-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaShifts in the Antibiotic Susceptibility, Serogroups, and Clonal Complexes of in Shanghai, China: A Time Trend Analysis of the Pre-Quinolone and Quinolone Eras
Minggui Wang and colleagues assess the genetic origin and changing prevalence of N. meningitidis resistance to ciprofloxacin.
Published in the journal: . PLoS Med 12(6): e32767. doi:10.1371/journal.pmed.1001838
Category: Research Article
doi: https://doi.org/10.1371/journal.pmed.1001838Summary
Minggui Wang and colleagues assess the genetic origin and changing prevalence of N. meningitidis resistance to ciprofloxacin.
Introduction
Neisseria meningitidis is a leading cause of bacterial meningitis worldwide. At least 1.2 million cases of meningococcal disease are estimated to occur in the world annually, with approximately 10% resulting in death [1]. Based on variants in the capsular polysaccharide, 12 serogroups have been described, with the majority of invasive meningococcal disease caused by A, B, C, X, Y, or W [1]. The use of multi-locus sequence typing (MLST)—where seven housekeeping genes are sequenced to establish sequence types and clonal complexes (CCs)—has contributed substantially to our understanding of the molecular epidemiology of N. meningitidis, including isolates from different serogroups but with similar antigenic or disease phenotypes [2]. Ciprofloxacin is one of the three antibiotics recommended for chemoprophylaxis worldwide [3]. A number of ciprofloxacin-resistant meningococcal isolates have emerged in some countries since 1992 [4–8], although they remain rare in other countries. Resistance is mostly due to point mutations in the quinolone-resistance-determining region (QRDR) of the gyrA gene, which codes for subunit A of DNA gyrase [9], with a minor proportion attributed to efflux pump mechanisms [10,11].
In China, meningococcal disease is notifiable, with specimens and isolates sent to the local Center for Disease Control and Prevention (CDC). Since 1950, the Shanghai CDC has acquired data on the incidence and case fatality rates for meningococcal disease and has collected bacterial isolates from patients and their close contacts when available. Meningococcal disease was highly prevalent in China before the 1990s [12], and Shanghai was one of the high incidence zones in the country, with a peak incidence of 434 cases per 100,000 population in 1967 [13]. From 2003 to 2005, an unusual increase of serogroup C meningococcal cases was reported in Anhui Province, China, including 264 cases with 18 deaths. The hypervirulent lineage ST-4821 complex (CC4821) was first identified during this period and appeared to be responsible for ten of the serogroup C outbreaks, accounting for 72 cases [14]. In response, serogroup A and C polysaccharide vaccine has been used in China for routine immunization since 2006 [15].
Fluoroquinolones, including ciprofloxacin and norfloxacin, have been recommended for meningococcal chemoprophylaxis for adult close contacts of cases in China since 2005, with an oral dose of 100 mg every 8 h for 2 d. However, fluoroquinolones have been widely used for the treatment of other infectious conditions including infectious diarrhea, urinary tract infection, and respiratory tract infection since 1986 [16–21]. Between 2001 and 2010, fluoroquinolone consumption increased by approximately 2 × 108 standard units and has become the third most consumed antibiotic in China [22]. Ciprofloxacin resistance was found to be highly prevalent (71%, 12/17) in serogroup W N. meningitidis isolates in Anhui Province during 2012–2013 [23]. However, data are scarce regarding the changes in serogroups, CCs, and antimicrobial susceptibility in N. meningitidis between the periods before and after the introduction of fluoroquinolones (the pre-quinolone and quinolone eras).
The goal of this study was to investigate the shifts in antimicrobial susceptibility, serogroups, and CCs of N. meningitidis associated with the introduction and expanding use of fluoroquinolones in Shanghai, using isolates from cerebrospinal fluid (CSF) and/or blood of invasive meningococcal disease cases that occurred in Shanghai in the pre-quinolone and quinolone eras and that were archived by the Shanghai CDC. Carriage isolates from pharyngeal carriage surveys conducted by the Shanghai CDC in both eras were also included to assess circulation of ciprofloxacin-nonsusceptible N. meningitidis in the healthy population.
Methods
Ethical Aspects
All specimens from meningococcal cases and close contacts were collected as part of the routine clinical management of patients, according to the national guidelines in China. Consequently, informed consent was not sought, and the study was approved by the Institutional Review Board of Huashan Hospital, Fudan University.
Study Settings
The study was conducted in Shanghai, a municipality with province-level status and also the economic and financial center of China. It lies on China’s central eastern coast at the mouth of the Yangtze River, with over 20 million permanent residents residing in an area of 6,340 km2. With four distinct seasons, Shanghai is characterized as having a subtropical monsoon climate with an annual average temperature of 16°C. Since 1950, the Shanghai CDC has continuously acquired data on the incidence of and case fatality rates from meningococcal disease. N. meningitidis isolates during the periods of 1965–1985 and 2005–2013 were collected, with an interruption during 1986–2004. Because fluoroquinolones were introduced into clinical practice in 1986 locally [22,24], we characterized the meningococcal disease isolates from 1965–1985 and 2005–2013, corresponding to the pre-quinolone and quinolone eras, respectively.
Meningococcal Surveillance in Shanghai
According to the National Notifiable Disease Surveillance System in China, a patient with a sudden onset of headache, fever, nausea, vomiting, petechial rash, or stiff neck, accompanied by delirium, coma or shock, or Gram-negative diplococci identified in the CSF defines a suspected case of meningococcal disease, which should be reported to the local CDC within 6 h. Specimens of all kinds (mainly CSF and blood) are cultured, and PCR is used for meningococcal identification if the cultures are negative. Under this mandate, all of the specimens and N. meningitidis isolate cultures from the CSF and/or blood of suspected cases in Shanghai have been sent to the Shanghai CDC since 2006. Before 2006, suspected cases of meningococcal disease were reported to the local CDC, but it was not mandatory to send the specimens or isolates to the CDC. Therefore, the Shanghai CDC collected and saved isolates only when the treating physicians elected to submit the specimens or isolates. During 1985–2004, a decrease in the incidence of meningococcal disease and the increased ability of hospital laboratories to culture and serogroup N. meningitidis without assistance from the Shanghai CDC resulted in interrupted collection of meningococcal isolates during this period.
For the present study, a confirmed case of meningococcal disease was defined by isolation of N. meningitidis from sterile sites (mainly CSF or blood), including culture-negative cases positive by Gram stain, latex agglutination, or PCR [14]. Close contacts were defined as those with history of working, living, studying, or nursing with a confirmed case, mainly including the household members, classmates, and roommates of the patients. Asymptomatic carriers were defined as healthy individuals with a pharyngeal swab growing N. meningitidis but without an epidemiological link to meningococcal cases.
Carriage Surveys
During 1966–1978, surveys of meningococcal carriage were performed by the Shanghai CDC annually except in 1968 and 1969 (11 surveys total). Each pharyngeal swab was taken by swabbing the posterior pharyngeal wall behind the uvula through the mouth (posterior pharyngeal swab), using a dry cotton-tipped wooden swab held with a copper rod. Samples were plated directly onto chocolate agar plates supplemented with vancomycin (3.3 μg/ml) and polymyxin B (25 IU/ml), and delivered to the laboratory within 4 h. A total of 23,083 swabs were collected, from toddlers in child care centers, students in schools, and people working in crowded circumstances, such as sales associates in department stores, railway station workers, and uniformed service members in the army. Only some isolates from these surveys were kept in storage and were thus available for this study.
Additionally, surveys of pharyngeal carriage were conducted in September 2007 and September 2010, prior to the epidemic seasons of meningococcal disease (winter and spring). Both of the surveys were performed in three districts of Shanghai to cover 630 people from seven age groups (<1 y, 1–2 y, 3–4 y, 5–6 y, 7–14 y, 15–19 y, and ≥20 y), with 30 individuals sampled per age group per district. The age groups <1 y and 1–2 y comprised infants living at home, who were swabbed when they presented to local community health centers for scheduled immunization. The age groups 3–4 y and 5–6 y were preschool children, mostly in kindergarten. The age groups 7–14 y and 15–19 y were students in different grades, while ≥20 y comprised teachers working in primary school. Each posterior pharyngeal swab was taken with a CLASSIQSwab (Copan), an absorbent cotton-tipped plastic swab, and was processed as described above.
Meningococcal Isolates and Bacterial Identification
The 374 isolates in this study comprised 305 isolates collected in 1965–1985 and 69 collected in 2005–2013. In 1965–1985, there were 126 disease isolates, 15 isolates from close contacts, and 164 carriage isolates. In 2005–2013, there were 37 disease isolates, 17 isolates from close contacts, and 15 carriage isolates. All of the isolates were confirmed as N. meningitidis by Neisseria-Haemophilus (NH) cards of the Vitek 2 automated system (bioMérieux). Serogroups were determined by slide agglutination using monoclonal antiserum to capsular polysaccharides A, B, C, W, X, Y, and Z (BD). Since 2007, PCR targeting ctrA and porA genes has been used for meningococcal identification of culture-negative cases [25,26]. PCR with primers for all 12 serogroups was used for serogrouping of culture-negative specimens and non-groupable strains [25].
Antimicrobial Susceptibility Testing
The minimum inhibitory concentrations (MICs) of 11 antimicrobial agents were determined by the agar dilution method, with susceptibility interpreted according to the 2013 guidelines of the Clinical and Laboratory Standards Institute [27]. Ciprofloxacin-nonsusceptible strains (MIC ≥ 0.12 μg/ml as resistant and MIC = 0.06 μg/ml as intermediate) were investigated for efflux pump activity by determination of ciprofloxacin MIC with the microdilution method in the presence and absence of 20 μg/ml reserpine (Sigma-Aldrich) [10]. Decreased susceptibility to penicillin (MIC ≥ 0.12 μg/ml) was confirmed by Etest (bioMérieux).
Molecular Typing for N. meningitidis
All isolates were characterized by MLST, typing of the variable regions of porin A (porA subtyping), and pulsed-field gel electrophoresis (PFGE) with the restriction endonuclease NheI (TaKaRa), as previously described [14,28]. The sequences of seven housekeeping genes (abcZ, adk, aroE, fumC, gdh, pdhC and pgm) were submitted to the Neisseria PubMLST database for assignment of sequence types (STs) [29]. A CC was defined as a group of related isolates whereby six or seven alleles are shared with at least one other member of the group [30]. In the same CC, single-locus variants (SLVs) represent two STs differing by one locus, and double-locus variants (DLVs) represent two STs differing by two loci. Clustering of PFGE patterns was assigned using the BioNumerics software package (version 6.0; Applied Maths), using the unweighted pair group method and an arithmetic averages (UPGMA) clustering algorithm [31], which was also used to construct dendrograms and minimum spanning trees based on MLST. To facilitate comparison of strains with different PFGE patterns, we used the method recommended by Popovic et al. for the investigation of meningococcal clonal dissemination [31]. With the position tolerance set at 1.5%, strains that had >95%, 85%–95%, and <85% relatedness were assigned to the same PFGE subtype, the same PFGE type, and different PFGE types, respectively, which represented strains that were closely related, possibly related, and genetically unrelated, respectively [31].
Sequencing of Quinolone-Resistance-Associated Genes
Previously described primers and procedures were used to amplify and sequence the QRDRs of the gyrase genes (gyrA and gyrB) and the topoisomerase IV genes (parC and parE) [3,9]. The mtrR gene, a negative transcriptional regulator of the MtrCDE efflux pump, was also sequenced. If any amino acid substitutions or insertions/deletions were found in MtrR, efflux pump involvement was considered [32]. In order to study the polymorphism of the gyrA QRDR, gyrA (115–639) genes differing by at least one nucleotide were assigned a unique gyrA allele [9]. The Lasergene software package (version 7.1; DNASTAR), Clustal X, and MEGA 5 were used to analyze nucleotide sequences and the deduced amino acid sequences [3]. The PFGE data of the 2004 Anhui outbreak isolate 053442 (GenBank accession number CP000381) was provided by PulseNet China [33,34].
Statistical Analyses
The incidence of meningococcal disease was calculated for 1950 to 2013 using population estimates from the Department of Acute Infectious Diseases of the Shanghai CDC. The chi-squared test for trend was used to determine linear trends of incidence over time, while regression splines were used to model nonlinear trends of incidence and case fatality rates during the period. Regression splines were performed using R (version 3.1.2; http://cran.r-project.org/). Incidences, case fatality rates, gender, age, predominant serogroups, predominant CCs, and susceptibility to ciprofloxacin, trimethoprim-sulfamethoxazole, and penicillin were compared between 1965–1985 and 2005–2013 for patients with meningococcal disease, close contacts, and asymptomatic N. meningitidis carriers using the chi-squared test (Fisher’s exact or Pearson’s). The odds ratio and 95% CI were calculated using the software OpenEpi module PersonTime2 (http://www.openepi.com/PersonTime2/PersonTime2.htm) [35]. Statistical analysis was performed using SPSS (version 20.0; IBM), and statistical significance was assessed at p < 0.05.
Results
Incidence and Case Fatality Rates of Meningococcal Disease in Shanghai
During the 64-y period from 1950 to 2013, 127,926 N. meningitidis cases were reported in Shanghai: 16,249 in 1950–1960, 105,671 in 1961–1970, 3,411 in 1971–1980, 1,957 in 1981–1990, 378 in 1991–2000, and 260 in 2001–2013, with the average incidences of 21.4, 97.6, 3.1, 1.6, 0.2, and 0.1 cases per 100,000 population for each period, respectively. The annual incidence peaked at 434 cases per 100,000 population in 1967 (Fig 1). The annual incidence for 1950–2013 was analyzed by regression spline (Table 1). The annual incidence fluctuated with time (Fig 1), with two waves observed in the 1950s–1980s and the 1980s–2000s. During 1965–1985, the average incidence was 43 cases per 100,000 population, compared with 0.08 during 2005–2013 (p < 0.001; Table 2).
Fig. 1. Annual incidence of reported cases of meningococcal disease in Shanghai from 1950 to 2013. 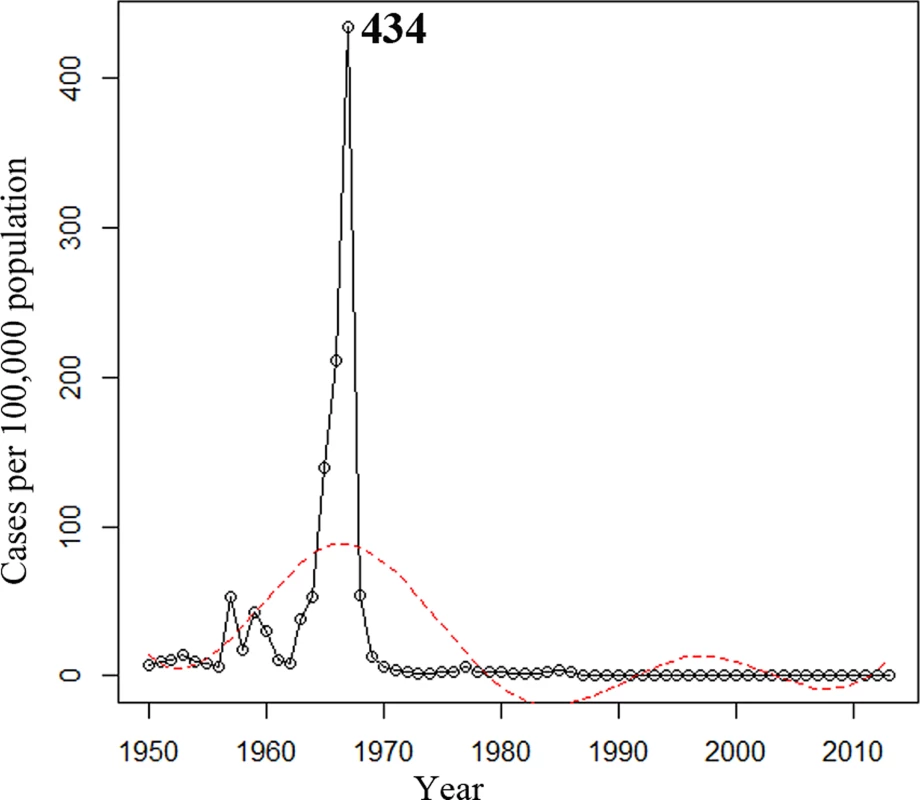
The trend was analyzed using regression splines (red dotted line). Tab. 1. Regression splines of annual incidence and case fatality rate of meningococcal disease for 1950–2013 in Shanghai. 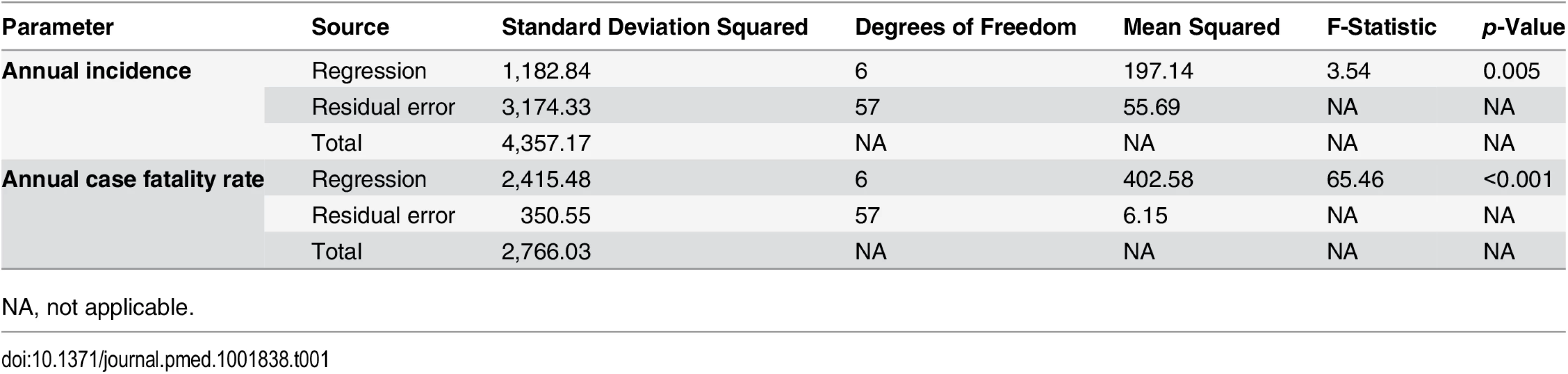
NA, not applicable. Tab. 2. Epidemiological information and antimicrobial susceptibility of meningococcal isolates from 1965–1985 and 2005–2013. 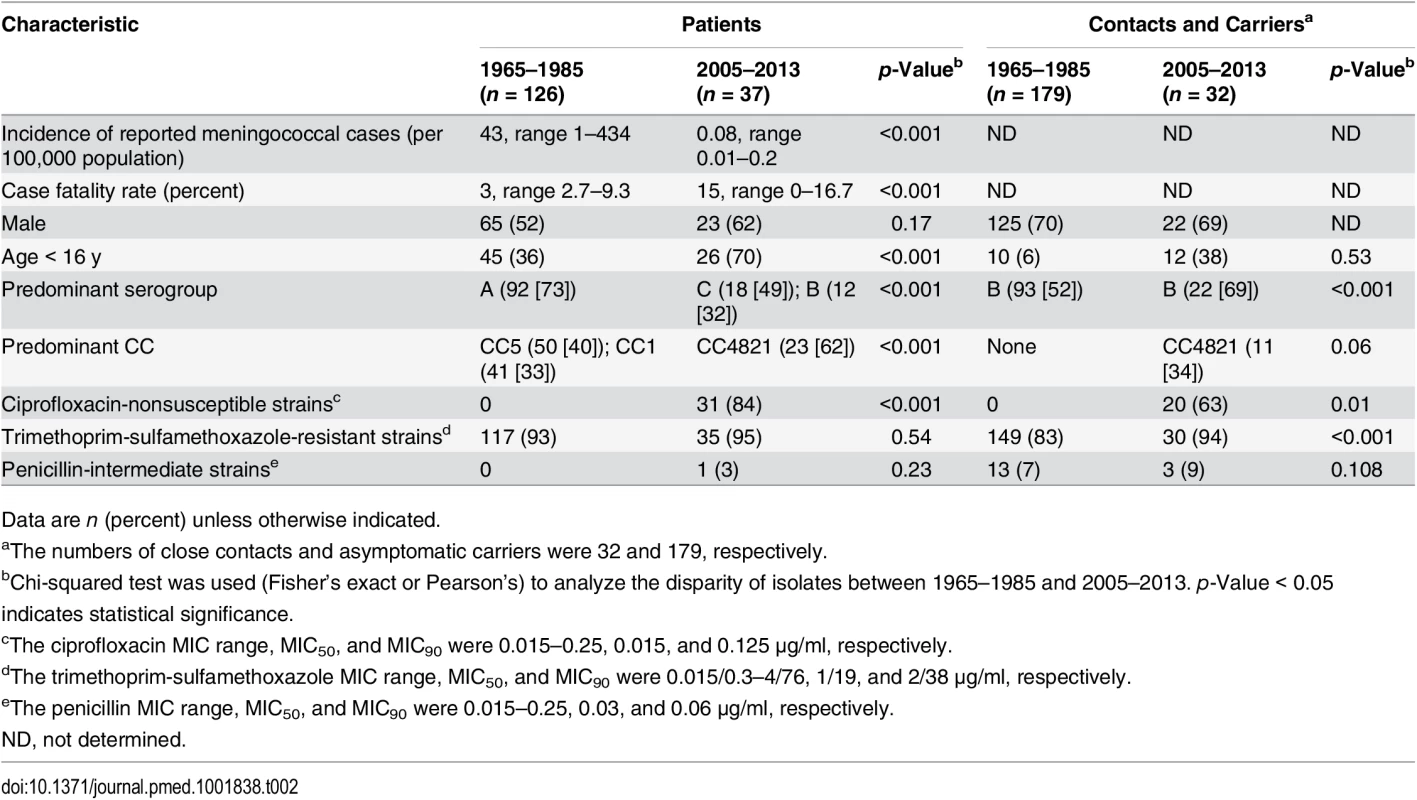
Data are n (percent) unless otherwise indicated. The case fatality rate of meningococcal disease was 15% in 1950–1960, 3% in 1961–1970, 5% in 1971–1980, 7% in 1981–1990, 4% in 1991–2000, and 10% in 2001–2013. The trend of case fatality rate over time for 1950–2013 was analyzed (Table 1). The case fatality rate was relatively stable after a sharp decrease during the 1950s (Fig 2).
Fig. 2. Case fatality rate of reported cases of meningococcal disease in Shanghai from 1950 to 2013. 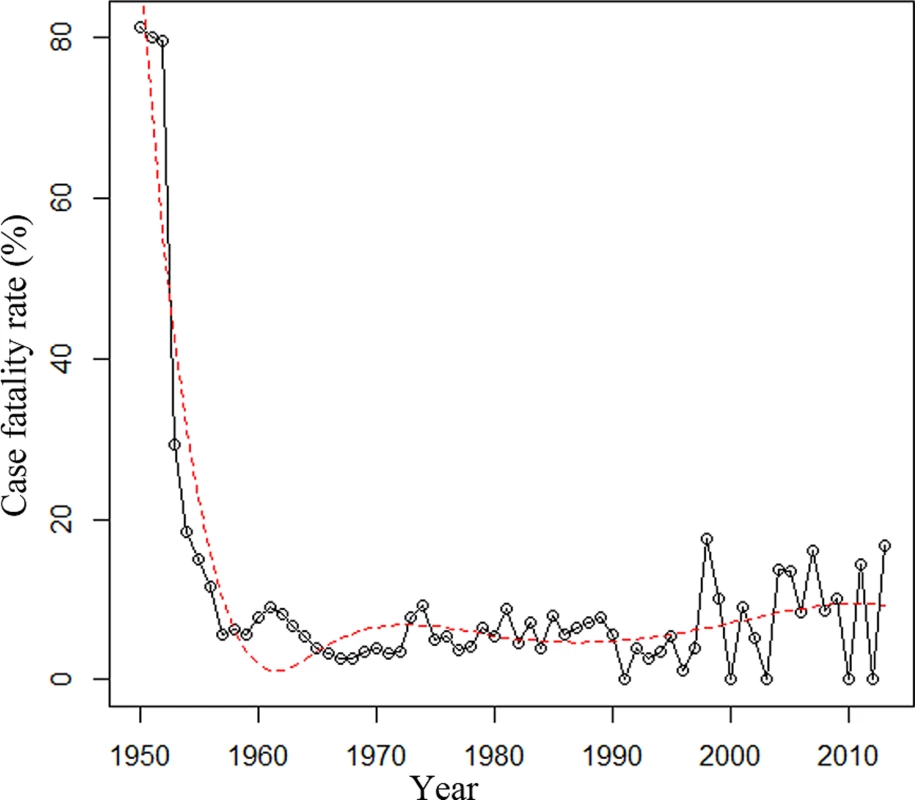
The trend was analyzed using regression splines (red dotted line). Clonal Complexes of N. meningitidis Disease Isolates over Time
Of the 126 N. meningitidis disease isolates from 1965–1985 that were available for this study, serogroup A was predominant (73%, 92/126), followed by B (17%) and C (7%). The serogroup A ST-5 complex/subgroup III complex (CC5; 84%, 48/57) was predominant in 1965–1973, and was responsible for the epidemic in 1967, while serogroup A ST-1 complex/subgroup I/II complex (CC1; 58%, 40/69) predominated in 1974–1985 (Fig 3).
Fig. 3. Minimum spanning tree analysis of multi-locus sequence types of N. meningitidis from patients. 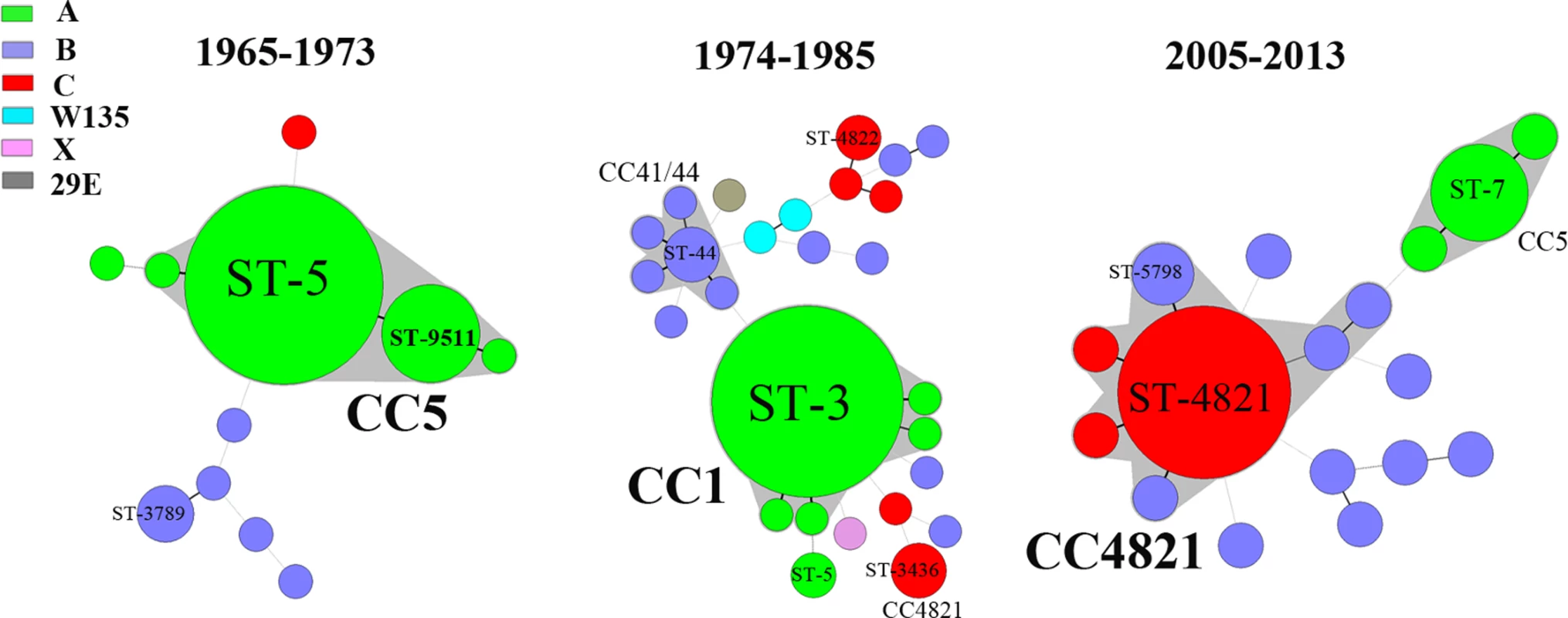
Isolates were obtained from patients during the periods 1965–1973, 1974–1985, and 2005–2013. STs are displayed as circles. The size of each circle indicates the number of isolates of this particular type. Serogroup is distinguished by different colors. The shaded halo surrounding the STs encompasses related STs that belong to the same CC. Heavy solid lines represent SLVs, and light solid lines represent DLVs. During 2005–2013, among the 125 suspected cases of meningococcal disease reported in total, 58 cases were confirmed, 47 by culture and 11 by PCR. With the exception of ten isolates from 2005 that became nonviable during storage in the hospital laboratory, 37 disease isolates were available for characterization. Serogroup C (49%, 18/37) was predominant, followed by serogroup B (32%, 12/37) and A (19%, 7/37). The ST-4821 complex (CC4821; 62%, 23/37) of serogroup C (n = 18) or B (n = 5) was predominant (Fig 3), while all seven serogroup A strains were assigned to CC5.
Carriage Surveys
During 1965–1985, 11 surveys of meningococcal carriage were performed. A total of 2,402 meningococcal isolates were isolated from the 23,083 pharyngeal swabs, with an average carriage rate of 10% (range, 2% in 1972 to 24% in 1967). The proportion of serogroup A strains in 1966 and 1967 was more than 70%; however, thereafter it decreased from 50% in 1970 to 0.5% in 1978 (p < 0.001), accompanied by an increase in serogroup B, serogroup C, or other serogroups (S2 Table). Only 164 isolates from these surveys were kept in storage and were thus available for this study.
Two surveys were performed during 2005–2013. In 2007, a total of 553 pharyngeal swabs were collected from healthy individuals with a median age of 5 y (range, 3 mo to 49 y). N. meningitidis was isolated from 11 survey participants (2%), from two age groups: 7–14 y (10%, 9/90) and ≥20 y (2%, 2/94). In 2010, 644 individuals with a median age of 5 y (range, 3 mo to 54 y) were investigated. Four (0.6%) were positive for N. meningitidis carriage, all in the age group ≥20 y (4%, 4/92). Among the 15 isolates from the two carriage surveys, 13 (87%) were serogroup B, one was W, and one was non-groupable, while no serogroup A or C strains were found (S2 Table).
Antimicrobial Susceptibility
A total of 374 N. meningitidis isolates were tested for antimicrobial susceptibility, including the 163 disease isolates, 32 isolates from close contacts, and 179 asymptomatic carriage isolates. All meningococcal isolates from meningococcal cases in 1965–1985 (n = 126) were susceptible to ciprofloxacin (MIC ≤ 0.015 μg/ml for all 126 isolates), while 84% (31/37) of the disease isolates from 2005–2013 (p < 0.001) were ciprofloxacin-nonsusceptible (Table 2), including 30 resistant and one intermediate isolates (MIC = 0.06 μg/ml [n = 1], 0.125 μg/ml [n = 29], and 0.25 μg/ml [n = 1]; MIC50 and MIC90 were both 0.125 μg/ml). The 31 ciprofloxacin-nonsusceptible strains had nalidixic acid MICs ranging from 16 to 128 μg/ml. The resistance frequencies for trimethoprim-sulfamethoxazole were 93% and 95% in 1965–1985 and 2005–2013 (p = 0.13), respectively. Only one isolate in 2005–2013 was intermediately resistant to penicillin (MIC = 0.38 μg/ml).
During 1965–1985, all of the 179 isolates from 15 close contacts and 164 asymptomatic carriers were ciprofloxacin susceptible (MIC ≤ 0.015 μg/ml), while 76% (13/17) of close contact isolates and 47% (7/15; five in 2007 and two in 2010) of carriage isolates (p = 0.005) in 2005–2013 were ciprofloxacin-nonsusceptible (MIC = 0.06–0.25 μg/ml). The frequency of resistance to trimethoprim-sulfamethoxazole was 83% in 1965–1985 and 94% in 2005–2013 (p = 0.10). Among the isolates from close contacts, the penicillin intermediate (MIC = 0.125–0.25 μg/ml) rate was 0% and 6% (1/17) during the two periods (p = 0.52), respectively, while among the carriage isolates, the rate was 8% (13/164) and 13% (2/15) during the two periods (p = 0.45), respectively (Table 2).
All 374 isolates were susceptible to the other antimicrobial agents tested, including cefotaxime, ceftriaxone, meropenem, chloramphenicol, azithromycin, minocycline, and rifampin.
Characterization of 31 Ciprofloxacin-Nonsusceptible Meningococcal Cases
Thirty-one cases of invasive meningococcal disease from February 2005 to April 2013 for which the isolates were available were caused by ciprofloxacin-nonsusceptible strains, including 17 serogroup C, seven serogroup B, and seven serogroup A. Patients were distributed across 12 of the 17 districts of Shanghai, especially in the central urban area (S1 Fig), and two cases in 2013 were likely imported cases from Jiangsu Province and Hong Kong. The median age of the patients was 4 y (range, 2 mo to 43 y), 13 (42%) were students, 20 (65%) were male, and all denied recent foreign travel. Fourteen patients had been immunized with serogroup A polysaccharide vaccine; ten of them were infected with serogroup C strains, one with a serogroup A strain, and three with serogroup B strains. Seven patients had not received any meningococcal vaccine, and there was no immunization record available for ten patients. The investigation identified no common exposures, social settings, or other epidemiological relationship among these patients.
Based on the classification system of clinical presentation [36], the clinical symptoms of 31 ciprofloxacin-nonsusceptible meningococcal disease cases could be divided into three groups, namely, fulminant meningococcal septicemia (48%, 15/31), distinct meningitis (32%, 10/31), and distinct meningitis and persistent septic shock (19%, 6/31). Despite appropriate treatment with penicillin, ceftriaxone, or meropenem, three (10%) patients died.
Close contacts of all 31 patients were investigated, and no additional meningococcal infections were detected. Among the 17 close contacts with carriage of N. meningitidis after 2005, one was administered norfloxacin, and the other 16, including two children, were given oral cefradine (n = 9) or trimethoprim-sulfamethoxazole (n = 7). Thirteen close contacts (age range 8 to 59 y) of nine cases, including five cases without proof of meningococcus by culture or PCR (clinically diagnosed meningococcal cases), were found to carry ciprofloxacin-nonsusceptible strains by pharyngeal swab culture (Fig 4).
Fig. 4. Dendrogram of 51 ciprofloxacin-nonsusceptible N. meningitidis strains from patients and carriers in Shanghai, China, 2005–2013, constructed using pulsed-field gel electrophoresis—NheI. 
The clones ChinaCC4821-R1-C/B and ChinaCC5-R14-A are indicated in grey shading, with the relatedness values marked on the roots. ●The PFGE data for the Anhui outbreak strain 053442 were obtained from PulseNet China. §Clonal dissemination between patients and their close contacts was observed. †Imported cases. NG, non-groupable. gyrA Mutations in 51 Ciprofloxacin-Nonsusceptible Strains
Mutations in the QRDR of gyrA associated with quinolone resistance substitutions were observed in all of the 51 ciprofloxacin-nonsusceptible strains from patients (n = 31), close contacts (n = 13), and asymptomatic carriers (n = 7), of which 49 harbored the typical substitution of threonine to isoleucine at amino acid position 91 (T91I). The other two ciprofloxacin-intermediate strains (MIC = 0.06 μg/ml) harbored aspartate to asparagine substitutions at amino acid position 95 (D95N) (Table 3). No additional mutations were observed in the QRDRs of gyrB, parC, or parE. In addition, there were no ciprofloxacin-nonsusceptible isolates with more than a 4-fold reduction in MIC for ciprofloxacin in the presence of reserpine, or with missense or nonsense mutations in mtrR.
Tab. 3. gyrA alleles in 51 ciprofloxacin-nonsusceptible N. meningitidis strains. 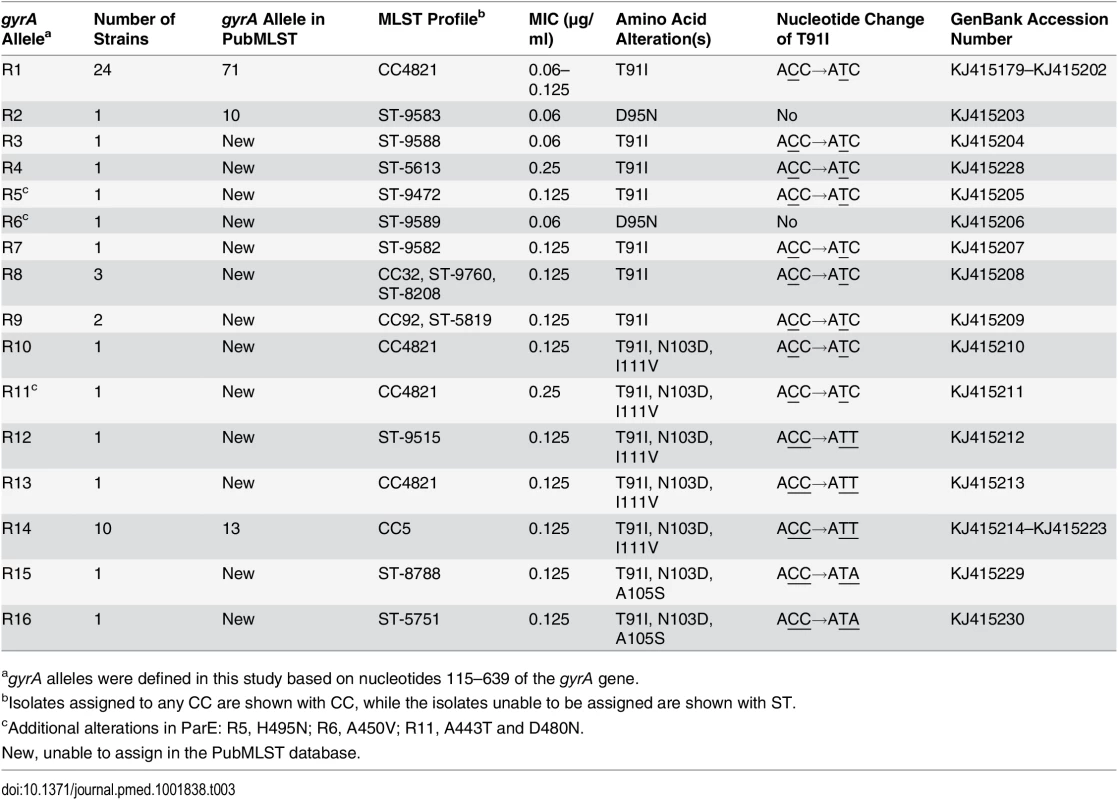
agyrA alleles were defined in this study based on nucleotides 115–639 of the gyrA gene. Sixteen gyrA alleles (R1–R16) were defined in 51 ciprofloxacin-nonsusceptible isolates (Table 3), and 12 alleles (S1–S12) in 323 ciprofloxacin-susceptible isolates (GenBank accession numbers KJ415179 to KJ415238). A phylogenetic analysis was performed using the gyrA QRDRs from 658 N. meningitidis, N. gonorrhoeae, and N. lactamica isolates, which included the 374 isolates in this study and 284 isolates from GenBank and the Neisseria PubMLST database. Isolates with gyrA alleles S1–S12 and R1–R2 were grouped within the N. meningitidis cluster, R3–R9 within the N. lactamica cluster, R10–R16 outside of the three Neisseria clusters, and none were grouped within the N. gonorrhoeae cluster (S2 Fig).
Identification of Two Predominant Ciprofloxacin-Nonsusceptible Clones
Among the 31 ciprofloxacin-nonsusceptible isolates from patients, 20 (65%) were assigned to CC4821, seven (23%) to CC5, and four to either singletons (n = 3) or CC32 (n = 1). Among the 13 ciprofloxacin-nonsusceptible strains from close contacts, six were assigned to CC4821 and three to CC5, while the remaining four isolates were singletons. Among the seven ciprofloxacin-nonsusceptible strains from asymptomatic carriers, five isolates were assigned to five different singletons and two isolates to two different CCs (CC4821 and CC92) (Fig 4).
Of 34 CC4821 strains in 2005–2013, 27 (79%) were ciprofloxacin-nonsusceptible with substitution T91I in GyrA, whereas all 18 CC4821 strains from 1965–1985 were ciprofloxacin-susceptible (p < 0.001). There were 21 ciprofloxacin-nonsusceptible CC4821 strains with PFGE patterns closely related (relatedness > 95%) to the Anhui outbreak strain 053442, representing a major outbreak clone (Fig 4). We named the clone ChinaCC4821-R1-C/B, characterized as having MLST profile ST-4821 or its SLV, plus gyrA allele R1, serogroup C or B (except one non-groupable), and PFGE subtype A1 (Fig 4 and S3 Fig). All the gyrA R1 alleles of the 21 ChinaCC4821-R1-C/B strains were grouped within the N. meningitidis cluster in the phylogenetic analysis. In the complete-length gyrA gene (2,751 nucleotides), only one nucleotide change (leading to T91I) was found among the ChinaCC4821-R1-C/B strains from the ciprofloxacin-susceptible CC4821 strain with the S1 allele isolated early in 1972.
All ten CC5 strains in 2005–2013 were ciprofloxacin-resistant, while all 63 CC5 strains isolated during 1965–1985 were ciprofloxacin-susceptible (p < 0.001). The ten ciprofloxacin-resistant CC5 strains showed a PFGE relatedness of more than 95%, representing another clone that we named ChinaCC5-R14-A. This clone is characterized as having ST-7 or its SLV, gyrA allele R14, serogroup A, PorA subtype P1.20,9,35–1, and PFGE subtype K (Fig 4 and S3 Fig). In addition to the R14 allele, which grouped outside the N. meningitidis cluster, at least 141 nucleotides in the whole sequence of the gyrA gene were found to differ between resistant and susceptible CC5 strains.
Circulation of Ciprofloxacin-Nonsusceptible Clones
Amongst those cases for which a viable isolate was available (37/47, 79%), ciprofloxacin resistance was evident in 31 cases of invasive meningococcal disease (with three deaths), among which 16 (52%) cases were caused by the ChinaCC4821-R1-C/B clone, seven (23%) by the ChinaCC5-R14-A clone, four by other CC4821 strains, one by CC32, and three by singleton strains. The cases were distributed in eight and five of the 17 districts in Shanghai, respectively (S1 Fig). No obvious epidemiological links were observed among these cases. However, transmission between patients and close contacts was presumed in at least four cases based on identification of the same clones in patients and the contacts (Fig 4).
The first case was a 21-y-old factory worker (Patient 15) hospitalized with CSF-culture-confirmed meningococcal meningitis during December 2005–January 2006. The second case was a 4-y-old child (Patient 110) with blood-culture-confirmed meningococcal septicemia in January 2011. Both patients were infected with the ChinaCC4821-R1-C/B clone and recovered with penicillin treatment. The same clone was also identified from the roommate of Patient 15 (Nm002) and the mother of Patient 110 (Nm061). The third case was a 4-mo-old infant (Patient 29) who was hospitalized with CSF-culture-confirmed meningococcal meningitis in April 2006 and recovered upon treatment with penicillin and ceftriaxone. An identical ChinaCC5-R14-A strain was isolated from a throat sample from his grandfather (Nm012). The fourth case was a 3-y-old boy (Patient 18), who was born in southwest China (Guizhou Province) then moved to Shanghai. He died of clinically diagnosed meningococcal meningitis in February 2006, without any specimens available for culture. Despite the absence of isolates from this patient to provide direct proof, the ChinaCC5-R14-A clone was recovered from both the patient’s father (Nm005) and uncle (Nm007), suggesting this clone was the culprit of the fatal disease in this child.
Discussion
Here, we present shifts in N. meningitidis ciprofloxacin susceptibility, serogroup prevalence, and CC prevalence associated with the introduction and expanding use of quinolones in Shanghai, China, based on meningococcal isolates that had been stored and were available at the time of the study. The findings are distinct from studies on meningococcal quinolone resistance conducted in North America, Europe, South Africa, and other parts of China in several aspects [3,9,23,37,38]. Ciprofloxacin-nonsusceptible strains in Shanghai were found with much higher frequency in the quinolone era than in the pre-quinolone era, not only in patients but also in the healthy population, and the majority of isolates were associated with two epidemic Chinese clones, ChinaCC4821-R1-C/B and ChinaCC5-R14-A, identified as circulating in Shanghai since 2005. Also, the strains acquired resistance by different means: via a point mutation in the gyrA gene and horizontal gene transfer from other Neisseria species (possessing different gyrA genes based on phylogenetic analysis).
Since the 1970s, the incidence of meningococcal disease in Shanghai has decreased rapidly. Since 1965, at least three clonal waves of N. meningitidis causing meningococcal disease in Shanghai have occurred. Serogroup A CC5 was epidemic in 1965–1973, while serogroup A CC1 was predominant in 1974–1985, and serogroup C or B CC4821 was predominant after 2005. These changes may have been promoted by meningococcal vaccination. From 1969 to 1980, children aged 1 to 6 y in Shanghai were immunized with serogroup A meningococcal whole cell vaccine. In 1980, national immunization with serogroup A polysaccharide vaccine was initiated in China, targeting children and adolescents aged 6 mo to 15 y with a three-dose schedule (at 6, 9, and 36 mo in infants) by subcutaneous injection in the upper arm [39]. At the same time, a catch-up campaign was implemented targeting high-risk populations, such as sales personnel in department stores, railway station workers, and soldiers in the army. Because of the Anhui serogroup C CC4821 outbreak during 2003–2005, the national meningococcal immunization program then expanded to include both serogroups A and C, with four injections in children aged 6 mo to 15 y. Polysaccharide vaccine A is administered to infants at age 6 and 9 mo, while polysaccharide vaccine A plus C is given at age 3 and 6 y.
The changes in meningococcal CCs in Shanghai reflect similar shifts taking place in the whole country. During the 1950s–1990s, the proportion of serogroup A was more than 95% in China [14], while in 2003–2008, it declined to 35%, and the proportion of serogroup C rose to 59% [40]. Correspondingly, CC4821 (55%) became predominant in China, followed by CC5 (37%) [40].
The carriage of serogroup A strains in the healthy population in Shanghai was found to have decreased significantly (p < 0.001) since 1970, just after the serogroup A vaccination campaign, and a low carriage (0.08%, 1/1,311) was also found in 2011 in Guangxi Province, China [15]. A similar trend has been observed in Africa, the main region of serogroup A meningococcal disease cases [1]. The incidence of serogroup A in South Africa decreased by 95% from 2003 to 2007 [41], and a similar decrease in the rate of carriage (94%) and incidence (95%) was observed in the African “meningitis belt” after the introduction of the serogroup A conjugate vaccine MenAfriVac in 2010 [42].
Furthermore, serogroup B and other serogroup strains, which comprised 30% (8/27) of the ciprofloxacin-nonsusceptible CC4821 strains (Fig 4), may have facilitated the spread of ciprofloxacin resistance, since no vaccines have been licensed in China to provide broad coverage that includes serogroup B meningococci. Bacterial dynamics should be monitored both in pharyngeal carriage of N. meningitidis and bacterial meningitis at the population level in order to prevent epidemics of these hypervirulent clones in the future.
This study found that up to 84% of N. meningitidis disease isolates were ciprofloxacin-nonsusceptible in 2005–2013 (quinolone era), even though quinolones were rarely used for meningococcal chemoprophylaxis in Shanghai during this study period. This nonsusceptibility rate was much higher than that observed in North Dakota and Minnesota in the United States between 2007 and 2008 (9%, 3/33) [3]. Besides in the disease isolates, a high frequency of ciprofloxacin nonsusceptibility was also found in isolates from asymptomatic carriers (47%) after 2005. In a report from the China CDC in 2014 [38], a ciprofloxacin nonsusceptibility rate of 72% (353/487) was mentioned, though no further details were provided. Current recommendations for meningococcal chemoprophylaxis in the national scheme for preventing and controlling meningococcal meningitis in China include the use of fluoroquinolones, cephalosporins, chloramphenicol, rifampin, or sulfafurazole, which have not been changed since 2005, when the scheme was approved by Ministry of Public Health of China. In practice, ciprofloxacin and amoxicillin were used as prophylaxis in the CC4821 outbreak (2004), and cefixime was used in a case of serogroup W (2012) in Anhui [43,44], while trimethoprim-sulfamethoxazole and cefradine tablets have been used in sporadic cases in Shanghai since 2006. Among these agents, fluoroquinolones should be used with caution for chemoprophylaxis based on the high frequency of ciprofloxacin-resistant N. meningitidis in China, given the presence of safe and effective alternatives such as ceftriaxone and rifampin. Sulfonamides and chloramphenicol should not be used, for the reason of either high frequency of resistance or frequent adverse effects. Azithromycin in a single 500-mg dose is effective to eradicate pharyngeal carriage and could be considered [4,45].
The high frequency of ciprofloxacin nonsusceptibility in Shanghai seems to be mainly linked with the emergence and spread of CC4821 and CC5 strains. Eighty-seven percent (27/31) of the ciprofloxacin-nonsusceptible strains from patients were assigned to either CC4821 or CC5, all with the substitution T91I in GyrA. In 2005–2013, CC4821 strains showed a ciprofloxacin nonsusceptibility rate of 79% (27/34), while all CC5 strains were resistant to ciprofloxacin. Given that CC4821 and CC5 strains not only accounted for 92% of meningococcal disease isolates in 2003–2008 in China [40] but also were responsible for a high proportion of ciprofloxacin-nonsusceptible cases, it is not surprising to find that ciprofloxacin-nonsusceptible N. meningitidis is highly prevalent in China [38]. Most of the CC4821 or CC5 strains are genetically homogenous, and we designated them ChinaCC4821-R1-C/B and ChinaCC5-R14-A, respectively. We found that these specific clones have been circulating in Shanghai since 2005, disseminating among some patients and their close contacts and facilitating the spread of ciprofloxacin resistance.
The ChinaCC4821-R1-C/B clone is unique to China, and appears to have acquired ciprofloxacin resistance by a point mutation in gyrA, leading to an amino acid change of T91I in GyrA, compared with the whole sequence of gyrA in the early ST-4821 strain in 1972. The Anhui outbreak strain 053442 was the earliest ChinaCC4821-R1-C/B clone we could identify (Fig 5). It was isolated from the CSF sample of a patient in the Anhui outbreak in 2004, and possessed the typical molecular traits of the epidemic CC4821 strains [14,34]. By analyzing its gyrA sequence, we found that strain 053442 possessed the T91I in GyrA, with a ciprofloxacin MIC of 0.064 μg/ml (data provided by the China CDC), suggesting that fluoroquinolone resistance was already present during the Anhui outbreak.
Fig. 5. Timeline for the introduction of fluroquinolones and vaccines, and clonal waves of N. meningitidis in China. 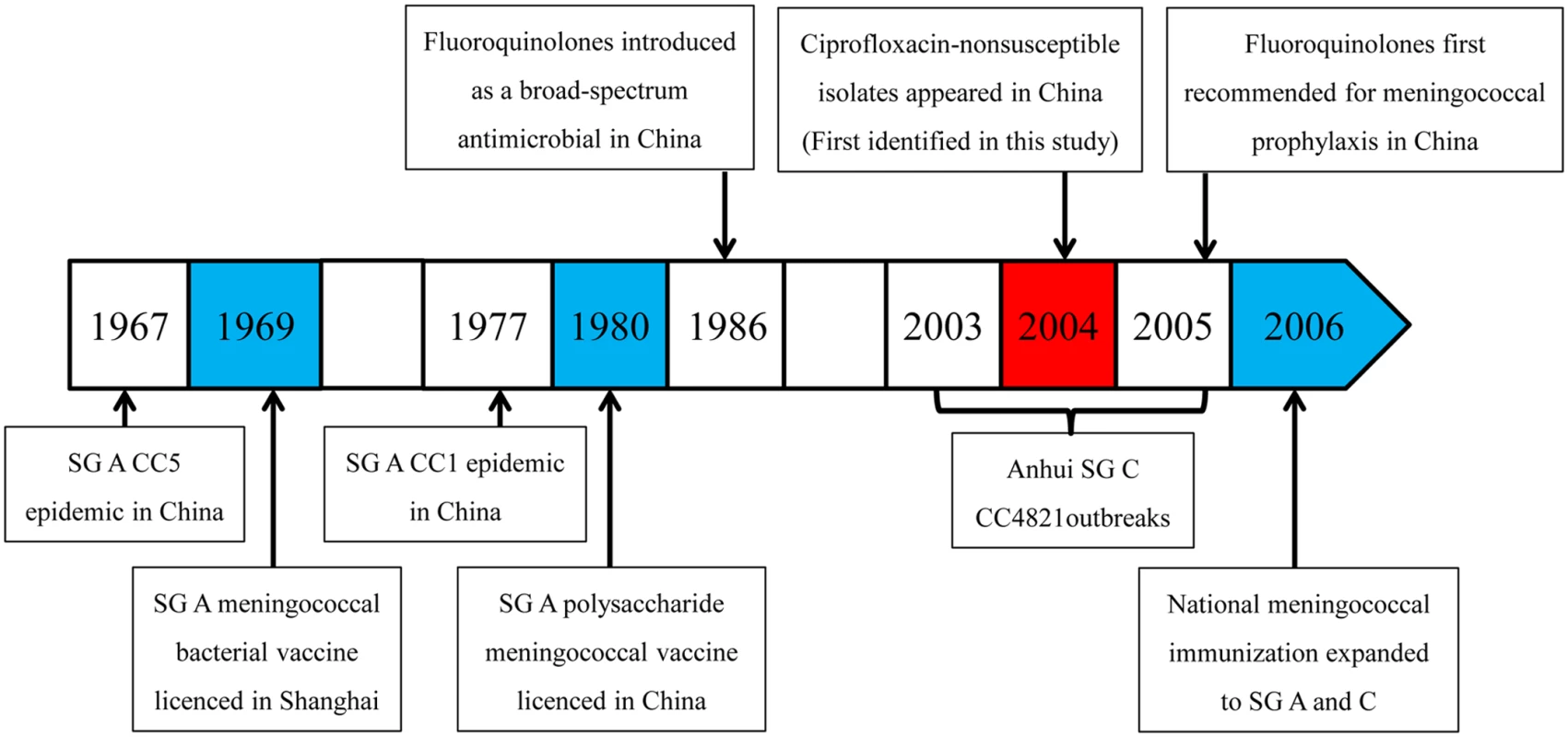
SG, serogroup. The ChinaCC5-R14-A clone belongs to the hypervirulent lineage CC5, and might have acquired resistance to ciprofloxacin via uptake and incorporation of gyrA fragments with T91I from other meningococci or Neisseria species. Responsible for three pandemics of epidemic meningitis, the serogroup A CC5 strains have spread from China to many parts of the world since the 1960s [46]. It is worth noting that ciprofloxacin-resistant CC5 strains have also been reported in Europe (n = 7) and Israel (n = 1) between 2004 and 2011 (S1 Table). In Europe, three CC5 strains were identified in 2006 and 2009 [5,29] that are genetically closely related to the ChinaCC5-R14-A clone (ST-7 or its SLV, serogroup A, PorA subtype of P1.20,9, and gyrA allele R14).
Among the 51 ciprofloxacin-nonsusceptible N. meningitidis strains, 14 strains showed diverse MLST profiles, with 79% of them belonging to serogroup B. In 2014, 71% (12/17) of serogroup W isolates were reported to be ciprofloxacin-resistant in Anhui Province [23], including four isolates from two fatal cases and 13 from close contacts. All 17 of the isolates showed the same molecular characteristics of ST-11 and P1.5,2, which started to spread in southeastern China in 2011 [15]. In our study, a serogroup W ciprofloxacin-resistant strain of CC92 from a carrier in 2010 with the R9 gyrA allele grouped within the N. lactamica cluster.
A limitation of this study is that no isolates from the period 1986–2004 in Shanghai were available, and only a small number of isolates from 1965–1985 were available. However, a study performed by the Children’s Hospital of Fudan University in Shanghai provides insights for the period 1984–2003. In that study, a total of 67 isolates were isolated from patients (age range 23 d to 13 y). It was reported that among the 67 isolates studied, 50 were isolated during 1984–1993, including 26 (52%) serogroup A, 21 (42%) B, and three non-groupable strains, while the other 17 isolates were isolated during 1994–2003 and were not tested for serogroup. Twenty-three isolates were chosen randomly to determine antimicrobial susceptibility, and all were susceptible to fluoroquinolones [47]. Therefore, it is likely that ciprofloxacin resistance emerged after the Anhui outbreak during 2003–2005. It would be interesting in the future to compare the whole genomes of ChinaCC4821-R1-C/B strains with those of the early CC4821 strains to understand the pathogenesis and evolution in virulence underpinning the expansion of this hypervirulent clone.
Our findings demonstrate that between the pre-quinolone and quinolone eras, shifts in ciprofloxacin susceptibility, serogroups, and CCs have occurred in Shanghai, China. Since 2005, ciprofloxacin nonsusceptibility in N. meningitidis has become highly prevalent in Shanghai, in association with the spread of the hypervirulent lineages CC4821 and CC5. The dissemination of two ciprofloxacin-nonsusceptible clones, ChinaCC4821-R1-C/B and ChinaCC5-R14-A, appears to have facilitated the spread of ciprofloxacin resistance.
Supporting Information
Zdroje
1. Jafri RZ, Ali A, Messonnier NE, Tevi-Benissan C, Durrheim D, Eskola J, et al. Global epidemiology of invasive meningococcal disease. Popul Health Metr. 2013;11 : 17. doi: 10.1186/1478-7954-11-17 24016339
2. Bratcher HB, Bennett JS, Maiden MC. Evolutionary and genomic insights into meningococcal biology. Future Microbiol. 2012;7 : 873–885. doi: 10.2217/fmb.12.62 22827308
3. Zalmanovici Trestioreanu A, Fraser A, Gafter-Gvili A, Paul M, Leibovici L. Antibiotics for preventing meningococcal infections. Cochrane Database Syst Rev. 2011;10:CD004785.
4. Wu HM, Harcourt BH, Hatcher CP, Wei SC, Novak RT, Wang X, et al. Emergence of ciprofloxacin-resistant Neisseria meningitidis in North America. N Engl J Med. 2009;360 : 886–892. doi: 10.1056/NEJMoa0806414 19246360
5. Lapadula G, Vigano F, Fortuna P, Dolara A, Bramati S, Soria A, et al. Imported ciprofloxacin-resistant Neisseria meningitidis. Emerg Infect Dis. 2009;15 : 1852–1854. doi: 10.3201/eid1511.090833 19891885
6. Skoczynska A, Alonso JM, Taha MK. Ciprofloxacin resistance in Neisseria meningitidis, France. Emerg Infect Dis. 2008;14 : 1322–1323. doi: 10.3201/eid1408.080040 18680672
7. Singhal S, Purnapatre KP, Kalia V, Dube S, Nair D, Deb M, et al. Ciprofloxacin-resistant Neisseria meningitidis, Delhi, India. Emerg Infect Dis. 2007;13 : 1614–1616. doi: 10.3201/eid1310.060820 18258023
8. Tzanakaki G, Blackwell CC, Kremastinou J, Kallergi C, Kouppari G, Weir DM. Antibiotic sensitivities of Neisseria meningitidis isolates from patients and carriers in Greece. Epidemiol Infect. 1992;108 : 449–455. 1601078
9. Hong E, Hedberg ST, Abad R, Fazio C, Enriquez R, Deghmane AE, et al. Target gene sequencing to define the susceptibility of Neisseria meningitidis to ciprofloxacin. Antimicrob Agents Chemother. 2013;57 : 1961–1964. doi: 10.1128/AAC.02184-12 23357770
10. Chu YW, Cheung TK, Tung V, Tiu F, Lo J, Lam R, et al. A blood isolate of Neisseria meningitidis showing reduced susceptibility to quinolones in Hong Kong. Int J Antimicrob Agents. 2007;30 : 94–95. 17408928
11. Corso A, Faccone D, Miranda M, Rodriguez M, Regueira M, Carranza C, et al. Emergence of Neisseria meningitidis with decreased susceptibility to ciprofloxacin in Argentina. J Antimicrob Chemother. 2005;55 : 596–597. 15728148
12. Wang JF, Caugant DA, Li X, Hu X, Poolman JT, Crowe BA, et al. Clonal and antigenic analysis of serogroup A Neisseria meningitidis with particular reference to epidemiological features of epidemic meningitis in the People’s Republic of China. Infect Immun. 1992;60 : 5267–5282. 1452360
13. Li J, Li Y, Yin Z, Yang J, Shao Z, Ning X, et al. [Epidemiological analysis of the meningococcal disease in China During 1997–2006.] China J Vaccine Immun. 2007;13 : 453–456.
14. Shao Z, Li W, Ren J, Liang X, Xu L, Diao B, et al. Identification of a new Neisseria meningitidis serogroup C clone from Anhui province, China. Lancet. 2006;367 : 419–423. 16458767
15. Zhou H, Liu W, Xu L, Deng L, Deng Q, Zhuo J, et al. Spread of Neisseria meningitidis serogroup W clone, China. Emerg Infect Dis. 2013;19 : 1496–1499. doi: 10.3201/eid1909.130160 23965378
16. Fagan M, Maehlen M, Lindbaek M, Berild D. Antibiotic prescribing in nursing homes in an area with low prevalence of antibiotic resistance: compliance with national guidelines. Scand J Prim Health Care. 2012;30 : 10–15. doi: 10.3109/02813432.2011.629156 22188479
17. Taur Y, Smith MA. Adherence to the Infectious Diseases Society of America guidelines in the treatment of uncomplicated urinary tract infection. Clin Infect Dis. 2007;44 : 769–774. 17304445
18. Guerrant RL, Van Gilder T, Steiner TS, Thielman NM, Slutsker L, Tauxe RV, et al. Practice guidelines for the management of infectious diarrhea. Clin Infect Dis. 2001;32 : 331–351. 11170940
19. Wood J, Butler CC, Hood K, Kelly MJ, Verheij T, Little P, et al. Antibiotic prescribing for adults with acute cough/lower respiratory tract infection: congruence with guidelines. Eur Respir J. 2011;38 : 112–118. doi: 10.1183/09031936.00145810 21233267
20. Hanlon JT, Aspinall SL, Semla TP, Weisbord SD, Fried LF, Good CB. et al. Consensus guidelines for oral dosing of primarily renally cleared medications in older adults. J Am Geriatr Soc. 2009;57 : 335–340. doi: 10.1111/j.1532-5415.2008.02098.x 19170784
21. Wang F, Zhu D, Wang Y. Surveillance of bacterial resistance to quinolones. Drugs. 1993;45 : 170–171.
22. Van Boeckel TP, Gandra S, Ashok A, Caudron Q, Grenfell BT, Levin SA, et al. Global antibiotic consumption 2000 to 2010: an analysis of national pharmaceutical sales data. Lancet Infect Dis. 2014;14 : 742–750. doi: 10.1016/S1473-3099(14)70780-7 25022435
23. Hu S, Zhang W, Li F, Hu Z, Ma E, Zheng T, et al. Neisseria meningitidis serogroup W135 sequence type 11, Anhui Province, China, 2011–2013. Emerg Infect Dis. 2014;20 : 1236–1238. doi: 10.3201/eid2007.131138 24960586
24. Song L, Ning Y, Shen J, Fan X, Zhang C. Investigation of resistance of animal Escherichia coli from food in different periods of China. Sci China Life Sci. 2009;39 : 692–698
25. Zhu H, Wang Q, Wen L, Xu J, Shao Z, Chen M, et al. Development of a multiplex PCR assay for detection and genogrouping of Neisseria meningitidis. J Clin Microbiol. 2012;50 : 46–51. doi: 10.1128/JCM.00918-11 22090406
26. Mothershed EA, Sacchi CT, Whitney AM, Barnett GA, Ajello GW, Schmink S, et al. Use of real-time PCR to resolve slide agglutination discrepancies in serogroup identification of Neisseria meningitidis. J Clin Microbiol. 2004;42 : 320–328. 14715772
27. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing: twenty-third informational supplement. M100-S23. Wayne (Pennsylvania): Clinical and Laboratory Standards Institute; 2013.
28. Birtles A, Hardy K, Gray SJ, Handford S, Kaczmarski EB, Edwards-Jones V, et al. Multilocus sequence typing of Neisseria meningitidis directly from clinical samples and application of the method to the investigation of meningococcal disease case clusters. J Clin Microbiol. 2005;43 : 6007–6014. 16333090
29. PubMLST. Neisseria sequence typing home page. Available: http://pubmlst.org/neisseria/. Accessed October 21, 2014.
30. Feil EJ, Li BC, Aanensen DM, Hanage WP, Spratt BG. eBURST: inferring patterns of evolutionary descent among clusters of related bacterial genotypes from multilocus sequence typing data. J Bacteriol. 2004;186 : 1518–1530. 14973027
31. Popovic T, Schmink S, Rosenstein NA, Ajello GW, Reeves MW, Plikaytis B, et al. Evaluation of pulsed-field gel electrophoresis in epidemiological investigations of meningococcal disease outbreaks caused by Neisseria meningitidis serogroup C. J Clin Microbiol. 2001;39 : 75–85. 11136752
32. Stefanelli P, Fazio C, La Rosa G, Marianelli C, Muscillo M, Mastrantonio P. Rifampicin-resistant meningococci causing invasive disease: detection of point mutations in the rpoB gene and molecular characterization of the strains. J Antimicrob Chemother. 2001;47 : 219–222. 11157912
33. Li W, Lu S, Cui Z, Cui J, Zhou H, Wang Y, et al. PulseNet China, a model for future laboratory-based bacterial infectious disease surveillance in China. Front Med. 2012;6 : 366–375. doi: 10.1007/s11684-012-0214-6 23124882
34. Peng J, Yang L, Yang F, Yang J, Yan Y, Nie H, et al. Characterization of ST-4821 complex, a unique Neisseria meningitidis clone. Genomics. 2008;91 : 78–87. 18031983
35. Dean AG, Sullivan KM, Soe MM. OpenEpi: Open Source Epidemiologic Statistics for Public Health, version 3.01a. Available: http://www.OpenEpi.com. Accessed September 22, 2014.
36. Brandtzaeg P. Classification of the clinical presentations. In: Frosch M, Maiden MCJ, editors. Handbook of meningococcal disease: infection biology, vaccination, clinical management. Weinheim: Wiley; 2006. pp. 427–480.
37. du Plessis M, de Gouveia L, Skosana H, Thomas J, Blumberg L, Klugman KP, et al. Invasive Neisseria meningitidis with decreased susceptibility to fluoroquinolones in South Africa, 2009. J Antimicrob Chemother. 2010;65 : 2258–2260. doi: 10.1093/jac/dkq291 20688769
38. Zhu B, Fan Y, Xu Z, Xu L, Du P, Gao Y, et al. Genetic diversity and clonal characteristics of ciprofloxacin-resistant meningococcal strains in China. J Med Microbiol. 2014;63 : 1411–1418. doi: 10.1099/jmm.0.078600-0 25082942
39. Li F. [Immunoprophylaxis of epidemic meningococcal meningitis.] In: Zuo W, Diao L, Xu A, editors. [Scheduled immunology.] Shanghai: Shanghai Scientific and Technological Literature Press; 2010. pp. 470–471.
40. Zhou H, Gao Y, Xu L, Li M, Li Q, Li Y, et al. Distribution of serogroups and sequence types in disease-associated and carrier strains of Neisseria meningitidis isolated in China between 2003 and 2008. Epidemiol Infect. 2012;140 : 1296–1303. doi: 10.1017/S0950268811001865 21929839
41. du Plessis M, Wolter N, Crowther-Gibson P, Hamstra HJ, Schipper K, Moodley C, et al. Meningococcal serogroup Y lpxL1 variants from South Africa are associated with clonal complex 23 among young adults. J Infect. 2014;68 : 455–461. doi: 10.1016/j.jinf.2013.12.011 24393652
42. Daugla DM, Gami JP, Gamougam K, Naibei N, Mbainadji L, Narbe M, et al. Effect of a serogroup A meningococcal conjugate vaccine (PsA-TT) on serogroup A meningococcal meningitis and carriage in Chad: a community study. Lancet. 2014;383 : 40–47. doi: 10.1016/S0140-6736(13)61612-8 24035220
43. Jin Y, Wang X, Hu Z, Wang H. [Analysis on the epidemic situation of cerebro-spinal meningitis and its prevention and control measures in 2004 in Hefei City.] Anhui J Prev Med. 2006;12 : 218–222.
44. Zhang F, He Y, Sheng W. [Investigation of the first case of meningococcal meningitis W135 in Tongling, Anhui.] Dis Surveill. 2012;27 : 923–924.
45. Girgis N, Sultan Y, Frenck RW Jr, El-Gendy A, Farid Z, Mateczun A. Azithromycin compared with rifampin for eradication of nasopharyngeal colonization by Neisseria meningitidis. Pediatr Infect Dis J. 1998;17 : 816–819. 9779768
46. Zhu P, van der Ende A, Falush D, Brieske N, Morelli G, Linz B, et al. Fit genotypes and escape variants of subgroup III Neisseria meningitidis during three pandemics of epidemic meningitis. Proc Natl Acad Sci U S A. 2001;98 : 5234–5239. 11287631
47. Li Y, Zeng M, Wang X, Wang D. [Clinical epidemiology of epidemic cerebrospinal meningitis in children in Shanghai area from 1984 to 2003.] Chin J Infect Chemother. 2004;4 : 321–323.
48. Hall BG. Building phylogenetic trees from molecular data with MEGA. Mol Biol Evol. 2013;30 : 1229–1235. doi: 10.1093/molbev/mst012 23486614
Štítky
Interní lékařství
Článek vyšel v časopisePLOS Medicine
Nejčtenější tento týden
2015 Číslo 6- Příznivý vliv Armolipidu Plus na hladinu cholesterolu a zánětlivé parametry u pacientů s chronickým subklinickým zánětem
- Alternativní léčebné možnosti u hypercholesterolemie při intoleranci statinů
- Vliv kombinace nutraceutik na remodelaci levé komory srdeční u osob s metabolickým syndromem
- Nutraceutika a jejich ovlivnění mírného kardiometabolického rizika
- Princip účinku medu v léčbě chronických i infikovaných ran
-
Všechny články tohoto čísla
- Inequitable and Ineffective: Exclusion of Mental Health from the Post-2015 Development Agenda
- Mistreatment of Women in Childbirth: Time for Action on This Important Dimension of Violence against Women
- HIV Programs for Sex Workers: Lessons and Challenges for Developing and Delivering Programs
- Efficacy of Handwashing with Soap and Nail Clipping on Intestinal Parasitic Infections in School-Aged Children: A Factorial Cluster Randomized Controlled Trial
- Assessing Development Assistance for Mental Health in Developing Countries: 2007–2013
- Achieving Systemic and Scalable Private Sector Engagement in Tuberculosis Care and Prevention in Asia
- Maximizing the Impact of Training Initiatives for Health Professionals in Low-Income Countries: Frameworks, Challenges, and Best Practices
- Shifts in the Antibiotic Susceptibility, Serogroups, and Clonal Complexes of in Shanghai, China: A Time Trend Analysis of the Pre-Quinolone and Quinolone Eras
- Transmission of Multidrug-Resistant and Drug-Susceptible Tuberculosis within Households: A Prospective Cohort Study
- The Potential for Reducing the Number of Pneumococcal Conjugate Vaccine Doses While Sustaining Herd Immunity in High-Income Countries
- Effects of Hormone Therapy on Cognition and Mood in Recently Postmenopausal Women: Findings from the Randomized, Controlled KEEPS–Cognitive and Affective Study
- The Mistreatment of Women during Childbirth in Health Facilities Globally: A Mixed-Methods Systematic Review
- Exposure to Second-Hand Smoke and the Risk of Tuberculosis in Children and Adults: A Systematic Review and Meta-Analysis of 18 Observational Studies
- Associations between Potentially Modifiable Risk Factors and Alzheimer Disease: A Mendelian Randomization Study
- PLOS Medicine
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- The Mistreatment of Women during Childbirth in Health Facilities Globally: A Mixed-Methods Systematic Review
- The Potential for Reducing the Number of Pneumococcal Conjugate Vaccine Doses While Sustaining Herd Immunity in High-Income Countries
- Mistreatment of Women in Childbirth: Time for Action on This Important Dimension of Violence against Women
- Associations between Potentially Modifiable Risk Factors and Alzheimer Disease: A Mendelian Randomization Study
Přihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



