-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Kariérní portál
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaAssociation between expansion of primary healthcare and racial inequalities in mortality amenable to primary care in Brazil: A national longitudinal analysis
In a national longitudinal study, Thomas Hone and colleagues show that the expansion of primary healthcare in Brazil was associated with improved health outcomes and reductions in health inequalities between racial groups.
Published in the journal: . PLoS Med 14(5): e32767. doi:10.1371/journal.pmed.1002306
Category: Research Article
doi: https://doi.org/10.1371/journal.pmed.1002306Summary
In a national longitudinal study, Thomas Hone and colleagues show that the expansion of primary healthcare in Brazil was associated with improved health outcomes and reductions in health inequalities between racial groups.
Introduction
Reducing inequalities within and among countries is the tenth goal of the Sustainable Development Goals (SDGs). This goal includes the target to “adopt policies, especially fiscal, wage and social protection policies” that “progressively achieve greater equality” (http://www.un.org/sustainabledevelopment/inequality/). Health systems are essential for social protection and, in addition to their contributions to other SDGs for health, may play a vital role in reducing inequalities [1]. Additionally, promoting equality in access to healthcare is a core principle of universal health coverage (UHC) [2]. Investment in primary healthcare (PHC), as part of efforts to achieve UHC, may be especially important in reducing health inequalities [3–5], but evidence is largely derived from North America and Europe.
Brazil is an important setting for evaluating the relationship of PHC with health inequalities. It is a middle-income country with one of the highest levels of income inequality globally (a Gini coefficient of 52.9 in 2013 [6]) and stark health inequalities across income, education, racial, and socio-economic groups [7–13]. Brazil’s considerable investments in social protection policies over the last two decades include the rollout of conditional cash transfers under the Bolsa Família programme and a commitment to UHC with the expansion of PHC through the Estratégia de Saúde da Família (ESF) (family health strategy) [14,15]. The ESF has rapidly expanded since the mid-1990s to become the largest community-based PHC program in the world [16]. In 2014, it covered ~121.2 million individuals (~62.5% of the population) [17]. Family health teams composed of a family doctor, nurses, and community health workers deliver a broad range of comprehensive and preventive healthcare services to defined local populations (approximately 3,400 individuals) [15]. Municipal governments are responsible for the provision of local ESF services, and financial incentives provided by the federal government encourage municipalities to adopt the ESF [18]. In general, municipalities with smaller populations, higher levels of poverty, and a higher proportion of residents from black/pardo (mixed race) racial groups exhibited greater uptake of the ESF (S1 Appendix, Figs. A–C) [19]. Expansion of the ESF has been associated with reductions in infant mortality [20–22], deaths from cardiovascular disease [4], and hospitalisations from ambulatory-care-sensitive conditions (ACSCs) [5], but there is little understanding of the associations between ESF expansion and changes in health inequalities. Recent financial and political crises in Brazil are threatening funding for social protection policies, including UHC [23]. Evidence of an association between the ESF and a reduction of inequalities in health outcomes would provide a strong argument for continued investment and political support.
Assessing racial inequalities is important for evaluating the ESF, given the complex historical, sociological, and political dimensions of race in Brazil [24,25]. In contrast to ancestral and ethnic classifications of race in the US and the UK [13], institutions in Brazil use skin colour. Official classifications are branco (white), preto (black), pardo (brown/mixed), amarello (Asian), and indigenous, with white, black, and pardo accounting for over 98% of the population. Self-reported classification, whilst reflecting ancestral and cultural roots, also reflects an individual’s perceived social identity [11,13,25]. Three main ancestral roots established the Brazilian population today—indigenous individuals, European colonisers, and African slaves [25]. Today, there is considerable admixture (evidenced by a sizeable pardo population), but sharp inequalities between racial groups persist [9–13]. Black and pardo populations have higher illiteracy, have lower average incomes, and use healthcare services less [9]. In health outcomes, they have lower life expectancy, are affected more by infectious diseases (including tuberculosis, leprosy, leishmaniasis, and schistosomiasis), and have higher mortality rates from external causes, drug overdoses, and homicides [9].
Few studies have examined the potential role of PHC in reducing health inequalities in low - and middle-income countries. This study seeks to address this important gap by examining associations between ESF coverage and mortality from ACSCs in white and black/pardo populations in Brazil. We test the hypothesis that expansion of PHC coverage through the ESF in Brazil is associated with reduced inequalities in mortality between racial groups [26].
Methods
Longitudinal (panel data) regression models were employed using routinely collected municipal-level data, which have been widely applied to evaluate the ESF previously [4,20,22,27–30]. These models estimated associations between ESF coverage and mortality from ACSCs among black/pardo and white populations over time, whilst controlling for other confounding factors. The main analysis was restricted to 1,622 municipalities based on previously assessed quality of vital statistics reporting to reduce bias from under-reporting of deaths [31]. Differences in our analytic approach from previous ESF evaluations were necessary to examine associations of ESF expansion and inequalities in mortality between racial groups. These were agreed before compilation and analysis of the data (which commenced in February 2016), and are set out in detail below. In response to reviewers’ suggestions after initial submission, we explored factors associated with ESF uptake, tested for pre-existing trends, tested for biases from ill-defined death adjustments, explored interactions with Bolsa Família, and conducted sensitivity analyses with alternative model specifications and, for comparison with ACSC mortality, on mortality from accidents.
Data sources
Data from individual death certificates for the years 2000–2013 were obtained from the Brazilian Ministry of Health DATASUS website [32]. Annual municipal population estimates by race and age group based on census data were obtained from the Instituto Brasileiro de Geografia e Estatística (IBGE) website [33]. Municipal-level covariate data, including illiteracy rate, poverty rate, urbanisation rate, and municipal gross domestic product (GDP), were obtained from the IBGE website [33]. Municipal ESF coverage, Bolsa Família coverage, public healthcare spending, the number of public hospital beds, the number of private hospital beds, and private health insurance coverage were obtained from the DATASUS website [32].
Variables
The mortality rate from ACSCs was the main outcome variable. ACSC deaths were encoded based on a list published by the Brazilian Ministry of Health (and restricted to those aged under 70 y) and ICD-10 codes reported on death certificates (Table 1) [34]. ACSCs were grouped by cause of death into infectious diseases, nutritional deficiencies and anaemia, chronic obstructive pulmonary disease (COPD) and asthma, cardiovascular disease, diabetes, epilepsy, and gastric ulcers. Redistribution of ill-defined deaths was performed using a published and previously utilised methodology to control for confounding trends from reductions in ill-defined deaths over time (S1 Text) [35].
Tab. 1. Ambulatory-care-sensitive conditions with International Classification of Diseases (ICD-10) codes. 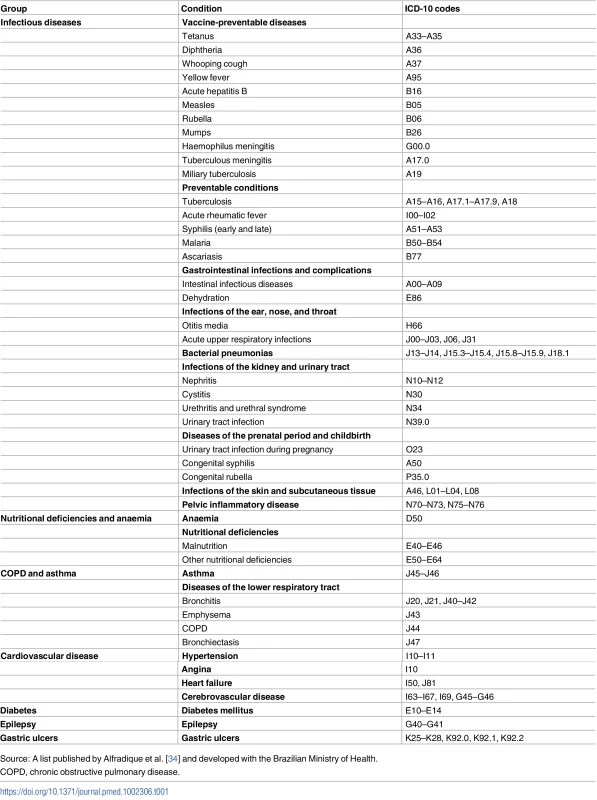
Source: A list published by Alfradique et al. [34] and developed with the Brazilian Ministry of Health. Race is recorded on death certificates and as part of the decennial census in Brazil. Census recording of race is self-reported. Individuals select branco (white), preto (black), pardo (brown/mixed), amarello (Asian), or indigenous. Recording of race on death certificates (using the same categories) is usually completed by the physician certifying the death and should be based on input from the family [13]. Amarello and indigenous deaths were very few and not examined. Black and pardo deaths were merged into one group, despite issues regarding differences between these populations [13]. This was to overcome potential differences in racial classification of individuals occurring either between censuses and death certificates, or over time as individuals and/or society changed reporting behaviour. Whilst evidence indicates overlap between black and pardo classifications in reporting of race, there are significantly clearer divisions between white and pardo classifications [36].
Reporting of race is near complete in censuses (99.29% in 2000 and 99.98% in 2010) and high on death certificates (total missing for 2000–2013 was 5.8%). For completeness, values were imputed for certificates with race missing using other death certificate variables (sex, age, education level, marital status, and location of death) and municipal population estimates of racial groups (S2 Text). For the period 2000–2013, race was imputed for 39,198 of the total 588,872 ACSC deaths (of those white or black/pardo and aged under 70 y) in the municipalities included in the analysis.
Using municipal census population data, population distributions by race (white and black/pardo) and age group (0–4, 5–9, 10–14, 15–19, 20–24, 25–29, 30–39, 40–49, 50–59, and 60–69 y) were calculated for each municipality for the census years (2000 and 2010), and were linearly interpolated and extrapolated for non-census years (2001–2009 and 2011–2013). Annual total municipal population estimates were used to calculate annual age and race group population estimates for each municipality. Direct age standardisation of cause of death by race was performed, producing annual age-adjusted mortality rates for total ACSCs and ACSC groups by race. The dependent variables (for each municipality and for each year) in the regression models were the expected (from age standardisation) number of deaths from ACSCs (in total and by ACSC group) for the black/pardo and white populations and the standardised rate ratio (SRR) between total black/pardo and white ACSC mortality rates. Rate ratios (RRs) are commonly used metrics for comparing rates between groups (e.g., between males and females) [37]. In this study, the ACSC mortality rate for the black/pardo population was divided by the ACSC mortality rate for white population.
The main variable of interest was municipal ESF coverage (percent) of the population, with official calculations based on one ESF team per 3,450 individuals [17]. A 2-y average (within the year and the year prior) of ESF coverage was employed, even though comparable results were obtained with just within-year coverage or including 2 - and 3-y lags. This approach was used to account for varying lagged and duration effects of the ESF that may differ between conditions and populations, to account for the time for ESF services to become fully operational and effective, and to permit simple comparison between the two racial groups.
Annual municipality-level covariate data were selected to include variables relating to socio-economic development, income, and the health system, which have been shown to affect mortality [38,39]. The covariates were scaled as percentages, in hundreds of Brazilian reais (R$100s) per person (adjusted for inflation), or per 1,000 inhabitants. Variables expressed as percentages were scaled between 0 and 1 so a one-unit increase would represent a 100% increase. Where necessary, logarithms were used to improve model fit. Covariates employed in all models were: Bolsa Família coverage (percent), illiteracy rate in those over 25 y (percent) (log-transformed), poverty rate (percent), population living in urban areas (percent), public healthcare spending (R$100s per person), public hospital beds per 1,000 population, private hospital beds per 1,000 population, private healthcare insurance (percent) (log-transformed), and GDP per person (R$100s per person) (log-transformed). An interaction between private healthcare insurance (percent) (log-transformed) and GDP per person (R$100s per person) (log-transformed) was included for model fit.
Statistical analysis
Descriptive analyses were undertaken, including national trends of ACSC mortality rates for black/pardo and white populations and the national SRR of the two rates.
Fixed-effects longitudinal regression was employed as an appropriate method for analysing annual observations of municipalities [40]. Fixed-effects models control for time-invariant unobserved factors that may affect mortality and could bias the results [40]. Consequently, only changes within municipalities over time are estimated rather than differences between municipalities. We tested for pre-intervention trends (i.e., mortality rates prior to ESF adoption and expansion) to determine whether time-varying unobserved factors could bias the results. Examining trends in the years 2000–2003 (when many municipalities still had relatively low coverage) and employing dummy variables for the years prior to ESF adoption revealed no evidence of pre-intervention trends.
In the models with dependent count variables (ACSC deaths), a Poisson model with a population offset term was employed, allowing the dependent variable (ACSC deaths) to be modelled as a rate (deaths per population). To aid interpretability, the coefficients were exponentiated and reported as RRs. These are interpreted as a ratio of the mortality rates for a one-unit increase in the independent variable (e.g., a 100% increase in ESF coverage or an additional year during the study period) (see S3 Text for more details). In other words, the difference between 1 and the RR can be interpreted as the percentage change in the rate given a one-unit increase in the independent variable. For the SRR, linear longitudinal regression was employed, and β coefficients reported. These are interpreted as the change in the SRR given a one-unit increase (i.e., from 0% to 100% coverage).
Two multiple regression models were undertaken examining the association between ESF expansion and ACSC mortality in the black/pardo and white populations separately. Differences in the effect sizes were tested for statistical significance (S4 Text). The p-values for the differences between the coefficients from the two models are reported in the text. The association between ESF coverage and the SRR was examined with a multiple regression model. Several regression models for the groups of ACSCs (infectious diseases, nutritional deficiencies and anaemia, COPD and asthma, cardiovascular disease, diabetes, epilepsy, and gastric ulcers) were employed in the black/pardo and white populations separately. Small numbers prohibited the use of SRR for groups of ACSCs. In all models, municipality-clustered robust standard errors were employed to account for possible auto-correlation and heteroscedasticity [40]. Stata 12 was used for statistical analysis.
Sensitivity analyses
Multiple sensitivity analyses were undertaken to check the robustness of the findings. First, alternative model specifications with sequential addition of covariates, random-effect models, and negative-binomial models were employed (S2 Appendix, Tables A and B). Second, varying classifications of ESF coverage were tested (S2 Table). Third, mortality from accidents (ICD-10 V01–X59) was tested, as an outcome that should have no association with ESF expansion (S3 Table). Fourth, the validity of imputing race on death certificates with race missing was assessed by excluding deaths where race was not recorded (S3 Appendix, Tables A and B). Fifth, the validity of redistributing ill-defined causes of death was tested (S4 Appendix, Tables A and B). Sixth, the analyses were repeated using data from all 5,565 municipalities in Brazil, not just those with adequate recording of vital statistics (S5 Appendix, Tables A and B). Seventh, because the potential for misclassification of race on death certificates exists (between the white and black/pardo populations), the effect of reclassifying black/pardo deaths (which are higher) as white was examined (S6 Appendix, Tables A–H). Eighth, an interaction between Bolsa Família and ESF coverage was examined (S7 Appendix).
Results
Between 2000 and 2013, there were 281,877 and 318,030 deaths from ACSC causes in the black/pardo and white populations, respectively (after age standardisation). Age-standardised ACSC mortality rates fell 37.9%, from 93.3 to 57.9 per 100,000, in the black/pardo population and by 34.9%, from 75.7 to 49.2 per 100,000, in the white population (Fig 1; S7 Appendix). Mortality from ACSC causes in the black/pardo population was between 17% and 23% higher than in the white population during the study period. There was a sizeable expansion of the ESF over the period, both in terms of the number of municipalities adopting the ESF and the average municipal ESF coverage (Fig 2).
Fig. 1. Age-standardised mortality rates for ambulatory-care-sensitive conditions in black/pardo and white populations in Brazil (2000–2013). 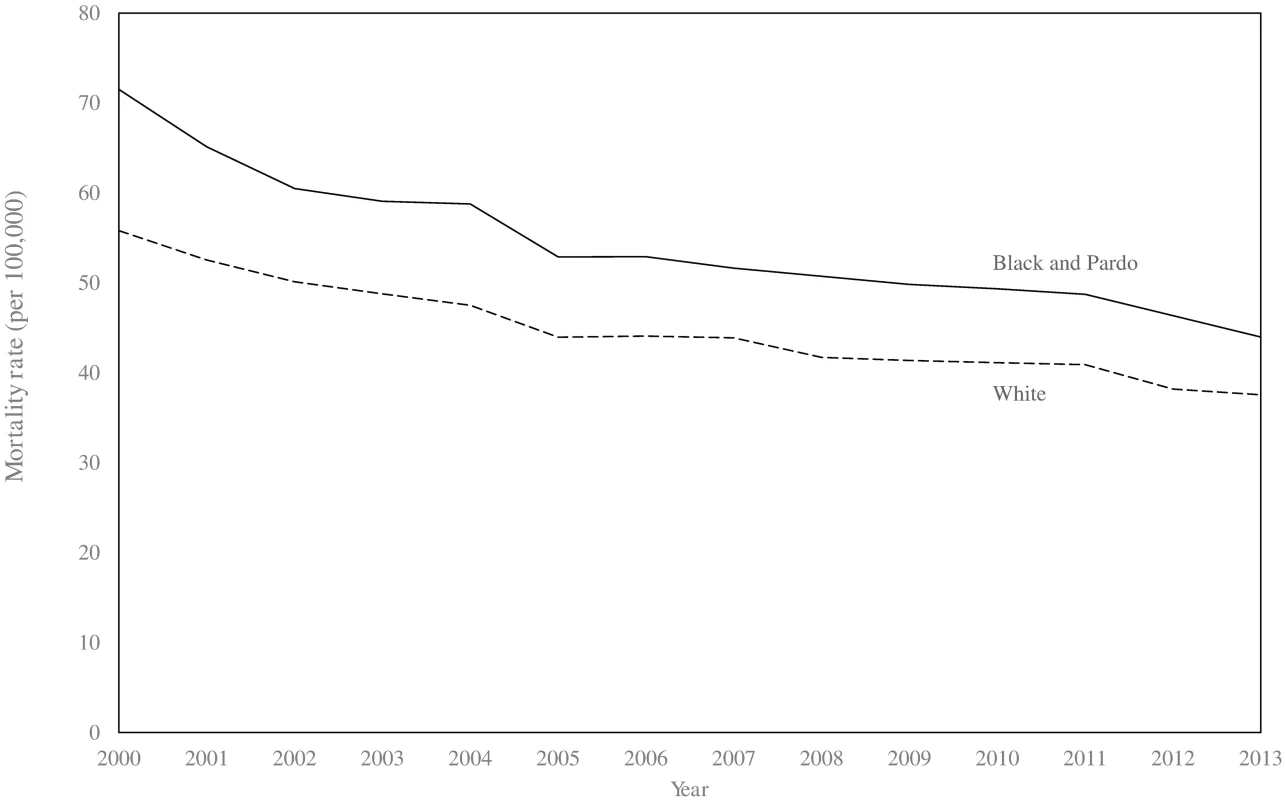
Data only from 1,622 municipalities with adequate reporting of vital statistics. Mortality rates are age standardised to the 2010 national population estimates. Fig. 2. Percentage of municipalities achieving any, 50%, and 100% Estratégia de Saúde da Família coverage (2000–2013). 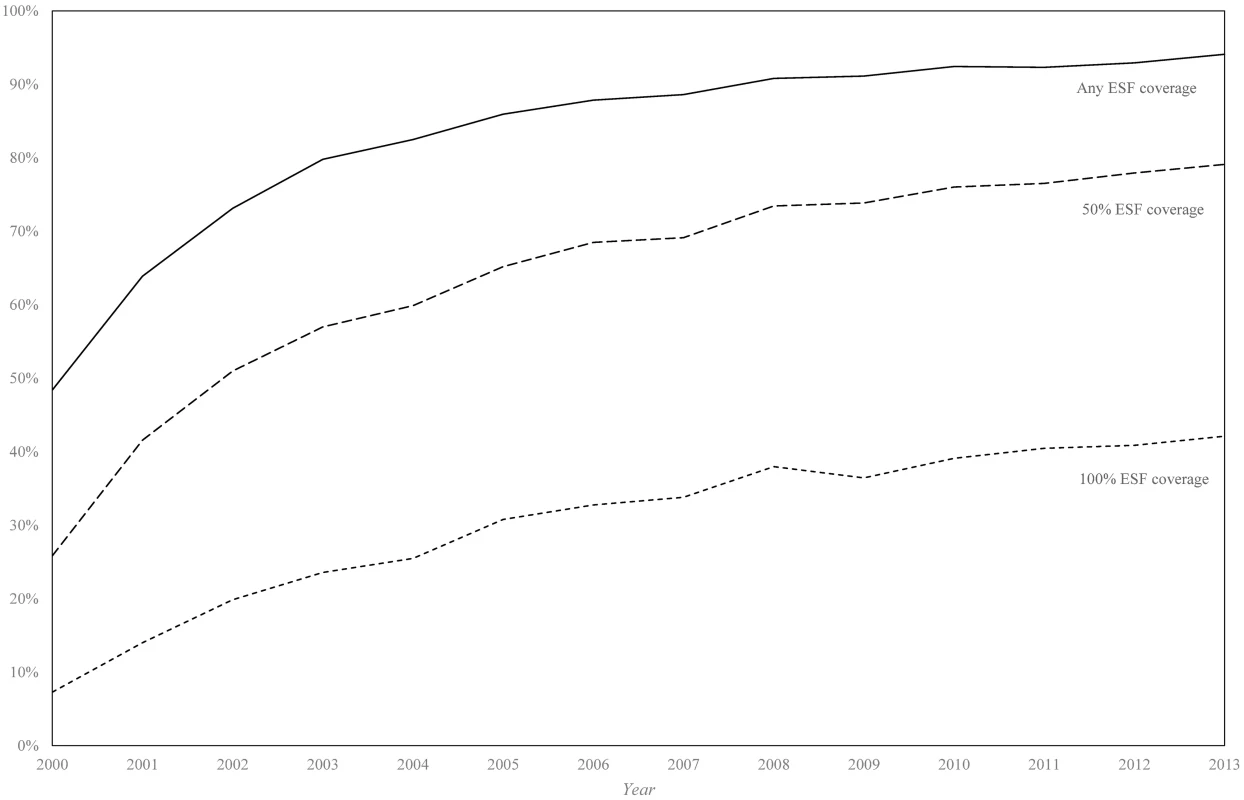
Only data from the 1,622 municipalities with adequate vital statistics reporting are included in this analysis. ESF, Estratégia de Saúde da Família. In longitudinal Poisson regression models, ACSC mortality decreased annually by 3.4% (RR: 0.966; 95% CI: 0.954–0.976) in the black/pardo population and by 2.9% (RR: 0.971; 95% CI: 0.963–0.979) in the white population in adjusted models (Table 2). ESF expansion (from 0% to 100% coverage) was associated with a 15.4% (RR: 0.846; 95% CI: 0.796–0.899) reduction in ACSC mortality in the black/pardo population and a 6.8% (RR: 0.932; 95% CI: 0.892–0.974) reduction in the white population. These coefficients were significantly different (p = 0.012).
Tab. 2. Results from longitudinal fixed-effects Poisson regression of mortality from ambulatory-care-sensitive conditions in black/pardo and white populations. 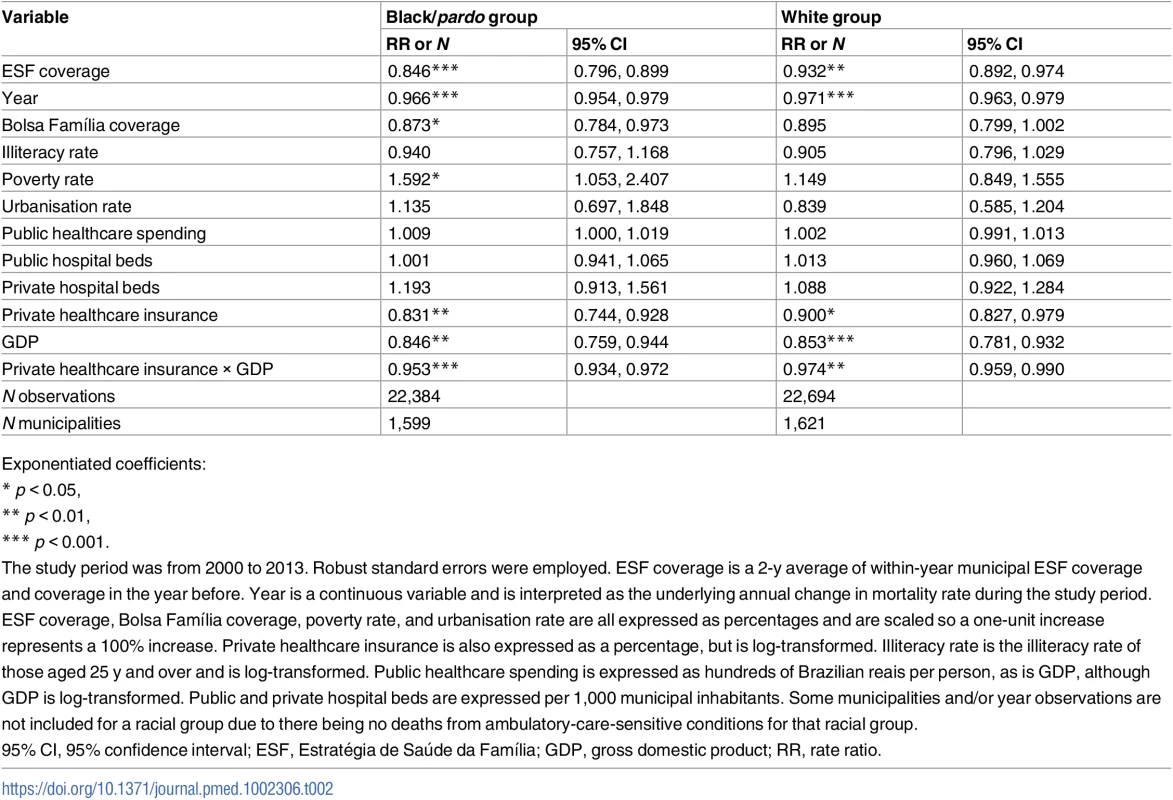
Exponentiated coefficients: ESF expansion (from 0% to 100% coverage) was associated with a 0.179 reduction (95% CI: 0.022–0.336) in the SRR (Table 3). Predicted SRRs from the model demonstrate that if ESF coverage were 0% in all municipalities, mortality amenable to PHC in the black/pardo population would be 29.6% higher than that in the white population (an estimated SRR of 1.296). With 100% ESF coverage in all municipalities, mortality amenable to PHC in the black/pardo population would be 11.7% higher than that in the white population (an estimated SRR of 1.117). Thus, expansion of the ESF (from 0% to 100%) yields a 60.5% reduction in the excess mortality that the black/pardo population experiences over the white population.
Tab. 3. Results from the longitudinal fixed-effects linear regression of standardised rate ratios for mortality from ambulatory-care-sensitive conditions in black/pardo and white populations. 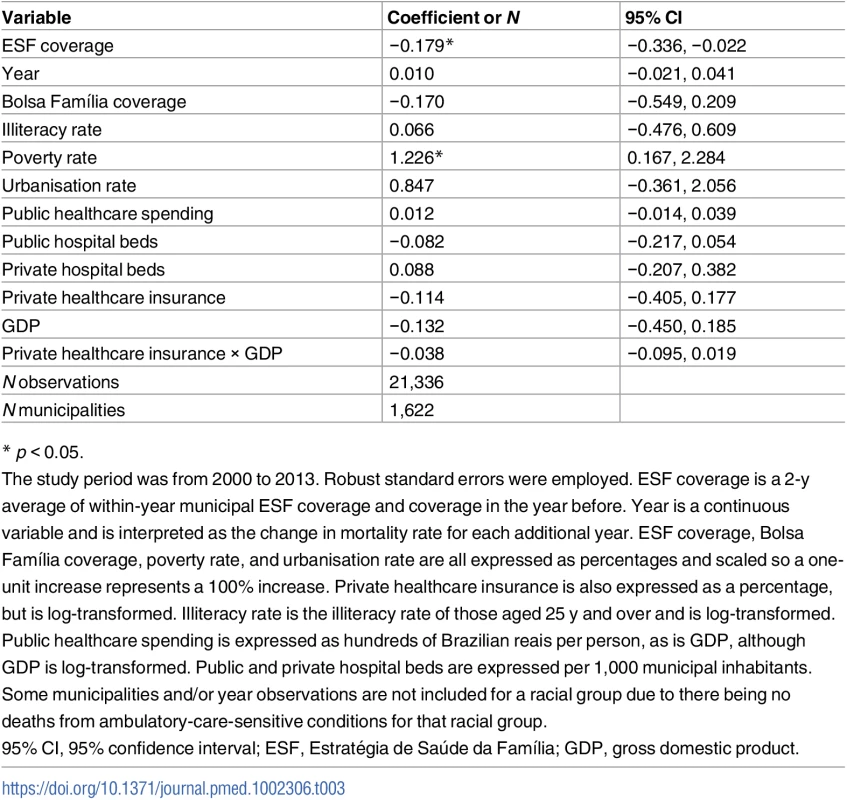
* p < 0.05. Associations with cause-specific mortality
Over the study period, mortality from COPD and asthma decreased annually by 4.1% (RR: 0.959; 95% CI: 0.933–0.985) in the black/pardo population and by 4.5% (RR: 0.955; 95% CI: 0.939–0.971) in the white population (Table 4). Mortality from cardiovascular disease decreased annually by 3.7% (RR: 0.963; 95% CI: 0.948–0.979) in the black/pardo population and by 2.7% (RR: 0.973; 95% CI: 0.962–0.984) in the white population. For the black/pardo population, mortality from diabetes decreased 2.7% per year (RR: 0.973; 95% CI: 0.952–0.994), whilst there were non-significant trends in infectious diseases, nutritional deficiencies and anaemia, epilepsy, and gastric ulcers. For the white population, mortality from infectious diseases decreased 2.8% annually (RR: 0.972; 95% CI: 0.948–0.997), mortality from nutritional deficiencies and anaemia decreased 4.9% annually (RR: 0.951; 95% CI: 0.909–0.994), and mortality from gastric ulcers decreased 4.9% annually (RR: 0.951; 95% CI: 0.922–0.981), but there were no significant trends in diabetes and epilepsy mortality.
Tab. 4. Results from longitudinal fixed-effects Poisson regressions for mortality by groups of ambulatory-care-sensitive conditions in black/pardo and white populations. 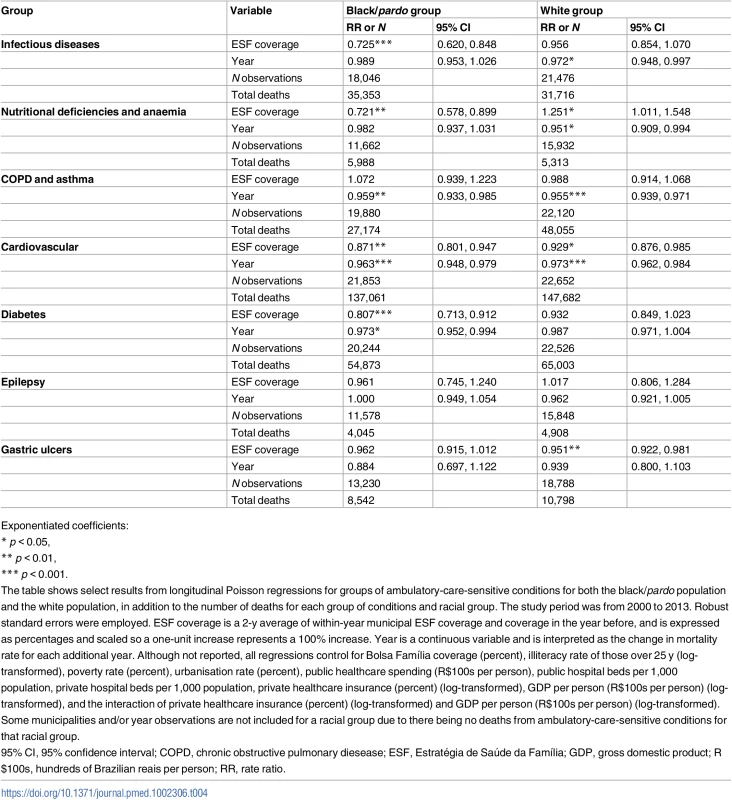
Exponentiated coefficients: ESF expansion (from 0% to 100%) was associated with a decrease in mortality from cardiovascular disease of 12.9% (RR: 0.871; 95% CI: 0.801–0.947) and 7.1% (RR: 0.929; 95% CI: 0.876–0.985) in the black/pardo and white populations, respectively. In the black/pardo population, ESF expansion was associated with 27.5% lower mortality from infectious diseases (RR: 0.725; 95% CI: 0.620–0.848) and 19.3% lower mortality from diabetes (RR: 0.807; 95% CI: 0.713–0.912), but there was no significant association with mortality for these ACSC groups in the white population. ESF expansion was associated with 17.9% lower mortality from nutritional deficiencies and anaemia (RR: 0.721; 95% CI: 0.478–0.899) in the black/pardo population, but in the white population, it was associated with 25.1% higher mortality (RR: 1.251; 95% CI: 1.011–1.548). For both the black/pardo and white populations, there was no significant association between ESF and mortality from COPD and asthma, epilepsy, or gastric ulcers.
Sensitivity analyses
Sensitivity analyses demonstrate the robustness of our findings. Alternative model specifications (S2 Appendix, Tables A and B) demonstrate the stability and robustness of the findings. We found that controlling for additional factors (fixed effects, covariates, and state-year fixed effects) did not change our findings; in fact, the differential associations of the ESF with black/pardo and white mortality became more apparent when these factors were taken into account. Alternative classifications of ESF coverage did not change the overall differences in the associations between ESF expansion and black/pardo and white mortality, although the results of the sensitivity analysis did suggest that greater reductions in mortality in the black/pardo population accrued over a longer period (S2 Table).
Examining mortality from accidents as a control outcome revealed no significant association of accident deaths with ESF coverage in either racial group, adding to the robustness of our findings (S3 Table). Excluding deaths with race not recorded yielded highly comparable results, demonstrating that imputation of missing race data was not a source of bias (S3 Appendix, Tables A and B). Repeating the analysis without adjustment for ill-defined deaths produced similar results (S4 Appendix, Tables A and B). An analysis with all 5,565 municipalities in Brazil (not just those with adequate reporting of vital statistics) found lower ACSC mortality associated with ESF expansion only in the white population yet a highly comparable association between ESF expansion and changes in the SRR (S5 Appendix, Tables A and B). The non-significance of the association of ACSC mortality with ESF expansion in the black/pardo population (when including municipalities with inadequate reporting of vital statistics) is expected given the likelihood of black/pardo deaths being under-reported and the role of the ESF in reducing under-reporting [41]. To examine the extent to which misclassification bias (i.e., deaths that were encoded as black/pardo when individuals self-identified as white in the census) could affect the results, we randomly reclassified 10%, 20%, and 30% of black/pardo deaths as white deaths (S6 Appendix, Tables A–H). A similar association between ESF expansion and race-specific mortality was found even when 30% of black/pardo deaths were reclassified, although associated reductions in inequality were lower. Lastly, an interaction between Bolsa Família and ESF coverage was non-significant (S7 Appendix, Tables A–C).
Discussion
Expansion of the ESF between 2000 and 2013 in Brazil was associated with a 2-fold greater reduction in ACSC mortality in the black/pardo compared to the white population. This differential benefit reduced racial inequalities in mortality and was driven by greater reductions in deaths from infectious diseases, nutritional deficiencies and anaemia, diabetes, and cardiovascular disease in the black/pardo population. This paper provides further evidence of the importance of expanding UHC in low - and middle-income countries.
Previous literature indicates that ESF expansion is associated with reduced child mortality, mortality from cardiovascular disease, and ACSC hospitalisations [4,5,20–22]. These changes are likely due to improved access to healthcare and a focus on prevention, health promotion, proactive outreach, and early management of conditions within the ESF [3]. Whilst there is local variation in how the ESF is implemented, federal guidelines specify minimum mandatory strategic areas ESF teams must cover, including the management of hypertension, diabetes, tuberculosis, and women’s and children’s health [42]. In this study, ESF expansion was associated with reductions in mortality in the ACSC groups that mirror these mandatory strategic areas. We found that ESF expansion was associated with reductions in cardiovascular mortality of 12.9% and 7.1% in the black/pardo and white groups, respectively. We found a 27.5% reduction in mortality from infectious diseases with ESF expansion in the black/pardo population. ESF expansion was also associated with a 17.9% reduction in mortality from nutritional deficiencies and anaemia in the black/pardo population, with children under 5 y accounting for over 25% of these deaths (compared to roughly 3% of all deaths from ACSCs). Additionally, ESF-associated reductions in mortality from respiratory diseases (COPD and asthma), epilepsy, and gastric ulcers are consistent with their inclusion within ACSC definitions and the fact that these conditions are considered amenable to PHC. We found no association between ESF expansion and mortality from accidents, which is not considered sensitive to primary care, providing reassurance that the associations of ESF expansion with ACSCs reported are not due to confounding.
The differential associations between ESF expansion and mortality in black/pardo and white populations may be explained by numerous factors, with socio-economic differences a key explanatory factor. Black/pardo populations are disproportionately affected by diseases of poverty, including infectious diseases, malnutrition, and anaemia [9,43], but these conditions may be more responsive to ESF services as they are generally easier to treat in PHC settings than complex non-communicable diseases. Additionally, black/pardo populations in Brazil have lower utilisation of healthcare and higher rates of forgone healthcare [9], suggesting ESF expansion may have facilitated access to healthcare and reduced unmet need. Lastly, the finding that ESF has benefitted black/pardo populations more than white populations may not be surprising given that ESF expansion had been prioritised within poorer areas and municipalities. Surveys indicate that black/pardo populations now have greater ESF coverage (at 57.3% in 2008) than white populations (44.6% in 2008), but lower coverage of private health insurance, suggesting they are more reliant on publicly funded and provided services, including the ESF [9].
Our findings are consistent with evidence derived largely from studies conducted in North America and Europe that show “equity-enhancing” associations from PHC expansion [3]. However, these studies mostly examine associations of PHC with health inequalities across income groups. There are fewer studies examining the association of PHC with health inequalities between racial groups. In a study in the US, increasing the supply of primary care physicians was associated with larger reductions in African-American mortality than white mortality [38]. Inequalities in low birth weight between African-American and white infants are lower among those using PHC [44]. No evidence exists on the association between PHC and race in Brazil, although a few studies have examined inequalities between municipalities. Previous Brazilian studies have shown that ESF expansion was associated with greater reductions in infant mortality in municipalities with higher infant mortality at baseline [20,45]. Another study demonstrated greater reductions in infant mortality in municipalities with lower human development, also implying improvements in equity [20].
There are important limitations to this study pertinent to the interpretation. First, these analyses were conducted on municipal-level aggregated data, and more complete, individual data with ESF enrolment, consultation rates, and associated health outcomes are required to elucidate the mechanisms determining the greater benefits experienced by the black/pardo population. Second, there could be biases from the methods employed and data manipulation. However, we conducted extensive sensitivity analyses that showed that our findings are robust to ill-defined death reclassification, varying classifications of ESF coverage, and alternative model specifications. We also found no evidence of pre-intervention trends that would bias the findings. Third, there are important caveats regarding the use of race in this study. There is potential for misclassification bias of race (with race in censuses self-reported and race in death certificates reported by either the family or physician), although sensitivity analyses indicate the robustness of the findings. Black/pardo would have to be incorrectly recorded for over 30% of black/pardo deaths for the differences found to be non-significant. Additionally, by grouping together black and pardo deaths, we do not account for the large amount of heterogeneity in health outcomes between these groups [13]. Fourth, lack of statistical power due to small numbers is apparent in our analysis of associations between ESF expansion and cause-specific deaths. This precluded any potential analysis with SRRs for ACSC groups. Fifth, we used mortality from ACSCs as our outcome measure rather than the more broadly defined concept of healthcare-amenable mortality [30,46]). This was principally to focus on conditions that have been defined as amenable to PHC within the Brazilian context and to exclude those that may be strongly influenced by hospital-based care. While previous research has generally examined hospital admissions for ACSCs, this was not feasible here due to low recording of race in hospital admission data in Brazil. We present a comparison of conditions included in the Brazilian Ministry of Health’s definition of ACSCs and healthcare-amenable mortality as defined by Nolte and McKee [46] in S4 Table.
Policy-makers should note that in Brazil, where sharp inequalities persist and an ambition to achieve UHC has been boldly pursued over the last 20 years, the equity-promoting associations of PHC are evident [30]. The strong positive relationship between PHC and reduced racial inequalities in mortality provides impetus for a renewed government commitment to the ESF. Current proposals that could limit public spending in Brazil and cause disinvestment from social protection programmes, including the ESF [23], may reverse the valuable progress made towards reducing health inequalities in the country. The health inequality impacts of policy changes influencing the ESF, which is the primary vehicle for UHC in Brazil, should be carefully monitored and evaluated.
Beyond the equity-enhancing nature of PHC itself, the impressive reductions in inequality in ACSC deaths between racial groups seen in Brazil may have been facilitated by numerous factors. These include the more rapid expansion of the ESF in poorer and more deprived areas, and the proactive outreach healthcare delivered by community health workers. Whilst challenges exist, including retaining health professionals in rural areas [15] and a lack of coverage for the urban poor [47], there are valuable lessons for other countries from Brazil’s efforts to achieve UHC. The pro-equity health gains demonstrated here reflect the country’s adoption of a pro-poor pathway to UHC. Universal access was embraced from the start, services are publicly financed, there is a focus on expanding access through community-based models of care, and strong political commitment has enabled rapid and sizeable expansion [48]. Valuable lessons may be derived from other settings including Costa Rica, which similarly expanded PHC in poorer areas preferentially [49], and countries such as Tanzania, Uganda, and Chile, which have accelerated coverage in underserved areas through flexible budget allocations [50].
In conclusion, expansion of the ESF in Brazil was associated with improved health outcomes and reductions in health inequalities between racial groups. As countries aim to “progressively achieve greater equality” as part of the SDGs, these findings reinforce the importance of strong PHC-focused health systems for improving health and reducing health inequities.
Supporting Information
Zdroje
1. Tangcharoensathien V, Mills A, Palu T. Accelerating health equity: the key role of universal health coverage in the Sustainable Development Goals. BMC Med. 2015;13(1):101.
2. World Health Organization. The world health report 2010: health system financing—the path to universal coverage. Geneva: World Health Organization; 2010.
3. Starfield B, Shi L, Macinko J. Contribution of primary care to health systems and health. Milbank Q. 2005;83(3):457–502. doi: 10.1111/j.1468-0009.2005.00409.x 16202000
4. Rasella D, Harhay MO, Pamponet ML, Aquino R, Barreto ML. Impact of primary health care on mortality from heart and cerebrovascular diseases in Brazil: a nationwide analysis of longitudinal data. BMJ. 2014;349:g4014. doi: 10.1136/bmj.g4014 24994807
5. Macinko J, Dourado I, Aquino R, Bonolo Pde F, Lima-Costa MF, Medina MG, et al. Major expansion of primary care in Brazil linked to decline in unnecessary hospitalization. Health Aff (Millwood). 2010;29(12):2149–60.
6. World Bank. Brazil: country at a glance. Washington (District of Columbia): World Bank; 2016 [cited 2016 Sep 8]. http://www.worldbank.org/en/country/brazil.
7. Szwarcwald CL, Souza-Júnior PR, Damacena GN. Socioeconomic inequalities in the use of outpatient services in Brazil according to health care need: evidence from the World Health Survey. BMC Health Serv Res. 2010;10 : 217. doi: 10.1186/1472-6963-10-217 20653970
8. Szwarcwald CL, Corrêa da Mota J, Damacena GN, Sardinha Pereira TG. Health inequalities in Rio de Janeiro, Brazil: lower healthy life expectancy in socioeconomically disadvantaged areas. Am J Public Health. 2011;101(3):517–23. doi: 10.2105/AJPH.2010.195453 21233437
9. Paixão MJ, Rossetto I, Montovanele F, Carvano LM. Relatório anual das desigualdades raciais no Brasil, 2009–10. Rio de Janeiro: Editora Garamond; 2010.
10. Cardoso AM, Santos RV, Coimbra CEA Jr. Mortalidade infantil segundo raça/cor no Brasil: o que dizem os sistemas nacionais de informação? Cad Saude Publica. 2005;21 : 1602–8.
11. Nyarko KA, Lopez-Camelo J, Castilla EE, Wehby GL. Explaining racial disparities in infant health in Brazil. Am J Public Health. 2013;103(9):1675–84. doi: 10.2105/AJPH.2012.301021 23409894
12. Rasella D, Machado D, Castellanos MEP, Paim J, Szwarcwald CL, Lima D, et al. Assessing the relevance of indicators in tracking social determinants and progress toward equitable population health in Brazil. Glob Health Action. 2016;9 : 29042.
13. Travassos C, Williams DR. The concept and measurement of race and their relationship to public health: a review focused on Brazil and the United States. Cad Saude Publica. 2004;20(3):660–78. doi: /S0102-311X2004000300003 15263977
14. Rasella D, Aquino R, Santos CAT, Paes-Sousa R, Barreto ML. Effect of a conditional cash transfer programme on childhood mortality: a nationwide analysis of Brazilian municipalities. Lancet. 2013;382(9886):57–64. doi: 10.1016/S0140-6736(13)60715-1 23683599
15. Harris M, Haines A. Brazil’s Family Health Programme. BMJ. 2010;341:c4945. doi: 10.1136/bmj.c4945 21115587
16. Macinko J, Harris MJ. Brazil’s Family Health Strategy—delivering community-based primary care in a universal health system. N Engl J Med. 2015;372(23):2177–81. doi: 10.1056/NEJMp1501140 26039598
17. Ministério da Saúde Departamento de Atenção Básica. Histórico de cobertura da saúde da família. Brasilia: Ministério da Saúde; 2015 [cited 2015 Apr 11]. http://dab.saude.gov.br/portaldab/historico_cobertura_sf.php.
18. Ministério da Saúde. Política Nacional de Atenção Básica. Brasilia: Ministério da Saúde; 2011.
19. Henrique F, Calvo MC. [The degree of implementation of the Family Health Program and social indicators.] Cien Saude Colet. 2009;14(Suppl 1):1359–65.
20. Aquino R, de Oliveira NF, Barreto ML. Impact of the family health program on infant mortality in Brazilian municipalities. Am J Public Health. 2009;99(1):87–93. doi: 10.2105/AJPH.2007.127480 19008516
21. Guanais FC. The combined effects of the expansion of primary health care and conditional cash transfers on infant mortality in Brazil, 1998–2010. Rev Panam Salud Publica. 2014;36(1):65–9. 25211681
22. Macinko J, Marinho de Souza MdF, Guanais FC, da Silva Simoes CC. Going to scale with community-based primary care: an analysis of the family health program and infant mortality in Brazil, 1999–2004. Soc Sci Med. 2007;65(10):2070–80. doi: 10.1016/j.socscimed.2007.06.028 17689847
23. Watts J. Brazil’s health system woes worsen in economic crisis. Lancet. 2016;387(10028):1603–4. doi: 10.1016/S0140-6736(16)30249-5 27116057
24. Telles EE. Race in another America: the significance of skin color in Brazil. Princeton (New Jersey): Princeton University Press; 2004.
25. Lima-Costa MF, Rodrigues LC, Barreto ML, Gouveia M, Horta BL, Mambrini J, et al. Genomic ancestry and ethnoracial self-classification based on 5,871 community-dwelling Brazilians (the Epigen Initiative). Sci Rep. 2015;5 : 9812. doi: 10.1038/srep09812 25913126
26. Purdy S, Griffin T, Salisbury C, Sharp D. Ambulatory care sensitive conditions: terminology and disease coding need to be more specific to aid policy makers and clinicians. Public Health. 2009;123(2):169–73. doi: 10.1016/j.puhe.2008.11.001 19144363
27. Macinko J, Guanais FC, de Fatima M, de Souza M. Evaluation of the impact of the Family Health Program on infant mortality in Brazil, 1990–2002. J Epidemiol Community Health. 2006;60(1):13–9. doi: 10.1136/jech.2005.038323 16361449
28. Guanais F, Macinko J. Primary care and avoidable hospitalizations: evidence from Brazil. J Ambul Care Manage. 2009;32(2):115–22. doi: 10.1097/JAC.0b013e31819942e51 19305223
29. Rocha R, Soares RR. Evaluating the impact of community-based health interventions: evidence from Brazil’s Family Health Program. Health Econ. 2010;19(Suppl):126–58.
30. Hone T, Rasella D, Barreto M, Atun R, Majeed A, Millett C. Large reductions in amenable mortality associated with Brazil’s primary care expansion and strong health governance. Health Aff (Millwood). 2017;36(1):149–58.
31. de Andrade CLT, Szwarcwald CL. Socio-spatial inequalities in the adequacy of Ministry of Health data on births and deaths at the municipal level in Brazil, 2000–2002. Cad Saude Publica. 2007;23(5):1207–16. 17486242
32. Ministério da Saúde. DATASUS. Brasilia: Ministério da Saúde; 2015 [cited 2015 Apr 4]. http://tabnet.datasus.gov.br/.
33. Instituto Brasileiro de Geografia e Estatística. Banco de dados. Rio de Janeiro: Instituto Brasileiro de Geografia e Estatística; 2016 [cited 2016 Dec 4]. http://www.ibge.gov.br/.
34. Alfradique ME, Bonolo PdF, Dourado I, Lima-Costa MF, Macinko J, Mendonca CS, et al. Ambulatory care sensitive hospitalizations: elaboration of Brazilian list as a tool for measuring health system performance (Project ICSAP--Brazil). Cad Saude Publica. 2009;25(6):1337–49. 19503964
35. França E, Teixeira R, Ishitani L, Duncan BB, Cortez-Escalante JJ, de Morais Neto OL, et al. Ill-defined causes of death in Brazil: a redistribution method based on the investigation of such causes. Rev Saude Publica. 2014;48(4):671–81. doi: 10.1590/S0034-8910.2014048005146 25210826
36. Travassos C, Laguardia J, Marques PM, Mota JC, Szwarcwald CL. Comparison between two race/skin color classifications in relation to health-related outcomes in Brazil. Int J Equity Health. 2011;10 : 35. doi: 10.1186/1475-9276-10-35 21867522
37. Mackenbach JP, Kunst AE, Cavelaars AEJM, Groenhof F, Geurts JJM. Socioeconomic inequalities in morbidity and mortality in western Europe. Lancet. 1997;349(9066):1655–9. 9186383
38. Shi L, Macinko J, Starfield B, Politzer R, Xu J. Primary care, race, and mortality in US states. Soc Sci Med. 2005;61(1):65–75. doi: 10.1016/j.socscimed.2004.11.056 15847962
39. Marmot M. Social determinants of health inequalities. Lancet. 2005;365(9464):1099–104. doi: 10.1016/S0140-6736(05)71146-6 15781105
40. Wooldridge J. Introductory econometrics: a modern approach. 5th ed. Boston: Cengage Learning; 2013. 878 p.
41. Rasella D, Aquino R, Barreto ML. Impact of the Family Health Program on the quality of vital information and reduction of child unattended deaths in Brazil: an ecological longitudinal study. BMC Public Health. 2010;10 : 380. doi: 10.1186/1471-2458-10-380 20587036
42. Ministério da Saúde. Programa Saúde da Família: ampliando a cobertura para consolidar a mudança do modelo de Atenção Básica. Rev Bras Saude Mater Infant. 2003;3 : 113–25.
43. Stevens P. Diseases of poverty and the 10/90 gap. London: International Policy Network; 2004.
44. Shi L, Stevens GD, Wulu JT Jr, Politzer RM, Xu J. America’s health centers: reducing racial and ethnic disparities in perinatal care and birth outcomes. Health Serv Res. 2004;39(6 Pt 1):1881–901. doi: 10.1111/j.1475-6773.2004.00323.x 15533192
45. Shei A. Brazil’s conditional cash transfer program associated with declines in infant mortality rates. Health Aff (Millwood). 2013;32(7):1274–81.
46. Nolte E, McKee CM. Measuring the health of nations: updating an earlier analysis. Health Aff (Millwood). 2008;27(1):58–71.
47. Costa NdR. The Family Health Strategy: primary health care and the challenge of Brazilian metropolises. Cien Saude Colet. 2016;21(5):1389–98. doi: 10.1590/1413-81232015215.24842015 27166889
48. Bump J, Cashin C, Chalkidou K, Evans D, González-Pier E, Guo Y, et al. Implementing pro-poor universal health coverage. Lancet Glob Health. 2015;4(1):e14–6. doi: 10.1016/S2214-109X(15)00274-0 26700794
49. Rosero-Bixby L. Spatial access to health care in Costa Rica and its equity: a GIS-based study. Soc Sci Med. 2004;58(7):1271–84. doi: 10.1016/S0277-9536(03)00322-8 14759675
50. World Health Organization. The world health report 2008: primary health care—now more than ever. Geneva: World Health Organization; 2008.
Štítky
Interní lékařství
Článek vyšel v časopisePLOS Medicine
Nejčtenější tento týden
2017 Číslo 5- Červená fermentovaná rýže účinně snižuje hladinu LDL cholesterolu jako vhodná alternativa ke statinové terapii
- Berberin: přírodní hypolipidemikum se slibnými výsledky
- Příznivý vliv Armolipidu Plus na hladinu cholesterolu a zánětlivé parametry u pacientů s chronickým subklinickým zánětem
- Alternativní léčebné možnosti u hypercholesterolemie při intoleranci statinů
- Vliv kombinace nutraceutik na remodelaci levé komory srdeční u osob s metabolickým syndromem
-
Všechny články tohoto čísla
- Rotavirus vaccine will have an impact in Asia
- Towards control of the global HIV epidemic: Addressing the middle-90 challenge in the UNAIDS 90–90–90 target
- Ebola exposure, illness experience, and Ebola antibody prevalence in international responders to the West African Ebola epidemic 2014–2016: A cross-sectional study
- Long-term inpatient disease burden in the Adult Life after Childhood Cancer in Scandinavia (ALiCCS) study: A cohort study of 21,297 childhood cancer survivors
- Contribution of systemic and somatic factors to clinical response and resistance to PD-L1 blockade in urothelial cancer: An exploratory multi-omic analysis
- Association between expansion of primary healthcare and racial inequalities in mortality amenable to primary care in Brazil: A national longitudinal analysis
- Vitamin D levels and susceptibility to asthma, elevated immunoglobulin E levels, and atopic dermatitis: A Mendelian randomization study
- Maternal age and severe maternal morbidity: A population-based retrospective cohort study
- Measuring personal beliefs and perceived norms about intimate partner violence: Population-based survey experiment in rural Uganda
- A universal testing and treatment intervention to improve HIV control: One-year results from intervention communities in Zambia in the HPTN 071 (PopART) cluster-randomised trial
- Estimation of the cost-effectiveness of HIV prevention portfolios for people who inject drugs in the United States: A model-based analysis
- First-trimester artemisinin derivatives and quinine treatments and the risk of adverse pregnancy outcomes in Africa and Asia: A meta-analysis of observational studies
- Comparison of artemether-lumefantrine and chloroquine with and without primaquine for the treatment of infection in Ethiopia: A randomized controlled trial
- Impact evaluation of different cash-based intervention modalities on child and maternal nutritional status in Sindh Province, Pakistan, at 6 mo and at 1 y: A cluster randomised controlled trial
- Tobacco control: Developing an innovative and effective global strategy
- Data sharing in clinical trials: An experience with two large cancer screening trials
- Mortality and kidnapping estimates for the Yazidi population in the area of Mount Sinjar, Iraq, in August 2014: A retrospective household survey
- PLOS Medicine
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Mortality and kidnapping estimates for the Yazidi population in the area of Mount Sinjar, Iraq, in August 2014: A retrospective household survey
- Vitamin D levels and susceptibility to asthma, elevated immunoglobulin E levels, and atopic dermatitis: A Mendelian randomization study
- Maternal age and severe maternal morbidity: A population-based retrospective cohort study
- Estimation of the cost-effectiveness of HIV prevention portfolios for people who inject drugs in the United States: A model-based analysis
Přihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



