-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Top novinky
ReklamaFresh fruit consumption in relation to incident diabetes and diabetic vascular complications: A 7-y prospective study of 0.5 million Chinese adults
Huaidong Du and colleagues study associations between fruit consumption and incidence of diabetes, along with complications of prevalent diabetes, in a Chinese population.
Published in the journal: . PLoS Med 14(4): e32767. doi:10.1371/journal.pmed.1002279
Category: Research Article
doi: https://doi.org/10.1371/journal.pmed.1002279Summary
Huaidong Du and colleagues study associations between fruit consumption and incidence of diabetes, along with complications of prevalent diabetes, in a Chinese population.
Introduction
Diabetes affects more than 400 million people globally, including about a quarter in China [1], with substantial risks of premature death and a range of macrovascular (e.g., ischaemic heart disease [IHD], stroke, and peripheral vascular disease) and microvascular (e.g., nephropathy, retinopathy, and neuropathy) complications. Healthy diet plays an important role in both prevention and appropriate management of diabetes [2], and diets rich in fruit and vegetables are generally recommended [3,4], even though evidence about their effects, particularly for fruit consumption, among diabetic patients is still rather limited.
Fruit and vegetables share many common nutritional properties, but are often consumed in different settings and manners, especially in China, where fresh fruit is usually consumed raw as a snack while fresh vegetables are usually fried or stewed (often together with meat, cooking oil, and salt) as main dishes. Moreover, the sugar content in fruit is generally higher than in vegetables, leading to concerns about its potential harmful impacts on diabetes [5]. A few prospective studies have tried to assess the effects of fruit intake on risk of diabetes, but the results have been inconsistent, with some studies showing a moderately strong inverse association [6,7] and others, including those in China [8] and in European populations [9], finding no association. Furthermore, there is very limited evidence about the effects of fruit consumption on risks of death and major vascular complications among people with established diabetes [10–12]. Reliable assessment of the effects of fruit consumption on risks of incident diabetes and, among those who have already developed diabetes, on diabetic complications is urgently needed to improve dietary recommendations, especially in low - and middle-income countries such as China and other Asian countries where avoidance of sweet-tasting food (including fresh fruit) is common among diabetic patients [13–15].
With data from the China Kadoorie Biobank study, a prospective cohort study of 0.5 million adults, we examined the association of fresh fruit consumption with the risk of developing diabetes among people without baseline diabetes, and with the risks of death and major vascular hospitalisations among people with prevalent diabetes at baseline.
Methods
The China Kadoorie Biobank study was conducted in accordance with a predefined study protocol [16,17], and data analyses were performed following a prespecified analysis plan (S1 Text).
Ethics statement
Ethics approval was obtained from the Oxford University Tropical Research Ethics Committee, the Chinese Academy of Medical Sciences Ethical Review Committee, the Chinese Center for Disease Control and Prevention Ethical Review Committee, and the scientific review boards in each of the ten regional centres.
Study population
The China Kadoorie Biobank study is a large nationwide prospective cohort study involving ten geographically diverse regional sites (five urban and five rural) in China, chosen to cover a wide range of risk exposures and disease patterns, all with good-quality death and disease registries and local capacity. The study design, methods, and population have been reported previously [16,17]. In brief, between June 2004 and July 2008, all non-disabled permanent residents within preselected communities aged 35 to 74 y were invited to participate in the study, and about one in three (33% in rural areas, 27% in urban areas) responded, yielding a total of 512,891 participants (including a few who were just outside the targeted age range); all participants provided written informed consent.
Data collection
At the local study clinics, trained health workers administered a laptop-based questionnaire on socio-economic status, smoking, alcohol intake, diet, physical activity [18], and medical history; measured anthropometrics and blood pressure; and took 10 ml of venous blood for on-site testing of random blood glucose (RBG) (with time since last eating or drinking any energy-containing foods or beverages recorded) and for long-term storage. RBG level was measured immediately following sample collection using the SureStep Plus System (Johnson & Johnson), which provided plasma-equivalent readings and was regularly calibrated with manufacturer control solutions. Individuals with no prior history of physician-diagnosed diabetes but with RBG levels between 7.8 and 11.1 mmol/l were invited back the following day for a fasting blood glucose test. At baseline, individuals were considered as having prevalent diabetes if they had either a self-reported prior history of physician-diagnosed diabetes or screen-detected diabetes, which was defined as having never been diagnosed with diabetes but having a measured RBG level ≥7.0 mmol/l with time since last food/beverages ≥8 h, or ≥11.1 mmol/l with time since last food/beverages <8 h, or a fasting blood glucose level ≥7.0 mmol/l on subsequent testing [19]. Dietary data covered 12 major food groups, including fresh fruit, fresh and preserved vegetables, meat, and dairy products, each with five frequency levels about habitual consumption during the past 12 mo (daily, 4–6 d/wk, 1–3 d/wk, monthly, or never/rarely) [20].
Following the baseline survey, 5%–6% of the surviving participants were randomly selected for resurveys in 2008 (first resurvey, response rate 80%) and 2013–2014 (second resurvey, response rate 76%), using procedures similar to those at baseline. During the second resurvey, in addition to frequency, the quantity of each food group consumed was also collected, which was used as a proxy to estimate the group average consumption for each baseline category (S1 Table).
Follow-up for mortality and morbidity
All participants were followed up (2,411 [0.5%] participants were lost to follow-up by 1 January 2014) for death and disease using information collected through linkages with death and disease registries and health insurance databases. The vital status of each participant was obtained periodically through China’s Disease Surveillance Points system [21], checked annually against local residential and health insurance records, and by street committees or village administrators. In addition, information on any episodes of hospitalisation was collected through linkages with disease registries (for stroke, IHD, cancer, and diabetes) and national health insurance claim databases. Cause-specific deaths and non-fatal events were coded, blinded to baseline information, by the trained staff using ICD-10 [16]. In addition, diagnosis descriptions of diabetes-related events (both fatal and non-fatal) were reviewed and standardised centrally by study clinicians blinded to baseline fruit consumption. For the present study, incident diabetes included all reported cases (fatal or not) of new onset diabetes that occurred between ages 35 and 79 y. Underlying causes of death were classified as diabetes, cardiovascular disease (CVD), or other. Diabetes-related microvascular complications included nephropathy, retinopathy, and neuropathy. Macrovascular complications included IHD, stroke, and other (S2 Table). Only the first non-fatal event for each endpoint was considered.
Statistical analysis
For baseline characteristics, means (standard deviations) or percentages of individuals by diabetes status were calculated, adjusting for age, sex, and region, where appropriate, using either multiple linear (for continuous outcomes) or logistic regression (for binary outcomes).
Among the 482,591 participants who were free of diabetes at baseline, hazard ratios (HRs) and 95% CIs for diabetes incidence by fresh fruit consumption level were estimated using Cox proportional hazard regression. Analyses were stratified by age at risk, sex, and region and were adjusted for education (four categories), annual household income (four categories), smoking (four categories), alcohol intake (four categories), physical activity (continuous variable), body mass index (BMI) (continuous variable), survey season (four categories), family history of diabetes (dichotomous), and consumption of meat (three categories), dairy products (three categories), and preserved vegetables (five categories).
Among those 30,300 participants with diabetes at baseline, Cox regression was used to investigate the association of fruit consumption with hospitalisations due to different diabetes complications. The main analyses grouped participants into three categories of fruit consumption (<1 d/wk, 1–3 d/wk, or >3 d/wk) in order to retain a reasonable number of cases in each group. In addition to the covariates mentioned above, baseline CVD and diabetes status (both as dichotomous variables) and anti-diabetic treatment (four categories) were also adjusted for.
Separate analyses of the first and second halves of the follow-up period showed no clear deviation from the proportionality assumption. The floating absolute risk method was used to calculate the confidence intervals of HRs in all categories (including the reference category) of fruit consumption. The traditional approach with an arbitrarily chosen reference group is unsatisfactory as the standard errors and associated CIs are dependent on the precision within the reference group; therefore, comparisons can only be made with the reference group. In contrast, the floating absolute risk method estimates standard errors and CIs using “floated” variances to provide appropriate variances to the log relative risk (i.e., HR in our analyses) for all exposure categories, including the reference category. Hence, valid comparisons can be made between any two exposure groups for polychotomous risk factors [22,23]. To further quantify the linear association between fruit consumption amount and disease risks and to account for regression dilution bias [24,25], we used data from the two resurveys to estimate mean usual fruit intake (portions/month) for each baseline category (S1 Table). The group mean levels of usual consumption were used to plot against the HRs in each baseline exposure category and to yield the effect size per one daily portion (i.e., 100 g/d) of usual fruit consumption through the Cox regression analyses.
Adjusted HRs for each one daily portion of usual fruit consumption were calculated across strata of potential effect modifiers, e.g., factors related to diabetes risk and diabetes stage, and chi-square tests for trend and heterogeneity were applied to the log HRs and their standard errors. In addition, sensitivity analyses investigated the potential impacts of excluding the first 2 y of follow-up, excluding participants with prevalent CVD at baseline, and additional adjustment for other dietary factors. For analyses of diabetes incidence, sensitivity analysis was also performed excluding participants with incident CVD (i.e., myocardial infarction and stroke) during follow-up.
All analyses were conducted in SAS (version 9.2), and graphs were plotted in R 3.0.2.
Results
Of the 512,891 participants, 30,300 (5.9%) had diabetes at baseline, including 16,162 with previously diagnosed diabetes and 14,138 with screen-detected diabetes (Table 1). Based on age at diagnosis being <30 y and insulin use, 0.2% of the cases were likely to be type 1 diabetes. Individuals with diabetes were older and were more likely to be women, to live in urban areas, to be less physically active, and to have higher levels of BMI, waist circumference, and blood pressure. Among men, the proportions with current regular smoking and alcohol intake were about 10% lower in those with previously diagnosed diabetes than in those with screen-detected diabetes or those without diabetes.
Tab. 1. Selected characteristics of participants by baseline diabetes status. 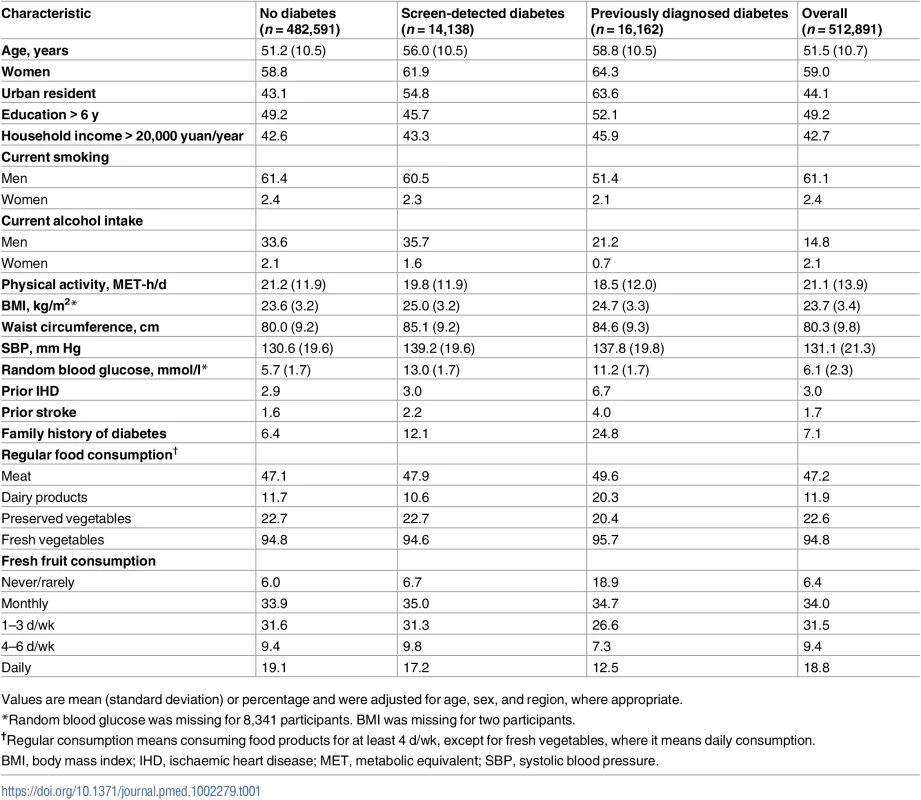
Values are mean (standard deviation) or percentage and were adjusted for age, sex, and region, where appropriate. Overall, 18.8% of participants reported consuming fresh fruit daily (daily consumers) and 6.4% never or rarely (non-consumers). The proportion of non-consumers among those with previously diagnosed diabetes (18.9%) was about three times higher than among those with screen-detected diabetes (6.7%) and those without diabetes (6.0%). There was an overall weak inverse association of fruit consumption with blood glucose (RBG was 0.6 mmol/l lower among daily consumers than non-consumers; S3 Table). A similar inverse association was seen among those with previously diagnosed diabetes, even after additionally adjusting for all other potential confounders including fasting time and anti-diabetic medications (S4 Table).
During ~7 y (3.4 million person-years) of follow-up, 9,504 new onset cases of diabetes were recorded among the 482,591 participants without diabetes at baseline, with an overall incidence rate of 2.8 per 1,000 person-years (Table 2). Participants with higher fruit consumption had a significantly lower risk of developing diabetes, with the adjusted HR for daily consumers versus non-consumers being 0.88 (95% CI 0.83–0.93) (Fig 1A). After adjusting for regression dilution bias, there was a clear log-linear dose–response relationship, with each one daily portion of fruit associated with an adjusted HR of 0.88 (95% CI 0.81–0.95) (p for trend = 0.01). This association was not significantly modified by sex, age, region, survey season, or a range of other factors including smoking, alcohol consumption, physical activity, BMI, and family history of diabetes (p for trend or heterogeneity ≥ 0.2 for all; S1 Fig). Excluding baseline or incident IHD and stroke, additionally adjusting for other dietary variables, or excluding the first 2 y of follow-up did not materially alter the results (S5 Table).
Tab. 2. Incidence rate and 5-y risk of incident diabetes and, among those with prevalent diabetes at baseline, diabetes complications by level of fresh fruit consumption. 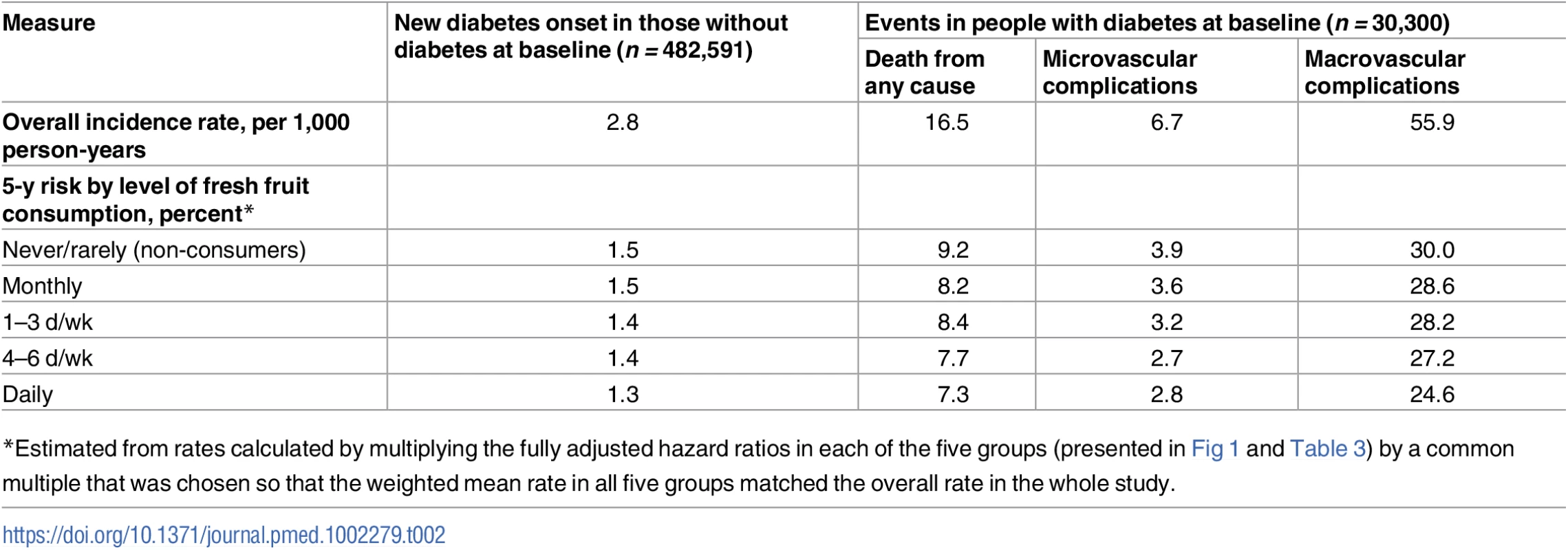
*Estimated from rates calculated by multiplying the fully adjusted hazard ratios in each of the five groups (presented in Fig 1 and Table 3) by a common multiple that was chosen so that the weighted mean rate in all five groups matched the overall rate in the whole study. Fig. 1. Adjusted hazard ratios for incident diabetes and all-cause mortality among those with diabetes at baseline, by fresh fruit consumption. 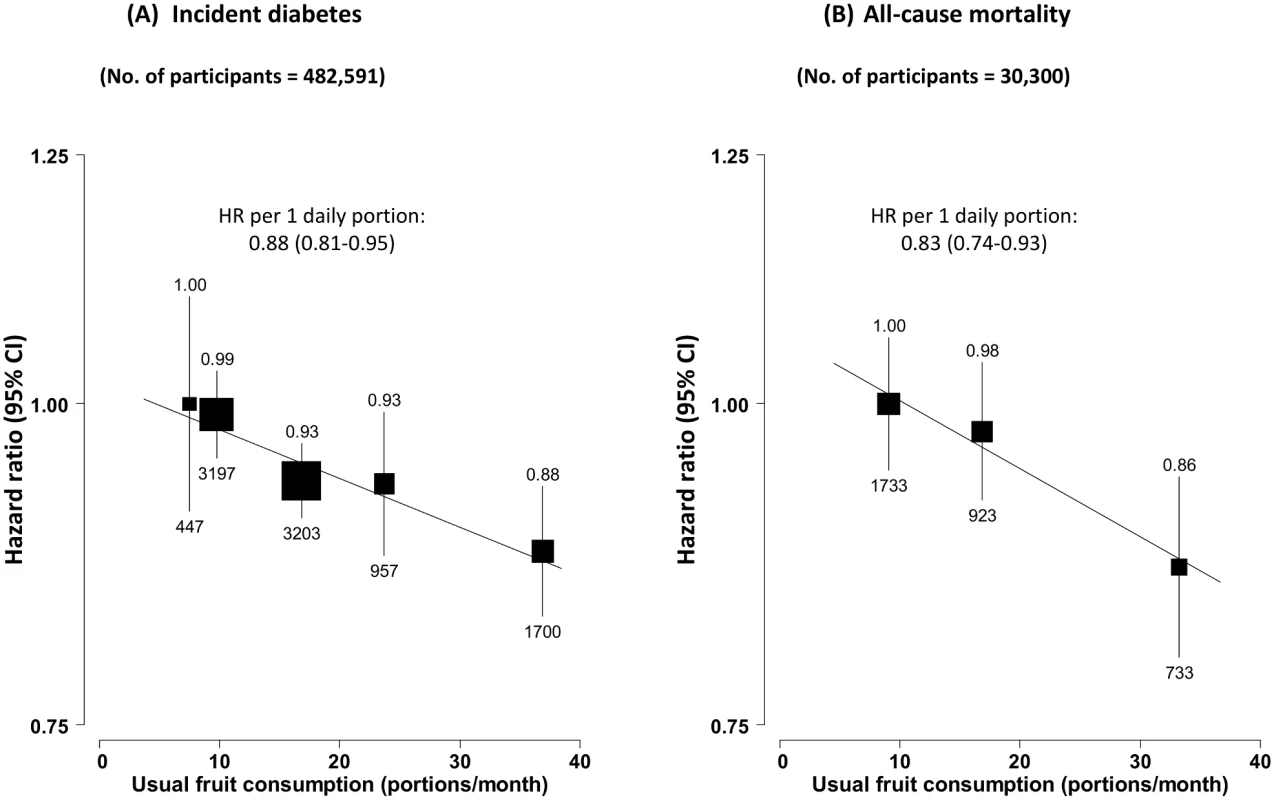
(A) Incident diabetes; (B) all-cause mortality among those with diabetes at baseline. Analyses were stratified by age at risk, sex, and region and were adjusted for education, income, alcohol intake, smoking, physical activity, survey season, BMI, family history of diabetes, and intakes of dairy products, meat, and preserved vegetables. The black boxes represent the hazard ratios (HRs), with the size inversely proportional to the variance of the log HRs, and the vertical lines represent the 95% confidence intervals. The values above the vertical lines are the point estimates of the HRs, and the values below them are the numbers of cases. Among the 30,300 participants who had prevalent diabetes at baseline, 3,389 (11.2%) died during follow-up (overall mortality rate 16.5 per 1,000), including 1,459 (43.1%) from CVD, 512 (15.1%) from diabetes (i.e., acute diabetic crises or other unspecified diabetes deaths without any immediate or antecedent cause of death, e.g., vascular or renal cause of death), and 1,418 (41.8%) from other causes, including cancer (n = 790). Fruit consumption was significantly and inversely associated with mortality from all causes (Fig 1B), diabetes, and CVD, but not with mortality from other causes (including cancer) (Fig 2). Compared to those who consumed fresh fruit <1 d/wk, individuals who consumed fruit >3 d/wk had adjusted HRs of 0.86 (95% CI 0.80–0.94) for all-cause mortality, 0.64 (0.48–0.86) for diabetes mortality, and 0.81 (0.72–0.92) for CVD mortality. These associations were approximately log-linear after correcting for regression dilution bias, with each one daily portion of fruit associated with HRs of 0.83 (95% CI 0.74–0.93), 0.59 (0.40–0.87), and 0.78 (0.65–0.93), respectively.
Fig. 2. Adjusted hazard ratios for selected cause-specific mortality by fresh fruit consumption among 30,300 participants with diabetes at baseline. 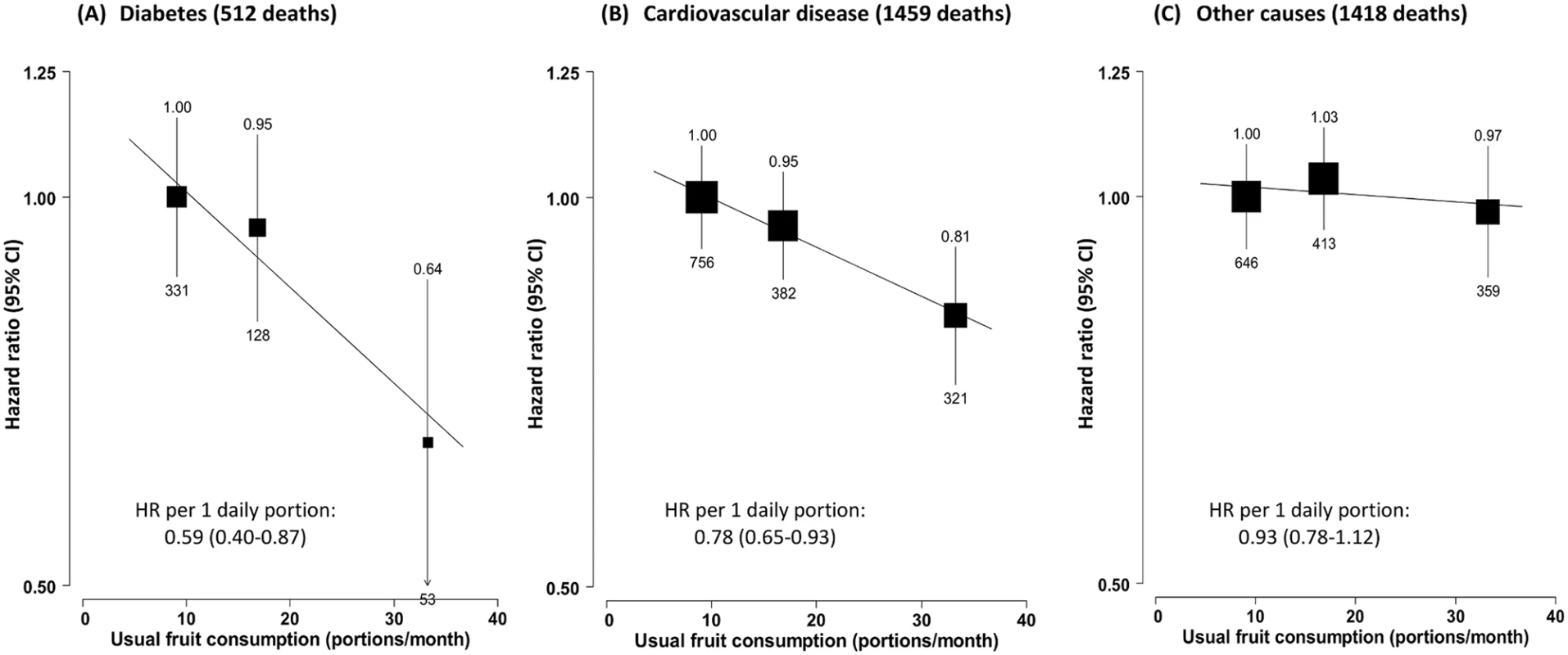
Mortality from (A) diabetes, (B) cardiovascular disease, and (C) other causes. Conventions as in Fig 1. Baseline status for cardiovascular disease, diabetes, and anti-diabetic treatment were also adjusted for. HR, hazard ratio. Fruit consumption was also inversely associated with risk of hospitalisation due to diabetic vascular complications. Compared to consuming fruit <1 d/wk, consuming fruit >3 d/wk was associated with a 26% (95% CI 16%–34%) lower risk of microvascular complications and 10% (6%–14%) lower risk of macrovascular complications. Each daily portion was associated with HRs of 0.72 (95% CI 0.61–0.87) and 0.87 (0.82–0.93), respectively (Fig 3). The strength of the association of fruit consumption with individual complications was similar (Fig 4).
Fig. 3. Adjusted hazard ratios for macro- and microvascular complications of diabetes by fresh fruit consumption among 30,300 participants with diabetes at baseline. 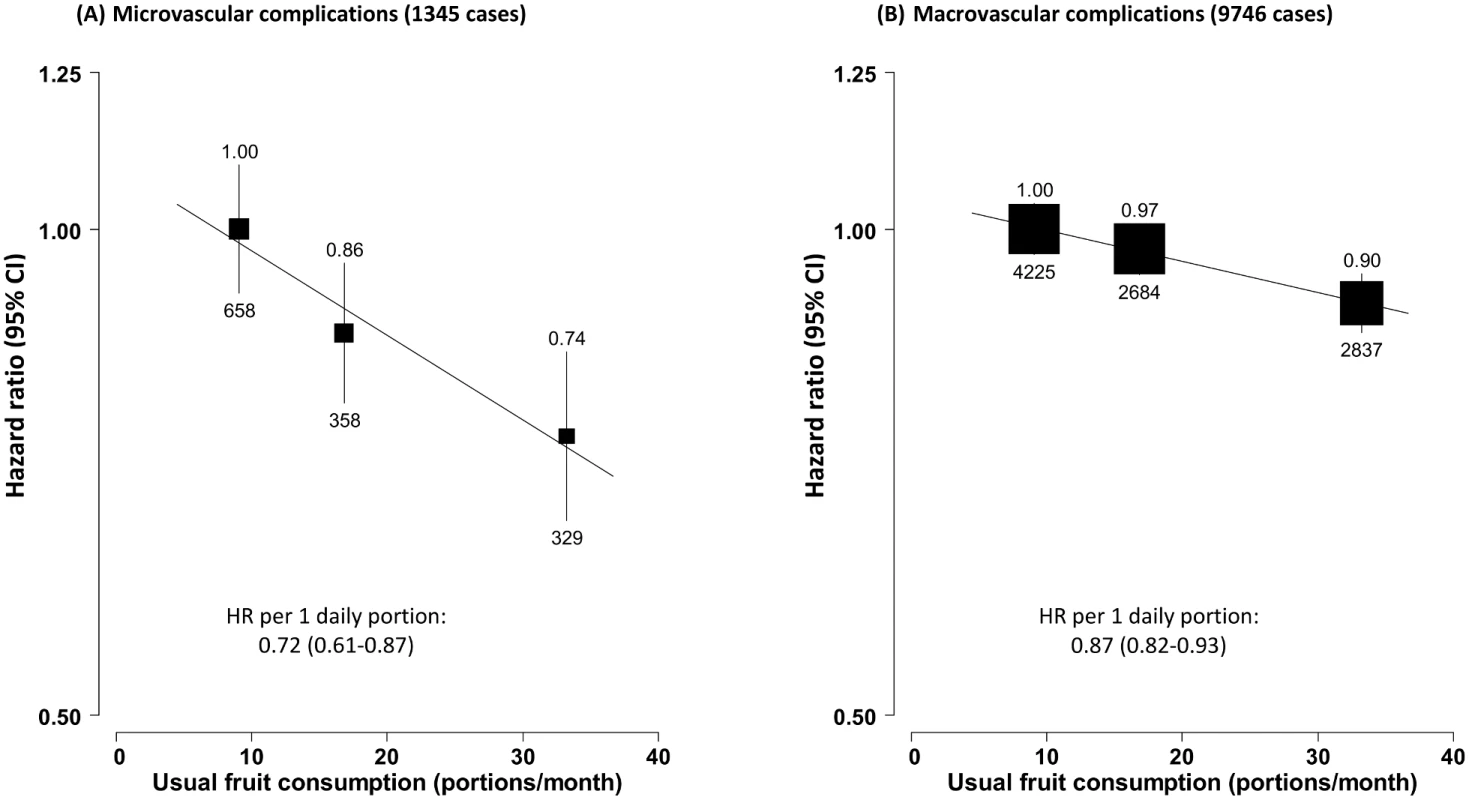
(A) Microvascular complications; (B) macrovascular complications. Conventions as in Fig 1. HR, hazard ratio. Fig. 4. Adjusted hazard ratios for major diabetes-related vascular complications by fresh fruit consumption among 30,300 participants with diabetes at baseline. 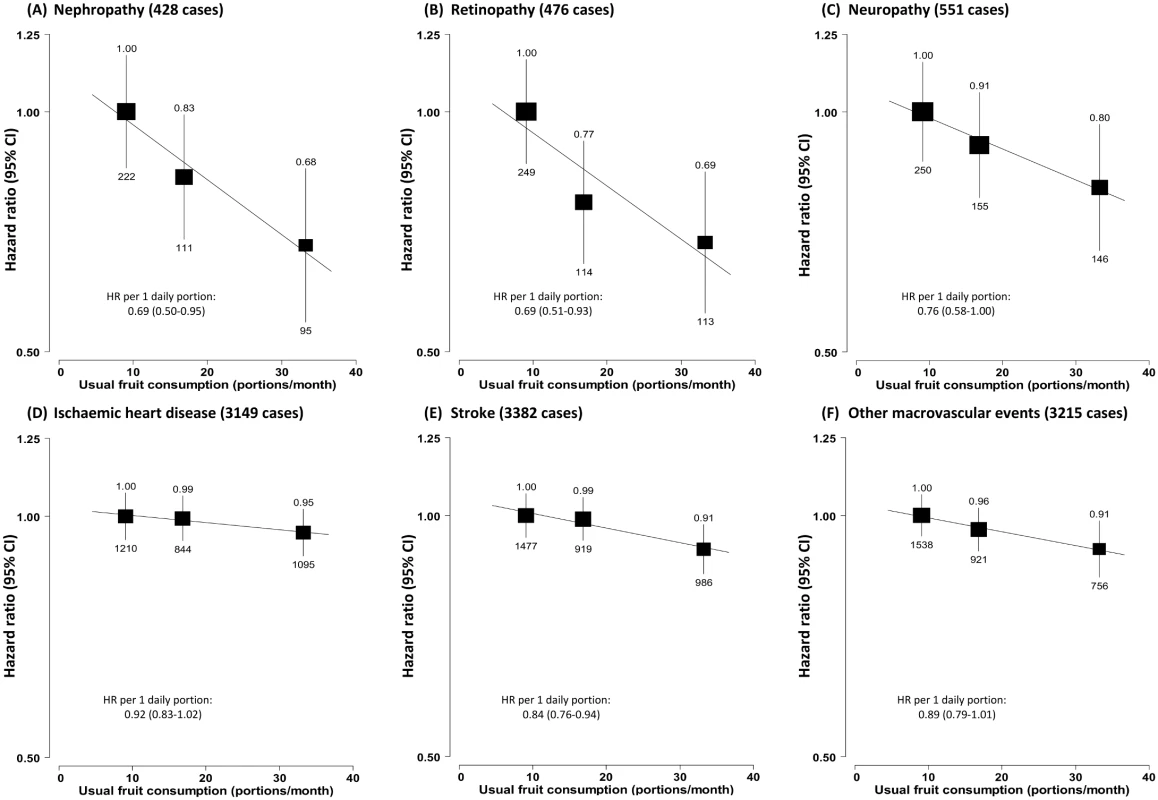
(A) Nephropathy; (B) retinopathy; (C) neuropathy; (D) ischaemic heart disease; (E) stroke; (F) other macrovascular events. Conventions as in Fig 1. HR, hazard ratio. The strength of the associations between fruit consumption and diabetic complications were largely consistent across subgroups of participants classified by baseline characteristics, survey season, baseline RBG level, and diabetes status (previously diagnosed versus screen-detected) (p for trend or heterogeneity ≥ 0.1 for all; S2 Fig). Anti-diabetic treatment, age of diabetes onset, and diabetes duration did not seem to modify the associations either (S3 Fig).
Similar findings were observed when fresh fruit consumption was analysed in the original five categories, although the trend was somewhat less consistent due to the smaller number of cases in each group (Table 3). Additional adjustment for other dietary variables, excluding the first 2 y of follow-up, or excluding diabetic participants with prevalent IHD or stroke at baseline (n = 3,885) did not materially alter the observed associations (S5 Table).
Tab. 3. Number of events and adjusted hazard ratios (95% CIs) for diabetic complications according to five categories of fresh fruit consumption. 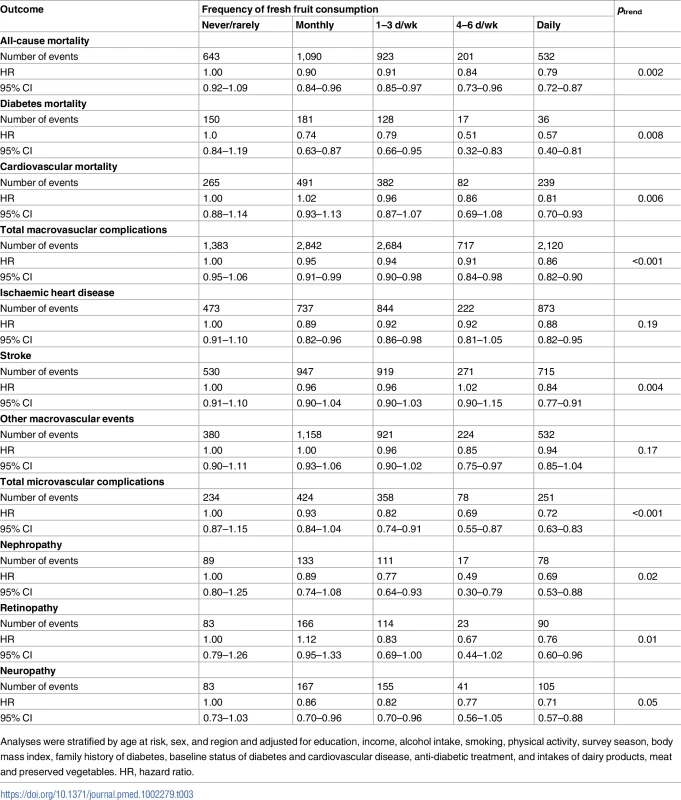
Analyses were stratified by age at risk, sex, and region and adjusted for education, income, alcohol intake, smoking, physical activity, survey season, body mass index, family history of diabetes, baseline status of diabetes and cardiovascular disease, anti-diabetic treatment, and intakes of dairy products, meat and preserved vegetables. HR, hazard ratio. Discussion
This large prospective study of Chinese adults with and without diabetes showed that higher fresh fruit consumption was significantly associated with a lower risk of developing diabetes, and also with a lower risk of dying or developing vascular complications among those who have already developed diabetes. These associations appeared to be similar in both men and women, in urban and rural residents, and in those with previously diagnosed and screen-detected diabetes. Moreover, higher fresh fruit consumption was not associated with elevated level of blood glucose.
Several prospective studies have previously assessed the association of fruit consumption with risk of diabetes, showing inconsistent findings [6,7,9,26–29]. For example, higher fruit consumption was significantly associated with diabetes incidence in the Nurses’ Health Study (with 6,358 cases) and a small Finnish study with 383 cases [6,30], with HRs of 0.82 (95% CI 0.72–0.92) and 0.69 (0.50–0.93), respectively, when comparing the highest with the lowest fruit consumption category. No significant association, however, was observed in the European Prospective Investigation into Cancer and Nutrition (EPIC)–InterAct study with nearly 11,000 incident diabetes cases [9] or a cohort study of Chinese women with approximately 1,600 new diabetes cases [8]. In the most recent meta-analysis of >400,000 participants from 11 studies with nearly 34,000 incident diabetes cases, higher fruit consumption was associated with a 9% (95% CI 4%–13%) lower risk of diabetes incidence [27]. These previous studies were conducted primarily among Western populations and tended to combine fresh fruit with processed fruit (sometimes including also fruit juice), in contrast to focusing only on fresh fruit, as in our study. This may partly explain the much stronger linear association observed in our study. In addition, the stronger association we observed might also be due to the very low level of fruit consumption among Chinese people and a non-linear dose–response association between fruit intake and diabetes risk [7,29]. Furthermore, our study has taken into account regression dilution bias when estimating the linear associations [24,25] while few previous large prospective studies have dealt with this important issue [31].
To date, very few large prospective studies have assessed the long-term health effects of fruit consumption in people with diabetes, even though fruit has been rather consistently associated with lower risk of CVD [20], a major complication of diabetes [19]. In two reports from the EPIC study, one involving approximately 6,000 [12] and the other involving 10,500 [11] individuals with self-reported diabetes, one portion of fruit per day was significantly associated with 12%–15% lower all-cause [12] and cardiovascular mortality [11]. Only one small study in Japan has reported the association of fruit consumption with diabetes-related microvascular complications. In that study of nearly 1,000 diabetic patients with 8 y of follow-up, individuals in the highest quartile of fruit consumption had approximately 50% (HR = 0.48 [95% CI 0.32–0.71]) lower risk of developing diabetic retinopathy (total 285 cases) [10]. Our study included a much larger number of participants and well-characterised disease outcomes, with new findings about the potential benefits of fresh fruit intake on a range of macro - and microvascular complications of diabetes. In addition, a separate analysis of data from more than 70,000 participants with existing CVD or hypertension in the China Kadoorie Biobank, with an overlap of about 10,000 participants with the current study, showed similar inverse associations of fresh fruit consumption with all-cause and cardiovascular mortality [32].
This is the first cohort study to our knowledge reporting beneficial associations of fresh fruit consumption with both incidence of diabetes and development of diabetes complications. Given the large sample size, our study findings are statistically robust even though the causality of the association cannot be established from such observational studies. However, the study also has limitations. First, our dietary questionnaire was not validated against another reference method, and information on fruit types was not collected. However, the previously observed inverse associations of fruit consumption with blood pressure and risk of CVD [20] could indicate an appropriate predictive validity of this method in estimating fruit consumption [33]. Moreover, the baseline level and secular trend of fruit consumption observed in our study population were in line with the findings from nationally representative nutrition surveys that used three consecutive 24-h recalls to collect habitual intake [20]. Fruits with relatively lower glycaemic index (i.e., apples, oranges, pears, and berries) may have larger beneficial effects on diabetes than those with a higher glycaemic index (i.e., bananas, grapes, and tropical fruits) [34], although previous studies have found both higher and lower glycaemic index fruits to be associated with lower risk of diabetes [6]. Based on data from the China Health and Nutrition Survey, the most frequently consumed fruits in China are apples, pears, and oranges, which are temperate climate/low glycaemic index fruit. Second, our baseline dietary data included consumption frequency only (not amount); therefore, the linear associations (i.e., HRs per 100 g/d) were estimated based on assumptions (assuming daily portions of fruit consumption did not change from baseline to second resurvey). Also, our analyses could not be adjusted for total energy or specific nutrient intake, and our data do not allow a reliable assessment of how much fruit per day is too much. Other dietary factors that may have an important association with diabetes, e.g., sugar-sweetened beverages [35], could not be adjusted for due to lack of information. This, however, should not confound our findings because the average sugar-sweetened beverage consumption level in our population was very low [36]. Third, no information on vascular diseases other than IHD and stroke was collected at baseline; thus, some cases of diabetes complications might be recurrent instead of new onset. This again should not invalidate our findings, given the very similar associations observed in individuals with newly detected diabetes (who should be less likely to have diabetes complications at baseline) and previously diagnosed diabetes. A proportion of diabetes cases might not have been detected at baseline because we did not use post-load blood glucose and HbA1c tests at baseline. Such misclassification should be non-differential (i.e., the proportion of undiagnosed diabetes cases should not be related to the level of fresh fruit consumption) and therefore should not invalidate or overestimate the observed associations of fruit consumption with diabetes incidence and vascular complications. Finally, although we carefully adjusted for potential confounders and there were consistent results across different participant subgroups, residual confounding (e.g., by socio-economic status) may still persist. In other words, it is not possible to determine reliably from this study whether the somewhat lower risks of diabetes incidence and diabetes complications observed among those with higher fruit consumption were caused by fruit consumption per se or were mainly due to other factors.
The exact mechanisms through which fresh fruit consumption may be protective against the development and deterioration of diabetes are not very well understood. Fruit contains sugars (i.e., glucose and fructose), which may have negative impacts on glycaemic control [37]. However, the natural sugars in fruit may not be metabolised in the same way as refined sugars [38]. In our study, fruit consumption had a weak inverse, instead of positive, association with levels of blood glucose, overall and in those with previously diagnosed diabetes. This is largely consistent with previous findings showing that fresh fruit consumption had no significant negative impact on glycaemic control, even in people with diabetes [5,34,39,40]. More importantly, fruit is a good source of dietary fibre [41,42], minerals (e.g., potassium [43]), and antioxidants (e.g., vitamins [44] and polyphenols [45]), which may work synergistically to confer several benefits on metabolism—including anti-oxidative, anti-inflammatory, anti-proliferative, anti-platelet, anti-hypertensive, anti-dyslipidaemic, anti-hyperglycaemic, and anti-atherogenic effects—and modulation of the composition and metabolic activity of gut microbiota [46–48], which could reduce the risk of diabetes as well as of vascular complications among those who have already developed diabetes [46,49].
In summary, our study demonstrated that, among Chinese adults, higher fresh fruit consumption was associated with lower risk of diabetes and diabetic vascular complications. Contrary to the common belief in China and many other low - and middle-income countries, fresh fruit consumption was not associated with an elevated blood glucose level in the present study, even in people with diabetes. These findings have public health and clinical implications and provide strong evidence in support of current dietary guidelines that fresh fruit consumption should be recommended for all, including those with diabetes [50]. In many developed countries, diabetes patients usually have higher fruit consumption than individuals without diabetes due to targeted health promotion and nutrition education [12,51]. However, in China people with previously diagnosed diabetes have a much lower level of fruit consumption, as observed in the present study, because of the incorrect belief that diabetes, or “sugar urine disease” in Chinese, will be better controlled if all sweet-tasting (or sugar-containing) foods, including fresh fruit, are restricted or avoided [5,52–54]. This situation emphasises the importance, given the present study findings, of better health promotion to improve public understanding of the role of fresh fruit in diabetes prevention and management. Such actions are urgently needed in China and other Asian countries, where diabetes prevalence is high and where, at the same time, there is widespread misunderstanding about eating fresh fruit among people with diabetes [40,55].
Supporting Information
Zdroje
1. International Diabetes Federation (2015) IDF diabetes atlas. 7th ed. Brussels: International Diabetes Federation.
2. Dyson PA, Kelly T, Deakin T, Duncan A, Frost G, Harrison Z, et al. (2011) Diabetes UK evidence-based nutrition guidelines for the prevention and management of diabetes. Diabet Med 28 : 1282–1288. doi: 10.1111/j.1464-5491.2011.03371.x 21699560
3. Bantle JP, Wylie-Rosett J, Albright AL, Apovian CM, Clark NG, Franz MJ, et al. (2008) Nutrition recommendations and interventions for diabetes: a position statement of the American Diabetes Association. Diabetes Care 31 (Suppl 1): S61–S78.
4. National Institute for Health and Care Excellence (2015) Type 2 diabetes in adults: management. NICE guideline NG28. UK: National Institute for Health and Care Excellence.
5. Christensen AS, Viggers L, Hasselstrom K, Gregersen S (2013) Effect of fruit restriction on glycemic control in patients with type 2 diabetes—a randomized trial. Nutr J 12 : 29. doi: 10.1186/1475-2891-12-29 23497350
6. Muraki I, Imamura F, Manson JE, Hu FB, Willett WC, van Dam RM, et al. (2013) Fruit consumption and risk of type 2 diabetes: results from three prospective longitudinal cohort studies. BMJ 347: f5001. doi: 10.1136/bmj.f5001 23990623
7. Li S, Miao S, Huang Y, Liu Z, Tian H, Yin X, et al. (2015) Fruit intake decreases risk of incident type 2 diabetes: an updated meta-analysis. Endocrine 48 : 454–460. doi: 10.1007/s12020-014-0351-6 25074631
8. Villegas R, Shu XO, Gao YT, Yang G, Elasy T, Li H, et al. (2008) Vegetable but not fruit consumption reduces the risk of type 2 diabetes in Chinese women. J Nutr 138 : 574–580. 18287369
9. Cooper AJ, Forouhi NG, Ye Z, Buijsse B, Arriola L, Balkau B, et al. (2012) Fruit and vegetable intake and type 2 diabetes: EPIC-InterAct prospective study and meta-analysis. Eur J Clin Nutr 66 : 1082–1092. doi: 10.1038/ejcn.2012.85 22854878
10. Tanaka S, Yoshimura Y, Kawasaki R, Kamada C, Horikawa C, Ohashi Y, et al. (2013) Fruit intake and incident diabetic retinopathy with type 2 diabetes. Epidemiology 24 : 204–211. doi: 10.1097/EDE.0b013e318281725e 23348071
11. Nothlings U, Schulze MB, Weikert C, Boeing H, van der Schouw YT, Bamia C, et al. (2008) Intake of vegetables, legumes, and fruit, and risk for all-cause, cardiovascular, and cancer mortality in a European diabetic population. J Nutr 138 : 775–781. 18356334
12. Sluik D, Boeing H, Li K, Kaaks R, Johnsen NF, Tjonneland A, et al. (2014) Lifestyle factors and mortality risk in individuals with diabetes mellitus: are the associations different from those in individuals without diabetes? Diabetologia 57 : 63–72. doi: 10.1007/s00125-013-3074-y 24132780
13. Sircar AR, Sircar S, Sircar J, Misra S (2010) Patients’ concepts and attitudes about diabetes. J Diabetes Complications 24 : 398–403. doi: 10.1016/j.jdiacomp.2009.07.004 19748288
14. Wang CY, Chan SM (2005) Culturally tailored diabetes education program for Chinese Americans: a pilot study. Nurs Res 54 : 347–353. 16224321
15. Choudhury SM, Brophy S, Williams R (2009) Understanding and beliefs of diabetes in the UK Bangladeshi population. Diabet Med 26 : 636–640. doi: 10.1111/j.1464-5491.2009.02741.x 19538240
16. Chen Z, Chen J, Collins R, Guo Y, Peto R, Wu F, et al. (2011) China Kadoorie Biobank of 0.5 million people: survey methods, baseline characteristics and long-term follow-up. Int J Epidemiol 40 : 1652–1666. doi: 10.1093/ije/dyr120 22158673
17. Chen Z, Lee L, Chen J, Collins R, Wu F, Guo Y, et al. (2005) Cohort profile: the Kadoorie Study of Chronic Disease in China (KSCDC). Int J Epidemiol 34 : 1243–1249. doi: 10.1093/ije/dyi174 16131516
18. Du H, Bennett D, Li L, Whitlock G, Guo Y, Collins R, et al. (2013) Physical activity and sedentary leisure time and their associations with BMI, waist circumference, and percentage body fat in 0.5 million adults: the China Kadoorie Biobank study. Am J Clin Nutr 97 : 487–496. doi: 10.3945/ajcn.112.046854 23364014
19. Bragg F, Li L, Yang L, Guo Y, Chen Y, Bian Z, et al. (2016) Risks and population burden of cardiovascular diseases associated with diabetes in China: a prospective study of 0.5 million adults. PLoS Med 13: e1002026. doi: 10.1371/journal.pmed.1002026 27379518
20. Du H, Li L, Bennett D, Guo Y, Key TJ, Bian Z, et al. (2016) Fresh fruit consumption and major cardiovascular disease in China. N Engl J Med 374 : 1332–1343. doi: 10.1056/NEJMoa1501451 27050205
21. Yang GH, Stroup DF, Thacker SB (1997) National public health surveillance in China: implications for public health in China and the United States. Biomed Environ Sci 10 : 1–13. 9099422
22. Plummer M (2004) Improved estimates of floating absolute risk. Stat Med 23 : 93–104. doi: 10.1002/sim.1485 14695642
23. Easton DF, Peto J, Babiker AG (1991) Floating absolute risk: an alternative to relative risk in survival and case-control analysis avoiding an arbitrary reference group. Stat Med 10 : 1025–1035. 1652152
24. Clarke R, Shipley M, Lewington S, Youngman L, Collins R, Marmot M, et al. (1999) Underestimation of risk associations due to regression dilution in long-term follow-up of prospective studies. Am J Epidemiol 150 : 341–353. 10453810
25. Lewington S, Thomsen T, Davidsen M, Sherliker P, Clarke R (2003) Regression dilution bias in blood total and high-density lipoprotein cholesterol and blood pressure in the Glostrup and Framingham prospective studies. J Cardiovasc Risk 10 : 143–148. 12668912
26. Hamer M, Chida Y (2007) Intake of fruit, vegetables, and antioxidants and risk of type 2 diabetes: systematic review and meta-analysis. J Hypertens 25 : 2361–2369. doi: 10.1097/HJH.0b013e3282efc214 17984654
27. Wang PY, Fang JC, Gao ZH, Zhang C, Xie SY (2016) Higher intake of fruits, vegetables or their fiber reduces the risk of type 2 diabetes: a meta-analysis. J Diabetes Investig 7 : 56–69.
28. Carter P, Gray LJ, Troughton J, Khunti K, Davies MJ (2010) Fruit and vegetable intake and incidence of type 2 diabetes mellitus: systematic review and meta-analysis. BMJ 341: c4229. doi: 10.1136/bmj.c4229 20724400
29. Wu Y, Zhang D, Jiang X, Jiang W (2015) Fruit and vegetable consumption and risk of type 2 diabetes mellitus: a dose–response meta-analysis of prospective cohort studies. Nutr Metab Cardiovasc Dis 25 : 140–147. doi: 10.1016/j.numecd.2014.10.004 25456152
30. Montonen J, Jarvinen R, Heliovaara M, Reunanen A, Aromaa A, Knekt P (2005) Food consumption and the incidence of type II diabetes mellitus. Eur J Clin Nutr 59 : 441–448. doi: 10.1038/sj.ejcn.1602094 15674312
31. Bhupathiraju SN, Wedick NM, Pan A, Manson JE, Rexrode KM, Willett WC, et al. (2013) Quantity and variety in fruit and vegetable intake and risk of coronary heart disease. Am J Clin Nutr 98 : 1514–1523. doi: 10.3945/ajcn.113.066381 24088718
32. Tian X, Du H, Li L, Bennett D, Gao R, Li S, et al. (2017) Fruit consumption and physical activity in relation to all-cause and cardiovascular mortality among 70,000 Chinese adults with pre-existing vascular disease. PLoS ONE 12(4): e0173054. doi: 10.1371/journal.pone.0173054
33. Kristal AR, Potter JD (2006) Not the time to abandon the food frequency questionnaire: counterpoint. Cancer Epidemiol Biomarkers Prev 15 : 1759–1760. doi: 10.1158/1055-9965.EPI-06-0727 17021349
34. Jenkins DJ, Srichaikul K, Kendall CW, Sievenpiper JL, Abdulnour S, Mirrahimi A, et al. (2011) The relation of low glycaemic index fruit consumption to glycaemic control and risk factors for coronary heart disease in type 2 diabetes. Diabetologia 54 : 271–279. doi: 10.1007/s00125-010-1927-1 20978741
35. Romaguera D, Norat T, Wark PA, Vergnaud AC, Schulze MB, van Woudenbergh GJ, et al. (2013) Consumption of sweet beverages and type 2 diabetes incidence in European adults: results from EPIC-InterAct. Diabetologia 56 : 1520–1530. doi: 10.1007/s00125-013-2899-8 23620057
36. Du H, Li L, Chen Z (2016) Fruit consumption and cardiovascular disease in China. N Engl J Med 375 : 487–489. doi: 10.1056/NEJMc1606624#SA1 27518674
37. Lustig RH, Schmidt LA, Brindis CD (2012) Public health: the toxic truth about sugar. Nature 482 : 27–29. doi: 10.1038/482027a 22297952
38. Sievenpiper JL, de Souza RJ, Jenkins DJ (2012) Sugar: fruit fructose is still healthy. Nature 482 : 470.
39. Wang B, Liu K, Mi M, Wang J (2014) Effect of fruit juice on glucose control and insulin sensitivity in adults: a meta-analysis of 12 randomized controlled trials. PLoS ONE 9: e95323. doi: 10.1371/journal.pone.0095323 24743260
40. Hegde SV, Adhikari P, M N, D’Souza V (2013) Effect of daily supplementation of fruits on oxidative stress indices and glycaemic status in type 2 diabetes mellitus. Complement Ther Clin Pract 19 : 97–100. doi: 10.1016/j.ctcp.2012.12.002 23561067
41. Silva FM, Kramer CK, de Almeida JC, Steemburgo T, Gross JL, Azevedo MJ (2013) Fiber intake and glycemic control in patients with type 2 diabetes mellitus: a systematic review with meta-analysis of randomized controlled trials. Nutr Rev 71 : 790–801. doi: 10.1111/nure.12076 24180564
42. Weickert MO, Pfeiffer AF (2008) Metabolic effects of dietary fiber consumption and prevention of diabetes. J Nutr 138 : 439–442. 18287346
43. Chatterjee R, Yeh HC, Edelman D, Brancati F (2011) Potassium and risk of Type 2 diabetes. Expert Rev Endocrinol Metab 6 : 665–672. doi: 10.1586/eem.11.60 22025927
44. Harding AH, Wareham NJ, Bingham SA, Khaw K, Luben R, Welch A, et al. (2008) Plasma vitamin C level, fruit and vegetable consumption, and the risk of new-onset type 2 diabetes mellitus: the European prospective investigation of cancer—Norfolk prospective study. Arch Intern Med 168 : 1493–1499. doi: 10.1001/archinte.168.14.1493 18663161
45. Kim Y, Keogh JB, Clifton PM (2016) Polyphenols and glycemic control. Nutrients 8: E17. doi: 10.3390/nu8010017 26742071
46. Bahadoran Z, Mirmiran P, Azizi F (2013) Dietary polyphenols as potential nutraceuticals in management of diabetes: a review. J Diabetes Metab Disord 12 : 43. doi: 10.1186/2251-6581-12-43 23938049
47. Chan HT, Yiu KH, Wong CY, Li SW, Tam S, Tse HF (2013) Increased dietary fruit intake was associated with lower burden of carotid atherosclerosis in Chinese patients with type 2 diabetes mellitus. Diabet Med 30 : 100–108. doi: 10.1111/j.1464-5491.2012.03764.x 22913415
48. Marchesi JR, Adams DH, Fava F, Hermes GD, Hirschfield GM, Hold G, et al. (2016) The gut microbiota and host health: a new clinical frontier. Gut 65 : 330–339. doi: 10.1136/gutjnl-2015-309990 26338727
49. Xu YJ, Tappia PS, Neki NS, Dhalla NS (2014) Prevention of diabetes-induced cardiovascular complications upon treatment with antioxidants. Heart Fail Rev 19 : 113–121. doi: 10.1007/s10741-013-9379-6 23436032
50. American Diabetes Association (2015) Standards of medical care in diabetes—2015. Diabetes Care 38: S1–S90.
51. Virtanen SM, Feskens EJ, Rasanen L, Fidanza F, Tuomilehto J, Giampaoli S, et al. (2000) Comparison of diets of diabetic and non-diabetic elderly men in Finland, The Netherlands and Italy. Eur J Clin Nutr 54 : 181–186. 10713738
52. Rodriguez MC, Parra MD, Marques-Lopes I, De Morentin BE, Gonzalez A, Martinez JA (2005) Effects of two energy-restricted diets containing different fruit amounts on body weight loss and macronutrient oxidation. Plant Foods Hum Nutr 60 : 219–224. doi: 10.1007/s11130-005-8622-2 16395633
53. Sargeant LA, Khaw KT, Bingham S, Day NE, Luben RN, Oakes S, et al. (2001) Fruit and vegetable intake and population glycosylated haemoglobin levels: the EPIC-Norfolk Study. Eur J Clin Nutr 55 : 342–348. doi: 10.1038/sj.ejcn.1601162 11378807
54. Carter P, Gray LJ, Talbot D, Morris DH, Khunti K, Davies MJ (2013) Fruit and vegetable intake and the association with glucose parameters: a cross-sectional analysis of the Let’s Prevent Diabetes Study. Eur J Clin Nutr 67 : 12–17. doi: 10.1038/ejcn.2012.174 23299789
55. Colles SL, Singh S, Kohli C, Mithal A (2013) Dietary beliefs and eating patterns influence metabolic health in type 2 diabetes: a clinic-based study in urban North India. Indian J Endocrinol Metab 17 : 1066–1072. doi: 10.4103/2230-8210.122626 24381886
Štítky
Interní lékařství
Článek vyšel v časopisePLOS Medicine
Nejčtenější tento týden
2017 Číslo 4- Alternativní léčebné možnosti u hypercholesterolemie při intoleranci statinů
- Vliv kombinace nutraceutik na remodelaci levé komory srdeční u osob s metabolickým syndromem
- Nutraceutika a jejich ovlivnění mírného kardiometabolického rizika
- Princip účinku medu v léčbě chronických i infikovaných ran
- Superoxidovaný roztok a jeho využití v léčbě ran
-
Všechny články tohoto čísla
- Implementation science: Relevance in the real world without sacrificing rigor
- Clinical decision tools are needed to identify HIV-positive patients at high risk for poor outcomes after initiation of antiretroviral therapy
- Silk garments plus standard care compared with standard care for treating eczema in children: A randomised, controlled, observer-blind, pragmatic trial (CLOTHES Trial)
- An open source pharma roadmap
- A new cascade of HIV care for the era of “treat all”
- Status and methodology of publicly available national HIV care continua and 90-90-90 targets: A systematic review
- Talking sensibly about depression
- Governing multisectoral action for health in low- and middle-income countries
- Effectiveness of a live oral human rotavirus vaccine after programmatic introduction in Bangladesh: A cluster-randomized trial
- Community-based strategies to strengthen men’s engagement in the HIV care cascade in sub-Saharan Africa
- Graft-derived cell-free DNA, a noninvasive early rejection and graft damage marker in liver transplantation: A prospective, observational, multicenter cohort study
- Risk prediction models for selection of lung cancer screening candidates: A retrospective validation study
- Fresh fruit consumption in relation to incident diabetes and diabetic vascular complications: A 7-y prospective study of 0.5 million Chinese adults
- The effects of implementing a point-of-care electronic template to prompt routine anxiety and depression screening in patients consulting for osteoarthritis (the Primary Care Osteoarthritis Trial): A cluster randomised trial in primary care
- Identification of genes associated with dissociation of cognitive performance and neuropathological burden: Multistep analysis of genetic, epigenetic, and transcriptional data
- Demographic transition and the dynamics of measles in six provinces in China: A modeling study
- Changes in prices, sales, consumer spending, and beverage consumption one year after a tax on sugar-sweetened beverages in Berkeley, California, US: A before-and-after study
- PLOS Medicine
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Silk garments plus standard care compared with standard care for treating eczema in children: A randomised, controlled, observer-blind, pragmatic trial (CLOTHES Trial)
- Talking sensibly about depression
- A new cascade of HIV care for the era of “treat all”
- Fresh fruit consumption in relation to incident diabetes and diabetic vascular complications: A 7-y prospective study of 0.5 million Chinese adults
Přihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



