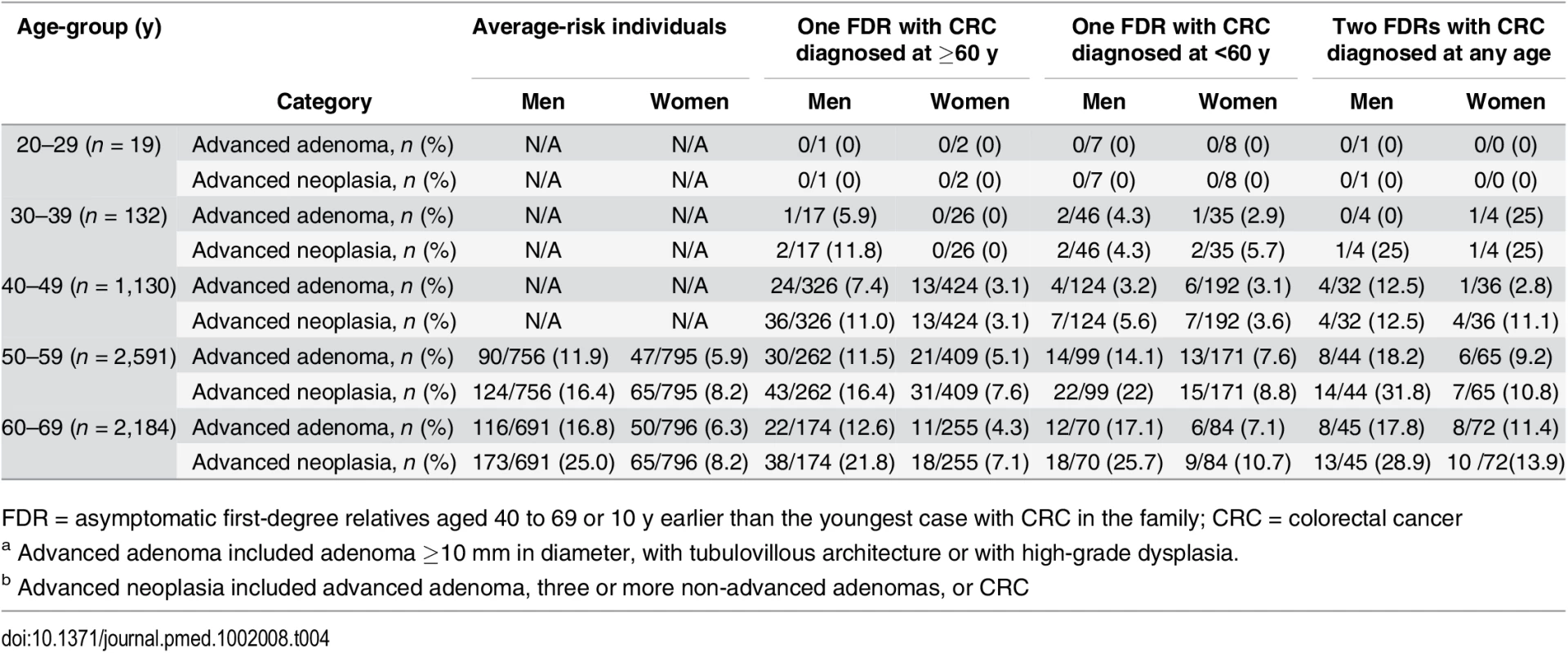
FDR = asymptomatic first-degree relatives aged 40 to 69 or 10 y earlier than the youngest case with CRC in the family; CRC = colorectal cancer Individuals having two FDR with colorectal cancer showed a significantly higher prevalence and risk of advanced adenomas than average-risk individuals both in distal (OR 2.08; 95% CI 1.35 to 3.19, p = 0.001) and proximal colon (OR 1.92; 95% CI 1.08 to 3.40, p = 0.026) (Table 5).
Tab. 5. Risk of advanced adenomaa stratified by location of lesionb. 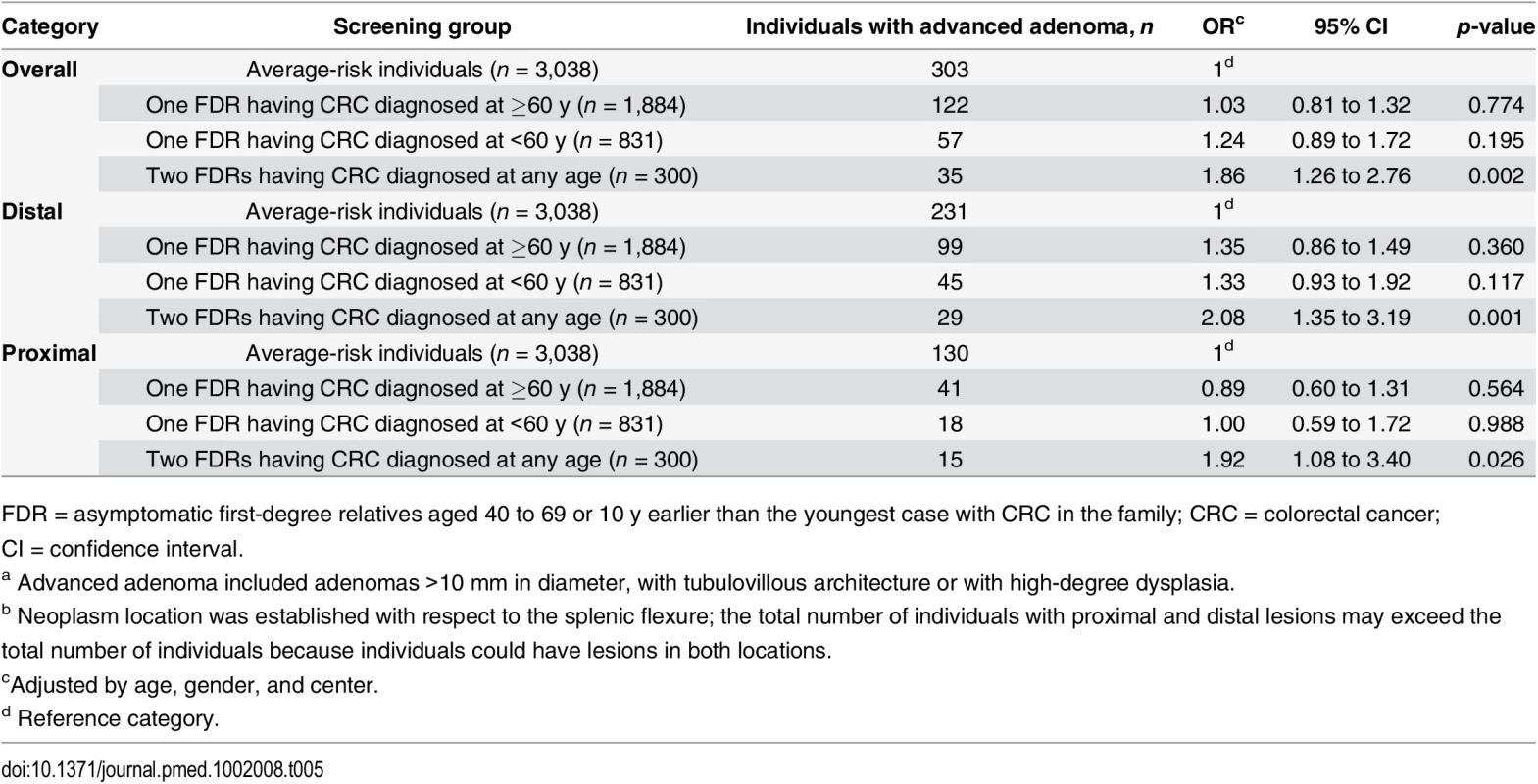
FDR = asymptomatic first-degree relatives aged 40 to 69 or 10 y earlier than the youngest case with CRC in the family; CRC = colorectal cancer; CI = confidence interval. No differences in the prevalence and risk of advanced adenomas categorized by location were observed for individuals having only one FDR with colorectal cancer compared to average-risk individuals.
Analysis of Resources
The number of colonoscopies needed to detect one advanced neoplasia was 7 (95% CI 5.4 to 10) in individuals having two FDR with colorectal cancer, 10.6 (95% CI 9.2 to 12.6) in individuals having only one FDR with colorectal cancer diagnosed at <60 y, 9 (95% CI 7.2 to 11.6) in individuals having one FDR diagnosed at >=60 y, and 11.6 (95% CI 10.4 to 13) in average-risk individuals. Approximately 2-fold fewer colonoscopies were needed to detect one advanced neoplasia in men than in women in all groups (Table 6).
Tab. 6. Number of colonoscopies needed to detect one advanced adenomaa and one advanced neoplasiab according to familial risk. 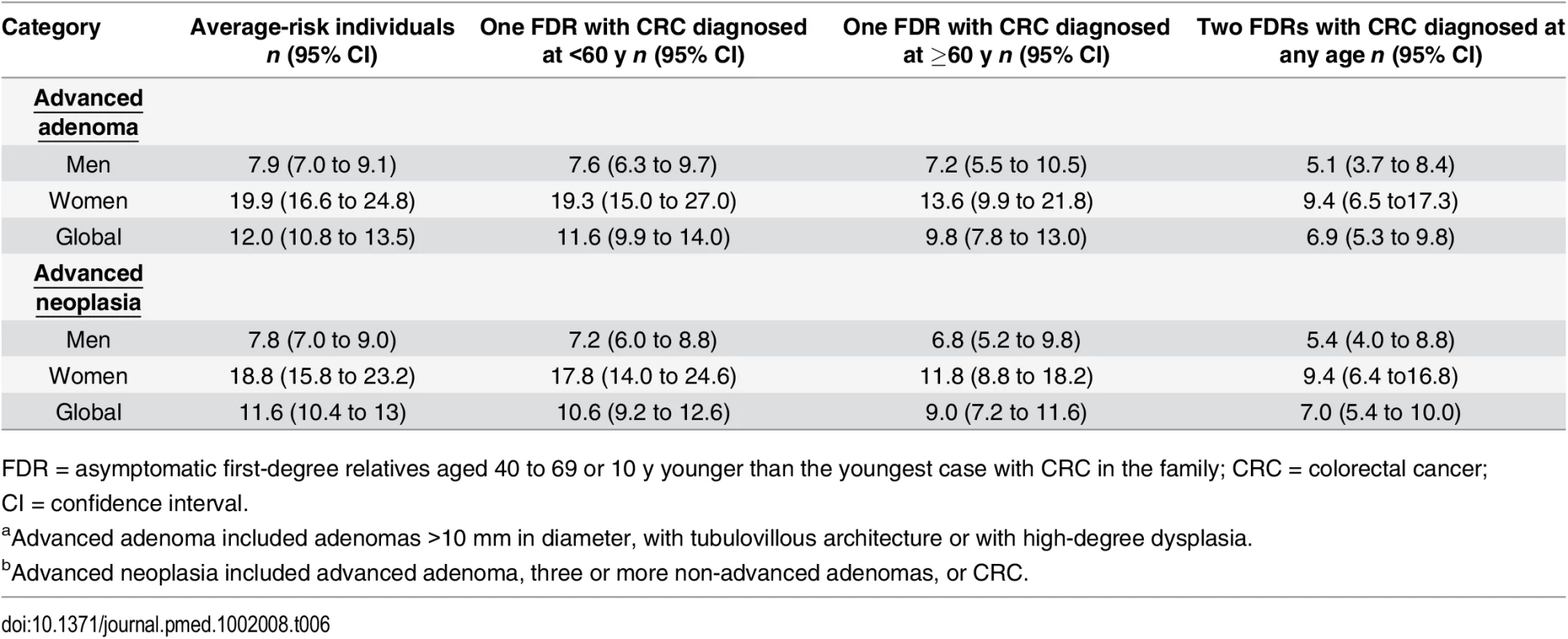
FDR = asymptomatic first-degree relatives aged 40 to 69 or 10 y younger than the youngest case with CRC in the family; CRC = colorectal cancer; CI = confidence interval. Discussion
The current study adds new insight on the risk and probability of colorectal neoplasia in the familial-risk population. Men and women having two FDR with colorectal cancer showed a significantly greater risk of advanced adenomas and advanced neoplasia than those with average-risk. In contrast, men and women having one FDR with colorectal cancer showed a similar risk of these lesions to average-risk individuals, regardless of index-case age at diagnosis of colorectal cancer. The risk of advanced neoplasia was almost 3-fold higher and appeared at least 10 y earlier in men than in women in both the familial - and average-risk groups.
Advanced adenoma and early colorectal cancer are surrogate endpoints of colorectal cancer screening because the detection and treatment of these lesions is associated with a significant reduction of colorectal cancer incidence and mortality [25,26]. Based on this principle, and on population-based studies reporting that FDR of patients with colorectal cancer have a higher relative risk of developing the disease than the average-risk population [12,27,28], current guidelines recommend screening colonoscopy every 5 [3,4,6–8,29] or 10 [3] y, depending on whether colorectal cancer in the index-case is diagnosed at <60 or ≥60 y. However, there is no clear evidence that the natural history of pre-cancerous lesions and cancer differs between familial - and average-risk populations. In this regard, the risk of adenoma recurrence is more related to the characteristics of the neoplasia at baseline colonoscopy and to demographic data (age and gender) than to family history, suggesting that screening intervals in individuals with familial colorectal cancer could be extended beyond 5 y, as most guidelines recommend [30,31].
Previous case-control studies have shown contradictory results with respect to adenoma prevalence in familial colorectal cancer. At least two studies [32,33] have reported a similar prevalence of adenomas in individuals having one FDR with colorectal cancer compared with average-risk individuals. In contrast, other studies have shown an increased prevalence of advanced adenomas in FDR of patients with colorectal cancer compared with average-risk individuals [11–13,34,35]. However, some of these studies had important methodological flaws: first, the control group was small in most of them [13,32,33,35] and frequently inappropriate, including autopsies [10], symptomatic patients [35], or volunteers paying for screening colonoscopy [13]. Second, the small sample size did not allow stratification according to the number of FDR affected [13,32,33,35]. Third, their retrospective design did not allow for ascertaining the indication for colonoscopy and exclusion of patients with hereditary colorectal cancer syndromes [11,12]. Finally, high-quality colonoscopy was not specifically assessed in many studies [10–12,32,33].
Our study apparently solved these drawbacks, corroborating that among relatives of patients with colorectal cancer, only those with two FDR affected showed a marked increase in the prevalence and risk of both adenomas and cancer compared with average-risk individuals in both the proximal and the distal colon. However, the risk of advanced neoplasia in individuals having only one FDR with colorectal cancer diagnosed before or after the age of 60 y was similar to that of average-risk individuals. These findings are in line with the results of a nested study performed within the randomized controlled Prostate, Lung, Colorectal, and Ovarian (PLCO) cancer-screening trial of flexible sigmoidoscopy versus usual care, showing that men and women having two FDR with colorectal cancer had a 2-fold increased risk of incident colorectal cancer, whereas those with one FDR affected were not associated with an increased risk in colorectal cancer incidence or mortality, regardless of their age at the time of diagnosis [36].
Interestingly, we observed that individuals with two FDR had also an increased risk of both non-advanced and advanced-adenomas compared with average-risk individuals. In addition, men showed an almost 3-fold higher risk of advanced neoplasia than women in all groups, and advanced neoplasia appeared at least 10 y earlier in men than in women. It is interesting to note that the number of colonoscopies needed to detect one advanced neoplasia was 2-fold higher in women than in men at all ages in both cohorts. Taken together, our results support the notion that screening colonoscopy may be delayed at least 10 y in women having one or even two FDR with colorectal cancer, as has been shown for women in the average-risk population [37–39].
Our study has several strengths. First, participants were recruited with strict selection criteria regarding colonoscopy quality and inclusion age. Only individuals with a first lifetime complete colonoscopy were eligible and an upper age limit of 69 y was established to minimize the effect of age on the risk of advanced neoplasia. Second, colonoscopies were performed by the same endoscopists who applied the same quality criteria in both cohorts. Third, to our knowledge, this is the largest study to compare the risk of advanced neoplasia between asymptomatic FDR of patients with colorectal cancer and average-risk individuals, thus allowing an accurate estimation of this parameter according to the number of close relatives with colorectal cancer, gender, and index-case age at diagnosis of the disease.
The study also has certain limitations. First, although average-risk individuals were consecutively included in the COLONPREV study [20] this was not the case in the familial-risk group. Fifty-five percent of the FDR were consecutively included in two prospective studies [18,19], whereas the remaining FDR were not. However, demographic characteristics and risk of advanced neoplasia were similar in FDR regardless of whether they were included consecutively or not (S1 Table). This potential bias was also minimized by performing a logistic regression analysis, controlling for confounding factors. Second, due to the study design, there was a substantial difference in age between individuals with family history of colorectal cancer and average risk individuals. Both logistic regression analyses and stratification by age suggested that age was not a main confounding factor in this study (S2 Table). However we cannot exclude an immortal time bias in the study. The risk of bias is built into the study design in a way that cannot be compensated by including an interaction test for age. Third, other confounding factors such as nonsteroidal anti-inflammatory drugs, acetylsalicylic acid, smoking, obesity, and diet were not recorded, and they could conceivably have influenced the results. Fourth, unfortunately, the prevalence of serrated polyps was not recorded because at the time the present study was initiated, classification of serrated polyps was still under debate. In fact, only recently clinical guidelines recommend colonoscopy surveillance for individuals with serrated polyps [1]. Fifth, colorectal cancer family history was obtained by interview and therefore could be underreported [40]. Additionally, the effect of the number of colorectal cancers in relation to the family size could not be analyzed. Sixth, although adenoma detection rate is widely used as a colonoscopy quality indicator, unfortunately this information was not available. Since the centers that provided individuals in the average-risk group (COLONPREV) and the ones that recruited individuals in the familial-risk cohort are virtually the same, we do not expect differences in colonoscopy quality between the two cohorts. Finally, some of the individuals included may have been members of families with Lynch syndrome, since we did not systematically exclude DNA mismatch repair deficiency in all cases with colorectal cancer. However, the overall results should not be greatly affected since this possibility would only involve a small number of individuals.
Our findings suggest that screening guidelines for the management of familial colorectal cancer, if not adjusted for the number of relatives affected and sex, may substantially overestimate the prevalence of advanced neoplasia, particularly in men and women having one FDR with colorectal cancer and in women having two FDR with colorectal cancer diagnosed before the age of 50. In fact, the same screening strategy as that for average-risk individuals could be recommended to men and women having only one FDR with colorectal cancer, but starting at the age of 40 or 45 y, in line with the results of previous studies [5,30], thus avoiding overuse of screening colonoscopy. In support of this recommendation, there are two recent prospective studies demonstrating that fecal immunochemical testing is as effective as colonoscopy to detect advanced neoplasia in colorectal cancer associated with familial risk [19,41]. In contrast, the higher prevalence and earlier presentation of advanced neoplasia in men having two FDR with colorectal cancer suggest that men have higher genetic penetrance, thus supporting screening colonoscopy from the age of 40, whereas it could be delayed until the age of 50 or older in women with two FDR.
In conclusion, our study demonstrates that the risk and predicted probability of advanced neoplasia are markedly increased in individuals having two FDR with colorectal cancer compared to average-risk individuals, but not in individuals having only one FDR with colorectal cancer, regardless of when they were diagnosed. Our results suggest that men having two FDR with colorectal cancer may benefit from an early screening colonoscopy (i.e., 40 y old or 10 y before the youngest case in the immediate family), whereas individuals having only one FDR with colorectal cancer could be recommended to undergo the same screening strategy as the average-risk population.
Supporting Information
Zdroje
1. Lieberman DA, Rex DK, Winawer SJ, Giardiello FM, Johnson DA, Levin TR. Guidelines for colonoscopy surveillance after screening and polypectomy: a consensus update by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2012;143(3):844–57. Epub 2012/07/06. doi: 10.1053/j.gastro.2012.06.001 22763141.
2. Rex DK, Johnson DA, Anderson JC, Schoenfeld PS, Burke CA, Inadomi JM. American College of Gastroenterology guidelines for colorectal cancer screening 2009 [corrected]. The American journal of gastroenterology. 2009;104(3):739–50. Epub 2009/02/26. doi: 10.1038/ajg.2009.104 19240699.
3. Davila RE, Rajan E, Baron TH, Adler DG, Egan JV, Faigel DO, et al. ASGE guideline: colorectal cancer screening and surveillance. Gastrointestinal endoscopy. 2006;63(4):546–57. Epub 2006/03/28. doi: 10.1016/j.gie.2006.02.002 16564851.
4. Cairns SR, Scholefield JH, Steele RJ, Dunlop MG, Thomas HJ, Evans GD, et al. Guidelines for colorectal cancer screening and surveillance in moderate and high risk groups (update from 2002). Gut. 2010;59(5):666–89. Epub 2010/04/30. doi: 10.1136/gut.2009.179804 20427401.
5. Fuchs CS, Giovannucci EL, Colditz GA, Hunter DJ, Speizer FE, Willett WC. A prospective study of family history and the risk of colorectal cancer. N Engl J Med. 1994;331(25):1669–74. Epub 1994/12/22. doi: 10.1056/NEJM199412223312501 7969357.
6. Castells A, Marzo-Castillejo M, Mascort JJ, Amador FJ, Andreu M, Bellas B, et al. [Clinical practice guideline. Prevention of colorectal cancer. 2009 update. Asociacion Espanola de Gastroenterologia]. Gastroenterologia y hepatologia. 2009;32(10):717 e1–58. Epub 2010/05/18. 20474100.
7. Levin B, Lieberman DA, McFarland B, Andrews KS, Brooks D, Bond J, et al. Screening and surveillance for the early detection of colorectal cancer and adenomatous polyps, 2008: a joint guideline from the American Cancer Society, the US Multi-Society Task Force on Colorectal Cancer, and the American College of Radiology. Gastroenterology. 2008;134(5):1570–95. 18384785. doi: 10.1053/j.gastro.2008.02.002
8. Smith RA, Brooks D, Cokkinides V, Saslow D, Brawley OW. Cancer screening in the United States, 2013: a review of current American Cancer Society guidelines, current issues in cancer screening, and new guidance on cervical cancer screening and lung cancer screening. CA Cancer J Clin. 2013;63(2):88–105. Epub 2013/02/05. doi: 10.3322/caac.21174 23378235.
9. Rustgi AK. The genetics of hereditary colon cancer. Genes Dev. 2007;21(20):2525–38. Epub 2007/10/17. doi: 10.1101/gad.1593107 17938238.
10. Lindgren G, Liljegren A, Jaramillo E, Rubio C, Lindblom A. Adenoma prevalence and cancer risk in familial non-polyposis colorectal cancer. Gut. 2002;50(2):228–34. Epub 2002/01/15. 11788565; PubMed Central PMCID: PMC1773092.
11. Tuohy TM, Rowe KG, Mineau GP, Pimentel R, Burt RW, Samadder NJ. Risk of colorectal cancer and adenomas in the families of patients with adenomas: a population-based study in Utah. Cancer. 2014;120(1):35–42. doi: 10.1002/cncr.28227 24150925.
12. Samadder NJ, Curtin K, Tuohy TM, Rowe KG, Mineau GP, Smith KR, et al. Increased risk of colorectal neoplasia among family members of patients with colorectal cancer: a population-based study in Utah. Gastroenterology. 2014;147(4):814–21 e5; quiz e15-6. doi: 10.1053/j.gastro.2014.07.006 25042087.
13. Armelao F, Paternolli C, Franceschini G, Franch R, Orlandi PG, Miori G, et al. Colonoscopic findings in first-degree relatives of patients with colorectal cancer: a population-based screening program. Gastrointest Endosc. 2011;73(3):527–34 e2. doi: 10.1016/j.gie.2010.12.025 21353850.
14. Bradshaw N, Holloway S, Penman I, Dunlop MG, Porteous ME. Colonoscopy surveillance of individuals at risk of familial colorectal cancer. Gut. 2003;52(12):1748–51. Epub 2003/11/25. 14633955; PubMed Central PMCID: PMC1773898.
15. Dowling DJ, John DJ St, Macrae FA, Hopper JL. Yield from colonoscopic screening in people with a strong family history of common colorectal cancer. J Gastroenterol Hepatol. 2000;15(8):939–44. Epub 2000/10/07. 11022837.
16. Neklason DW, Thorpe BL, Ferrandez A, Tumbapura A, Boucher K, Garibotti G, et al. Colonic adenoma risk in familial colorectal cancer—a study of six extended kindreds. Am J Gastroenterol. 2008;103(10):2577–84. Epub 2008/08/02. doi: 10.1111/j.1572-0241.2008.02019.x 18671820; PubMed Central PMCID: PMC2922112.
17. van der Meulen-de Jong AE, Morreau H, Becx MC, Crobach LF, van Haastert M, ten Hove WR, et al. High detection rate of adenomas in familial colorectal cancer. Gut. 2011;60(1):73–6. 20833659. doi: 10.1136/gut.2010.217091
18. Castro I, Cubiella J, Rivera C, Gonzalez-Mao C, Vega P, Soto S, et al. Fecal immunochemical test accuracy in familial risk colorectal cancer screening. Int J Cancer. 2014;134(2):367–75. doi: 10.1002/ijc.28353 23818169.
19. Quintero E, Carrillo M, Gimeno-Garcia AZ, Hernandez-Guerra M, Nicolas-Perez D, Alonso-Abreu I, et al. Equivalency of fecal immunochemical tests and colonoscopy in familial colorectal cancer screening. Gastroenterology. 2014;147(5):1021–30 e1; quiz e16-7. doi: 10.1053/j.gastro.2014.08.004 25127679.
20. Quintero E, Castells A, Bujanda L, Cubiella J, Salas D, Lanas A, et al. Colonoscopy versus fecal immunochemical testing in colorectal-cancer screening. N Engl J Med. 2012;366(8):697–706. 22356323. doi: 10.1056/NEJMoa1108895
21. Jover R, Herraiz M, Alarcon O, Brullet E, Bujanda L, Bustamante M, et al. Clinical practice guidelines: quality of colonoscopy in colorectal cancer screening. Endoscopy. 2012;44(4):444–51. doi: 10.1055/s-0032-1306690 22438159.
22. Parra-Blanco A, Nicolas-Perez D, Gimeno-Garcia A, Grosso B, Jimenez A, Ortega J, et al. The timing of bowel preparation before colonoscopy determines the quality of cleansing, and is a significant factor contributing to the detection of flat lesions: a randomized study. World J Gastroenterol. 2006;12(38):6161–6. 17036388.
23. Small KA H C. Multinomial Logit Specification Tests. Int Econ Rev. 1985;26 : 619–27.
24. Austin PC. Absolute risk reductions and numbers needed to treat can be obtained from adjusted survival models for time-to-event outcomes. J Clin Epidemiol. 2010;63(1):46–55. doi: 10.1016/j.jclinepi.2009.03.012 19595575.
25. Winawer SJ, Zauber AG, Gerdes H, O'Brien MJ, Gottlieb LS, Sternberg SS, et al. Risk of colorectal cancer in the families of patients with adenomatous polyps. National Polyp Study Workgroup. N Engl J Med. 1996;334(2):82–7. doi: 10.1056/NEJM199601113340204 8531963.
26. Zauber AG, Winawer SJ, O'Brien MJ, Lansdorp-Vogelaar I, van Ballegooijen M, Hankey BF, et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med. 2012;366(8):687–96. doi: 10.1056/NEJMoa1100370 22356322; PubMed Central PMCID: PMC3322371.
27. Butterworth AS H J, Pharoah P. Relative and absolute risk of colorectal cancer for individuals with a family history:a mate-analysis. Eur J Cancer. 2006;42(216–227).
28. John DJ St, McDermott FT, Hopper JL, Debney EA, Johnson WR, Hughes ES. Cancer risk in relatives of patients with common colorectal cancer. Annals of internal medicine. 1993;118(10):785–90. Epub 1993/05/15. 8470852.
29. Desch CE, Benson AB 3rd, Somerfield MR, Flynn PJ, Krause C, Loprinzi CL, et al. Colorectal cancer surveillance: 2005 update of an American Society of Clinical Oncology practice guideline. J Clin Oncol. 2005;23(33):8512–9. Epub 2005/11/02. doi: 10.1200/JCO.2005.04.0063 16260687.
30. Dove-Edwin I S P, Adams J, Thomas HJW. Prevention of colorectal cancer by colonoscopic surveillance in individuals with a family history of colorectal cancer:16 year prospective, follow-up study. Br Med J. 2005;331 : 1047–52.
31. Martinez ME, Baron JA, Lieberman DA, Schatzkin A, Lanza E, Winawer SJ, et al. A pooled analysis of advanced colorectal neoplasia diagnoses after colonoscopic polypectomy. Gastroenterology. 2009;136(3):832–41. Epub 2009/01/28. doi: 10.1053/j.gastro.2008.12.007 19171141; PubMed Central PMCID: PMC3685417.
32. Aitken JF, Bain CJ, Ward M, Siskind V, MacLennan R. Risk of colorectal adenomas in patients with a family history of colorectal cancer: some implications for screening programmes. Gut. 1996;39(1):105–8. 8881819; PubMed Central PMCID: PMC1383241.
33. Guillem JG, Forde KA, Treat MR, Neugut AI, O'Toole KM, Diamond BE. Colonoscopic screening for neoplasms in asymptomatic first-degree relatives of colon cancer patients. A controlled, prospective study. Dis Colon Rectum. 1992;35(6):523–9. 1587168.
34. Ng SC, Lau JY, Chan FK, Suen BY, Leung WK, Tse YK, et al. Increased risk of advanced neoplasms among asymptomatic siblings of patients with colorectal cancer. Gastroenterology. 2013;144(3):544–50. Epub 2012/11/20. doi: 10.1053/j.gastro.2012.11.011 23159367.
35. Pariente A, Milan C, Lafon J, Faivre J. Colonoscopic screening in first-degree relatives of patients with 'sporadic' colorectal cancer: a case-control study. The Association Nationale des Gastroenterologues des Hopitaux and Registre Bourguignon des Cancers Digestifs (INSERM CRI 9505). Gastroenterology. 1998;115(1):7–12. 9649451.
36. Schoen RE, Razzak A, Yu KJ, Berndt SI, Firl K, Riley TL, et al. Incidence and Mortality of Colorectal Cancer in Individuals With a Family History of Colorectal Cancer. Gastroenterology. 2015;149(6):1438–45 e1. doi: 10.1053/j.gastro.2015.07.055 26255045; PubMed Central PMCID: PMCPMC4628587.
37. Ferlitsch M, Reinhart K, Pramhas S, Wiener C, Gal O, Bannert C, et al. Sex-specific prevalence of adenomas, advanced adenomas, and colorectal cancer in individuals undergoing screening colonoscopy. JAMA. 2011;306(12):1352–8. doi: 10.1001/jama.2011.1362 21954479.
38. Kolligs FT, Crispin A, Munte A, Wagner A, Mansmann U, Goke B. Risk of advanced colorectal neoplasia according to age and gender. PLoS ONE. 2011;6(5):e20076. doi: 10.1371/journal.pone.0020076 21629650; PubMed Central PMCID: PMC3101231.
39. Regula J, Rupinski M, Kraszewska E, Polkowski M, Pachlewski J, Orlowska J, et al. Colonoscopy in colorectal-cancer screening for detection of advanced neoplasia. N Engl J Med. 2006;355(18):1863–72. 17079760.
40. Mitchell RJ, Brewster D, Campbell H, Porteous ME, Wyllie AH, Bird CC, et al. Accuracy of reporting of family history of colorectal cancer. Gut. 2004;53(2):291–5. 14724166; PubMed Central PMCID: PMCPMC1774933.
41. Cubiella J, Castro I, Hernandez V, Gonzalez-Mao C, Rivera C, Iglesias F, et al. Diagnostic accuracy of fecal immunochemical test in average - and familial-risk colorectal cancer screening. United European Gastroenterol J. 2014;2(6):522–9. doi: 10.1177/2050640614553285 25452848; PubMed Central PMCID: PMC4245302.
Štítky
Interní lékařství
Článek vyšel v časopisePLOS Medicine
Nejčtenější tento týden
2016 Číslo 5- Berberin: přírodní hypolipidemikum se slibnými výsledky
- Červená fermentovaná rýže účinně snižuje hladinu LDL cholesterolu jako vhodná alternativa ke statinové terapii
- Příznivý vliv Armolipidu Plus na hladinu cholesterolu a zánětlivé parametry u pacientů s chronickým subklinickým zánětem
- Alternativní léčebné možnosti u hypercholesterolemie při intoleranci statinů
- Vliv kombinace nutraceutik na remodelaci levé komory srdeční u osob s metabolickým syndromem
-
Všechny články tohoto čísla
- Epidemiology and Reporting Characteristics of Systematic Reviews of Biomedical Research: A Cross-Sectional Study
- Steroid-Based Therapy and Risk of Infectious Complications
- How Much Can the USA Reduce Health Care Costs by Reducing Smoking?
- Interpreting the Global Enteric Multicenter Study (GEMS) Findings on Sanitation, Hygiene, and Diarrhea
- Health Research and the World Humanitarian Summit—Not a Thousand Miles Apart
- A Public Health Paradox: The Women Most Vulnerable to Malaria Are the Least Protected
- Toward a Common Secure Future: Four Global Commissions in the Wake of Ebola
- The Clinical Challenge of Sepsis Identification and Monitoring
- All-Cause Mortality of Low Birthweight Infants in Infancy, Childhood, and Adolescence: Population Study of England and Wales
- Smoking Behavior and Healthcare Expenditure in the United States, 1992–2009: Panel Data Estimates
- Estimating the Risk of Chronic Pain: Development and Validation of a Prognostic Model (PICKUP) for Patients with Acute Low Back Pain
- Initiating Antiretroviral Therapy for HIV at a Patient’s First Clinic Visit: The RapIT Randomized Controlled Trial
- Prioritizing Surgical Care on National Health Agendas: A Qualitative Case Study of Papua New Guinea, Uganda, and Sierra Leone
- Effectiveness of and Financial Returns to Voluntary Medical Male Circumcision for HIV Prevention in South Africa: An Incremental Cost-Effectiveness Analysis
- Risk of Advanced Neoplasia in First-Degree Relatives with Colorectal Cancer: A Large Multicenter Cross-Sectional Study
- Common Infections in Patients Prescribed Systemic Glucocorticoids in Primary Care: A Population-Based Cohort Study
- Sanitation and Hygiene-Specific Risk Factors for Moderate-to-Severe Diarrhea in Young Children in the Global Enteric Multicenter Study, 2007–2011: Case-Control Study
- A Revolution in Treatment for Hepatitis C Infection: Mitigating the Budgetary Impact
- Nondisclosure of Financial Interest in Clinical Practice Guideline Development: An Intractable Problem?
- Financial Relationships between Organizations That Produce Clinical Practice Guidelines and the Biomedical Industry: A Cross-Sectional Study
- Prices, Costs, and Affordability of New Medicines for Hepatitis C in 30 Countries: An Economic Analysis
- PLOS Medicine
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Estimating the Risk of Chronic Pain: Development and Validation of a Prognostic Model (PICKUP) for Patients with Acute Low Back Pain
- Prioritizing Surgical Care on National Health Agendas: A Qualitative Case Study of Papua New Guinea, Uganda, and Sierra Leone
- A Revolution in Treatment for Hepatitis C Infection: Mitigating the Budgetary Impact
- Toward a Common Secure Future: Four Global Commissions in the Wake of Ebola
Přihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



