-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praxe
Top novinky
ReklamaThe GATA Factor Regulates . Developmental Timing by Promoting Expression of the Family MicroRNAs
In the nematode roundworm C. elegans, seam cells, a type of adult stem cell, divide in a completely predictable manner throughout post-embryonic development. Study of the control of the timing of these cells’ division and differentiation led to the discovery of the first microRNAs, which are small non-coding RNAs that regulate the expression of protein-coding mRNAs, but knowledge of the regulation of expression of microRNAs themselves within C. elegans stem cells remains incomplete. In this study, the GATA-family transcription factor elt-1, known to be important for the formation and maintenance of tissues during embryonic and post-embryonic development, is found to regulate the expression of let-7 family microRNAs in stem cells during late developmental stages. It is found to do so redundantly with daf-12, the only other transcription factor previously known to directly regulate microRNA expression in C. elegans. In addition, the presence of ELT-1 in vivo binding near microRNA coding DNA sequences suggests that its contribution to the regulation of microRNA expression is likely through direct regulation of transcription. Stem cells are important in development, tissue homeostasis, and malignancy, so additional knowledge of the mechanisms underlying their maintenance, renewal, and differentiation is of broad interest.
Published in the journal: . PLoS Genet 11(3): e32767. doi:10.1371/journal.pgen.1005099
Category: Research Article
doi: https://doi.org/10.1371/journal.pgen.1005099Summary
In the nematode roundworm C. elegans, seam cells, a type of adult stem cell, divide in a completely predictable manner throughout post-embryonic development. Study of the control of the timing of these cells’ division and differentiation led to the discovery of the first microRNAs, which are small non-coding RNAs that regulate the expression of protein-coding mRNAs, but knowledge of the regulation of expression of microRNAs themselves within C. elegans stem cells remains incomplete. In this study, the GATA-family transcription factor elt-1, known to be important for the formation and maintenance of tissues during embryonic and post-embryonic development, is found to regulate the expression of let-7 family microRNAs in stem cells during late developmental stages. It is found to do so redundantly with daf-12, the only other transcription factor previously known to directly regulate microRNA expression in C. elegans. In addition, the presence of ELT-1 in vivo binding near microRNA coding DNA sequences suggests that its contribution to the regulation of microRNA expression is likely through direct regulation of transcription. Stem cells are important in development, tissue homeostasis, and malignancy, so additional knowledge of the mechanisms underlying their maintenance, renewal, and differentiation is of broad interest.
Introduction
Extensive study of postembryonic development in the nematode Caenorhabditis elegans has advanced our understanding of the temporal regulation of development and the roles of microRNAs (miRNAs) in controlling gene expression [1–5]. In C. elegans, developmental timing is regulated by the heterochronic gene network, which directs the transitions among discrete developmental stages largely by initiating the stage-specific of miRNAs that down-regulate other stage-specific factors [6–8]. Many gene products of the C. elegans heterochronic regulatory network are conserved in metazoans, including the LET-7 family of miRNAs and LIN-28, a post-transcriptional repressor of these miRNAs [9–11]. LET-7 family miRNAs regulate the expression of multiple targets, including LIN-41, and the LIN-28-LET-7-LIN-41 pathway has been shown to regulate differentiated states of stem cells in both C. elegans and mammals [3,4,12–17]. The LIN-28-LET-7 axis is important in human physiology and disease, as it is involved in induced pluripotency [17–19], adult intestinal stem cell function [20], tissue repair [21], and malignancy [22,23].
During normal development, dafachronic acid steroid hormones are synthesized by C. elegans in response to favorable growth conditions [24]. They stimulate the nuclear hormone receptor (NHR) DAF-12, the vitamin D NHR ortholog, to promote progression from the 2nd larval stage (L2) to the 3rd larval stage (L3) [24–26] by, in part, initiating expression of the LET-7 family of miRNAs, miR-48, miR-84, and miR-241, during or near the L2-to-L3 molt [27,28]. In this way, the nuclear-hormone receptor DAF-12 acts as a key switch in the regulation of developmental fate [27–29]. Expression of miRs has been proposed to drive transition from one larval stage to the next [8]. DAF-12/NHR is known to regulate miRNA expression in this system [7], but cannot itself account for all of the upstream transcriptional regulation of the LET-7 family of miRNA, as its null phenotype is much weaker than that of the LET-7 family itself [25,26,30]. LET-7 is known to be under-expressed in both daf-12 mutants [28] and alg-1 mutants [31], and a portion of its promoter region has been identified to be required for correct temporal expression [32], but the factor(s) that directly regulate its transcription are not yet known. Additionally, the transcriptional regulation of LIN-4 remains largely unknown. A previous study determined that LIN-66 provides regulation of developmental timing in parallel to daf-12, but the molecular function of the LIN-66 protein remains unknown [33]. Recently, the PERIOD homolog lin-42 has been found to negatively regulate the expression of multiple microRNAs, including LIN-4 and LET-7 [34]. The presence of other regulatory factors that act on the transcription of these miRs is implied, and the identification of these factors would significantly advance our understanding of developmental timing regulation as well of miRNA function in general.
In this study, we performed a forward genetic screen to identify enhancers of the heterochronic phenotype of daf-12(null) animals; the purpose was to identify new factors that act in parallel to it in the regulation of the heterochronic genetic network. A partial loss-of-function allele of the GATA transcription factor elt-1 was positionally cloned, and the role of ELT-1 in the heterochronic gene network is described.
Results
ELT-1 and DAF-12 redundantly regulate seam cell fate during post-embryonic development
An EMS-mutagenesis screen was performed to identify mutations that enhance the heterochronic phenotype of daf-12(rh61rh411) animals. One such enhancer allele was identified and mapped to the elt-1/GATA gene by genetic mapping techniques including genetic and SNP markers, whole-genome shotgun sequencing to identify candidate variations, and transgene-mediated phenotype complementation. As shown in Figs. 1, 2A–2F, and S1, and Table 1, the elt-1/GATA(ku491) mutation causes delayed heterochronic phenotypes when animals are double-mutant for daf-12(rh61rh411), but not when animals are daf-12(+). Specifically, the number of seam cells in the lateral hypodermis is dramatically higher than normal during the 4th larval (L4) and young adult (Y.A.) stages (Fig. 1). In addition, the double-mutants have an L4-stage bursting vulva phenotype (Table 1), similar to that caused by mutation of delayed heterochronic genes, including let-7 and the three other let-7 family miR genes (mir-48, mir-84, and mir-241) [3,30]. These results indicate that elt-1/GATA has an important role in regulating developmental timing in parallel to daf-12. daf-12 is known to regulate developmental timing by promoting the expression of the three LET-7 family miRNAs [27,28], suggesting that elt-1 may regulate developmental timing by acting either in parallel to or upstream of the miRNAs.
Fig. 1. ELT-1/GATA acts in parallel to DAF-12/NHR to regulate developmental timing in C. elegans. 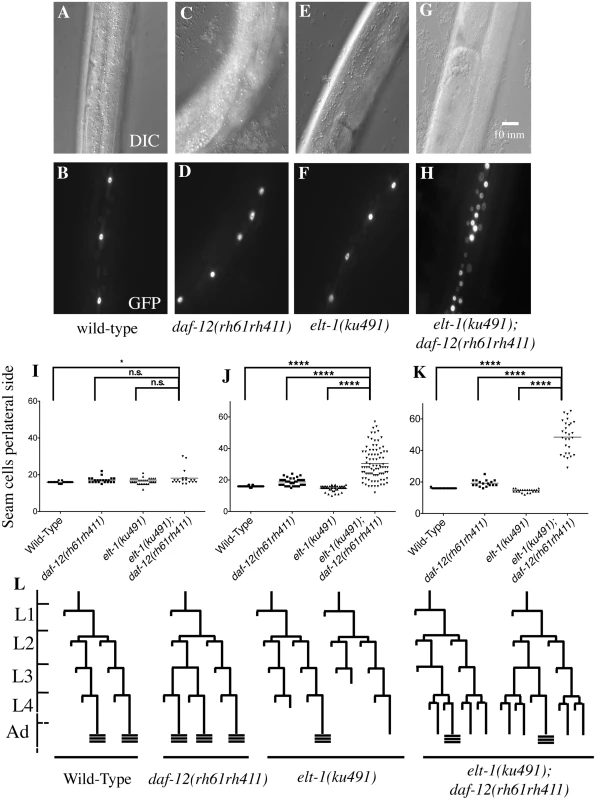
A-H, DIC and fluorescence images of representative young-adult stage animals with the indicated genotypes, showing that seam cell numbers, visualized with a scm::GFP marker, are drastically increased in elt-1(ku491); daf-12(rh61rh411) double mutants but not in each single mutant. I-K, Scatter plots showing the distribution of the number of seam cells per lateral side for each genotype at L4 and young adult stages; horizontal bar is mean. L, cellular lineage diagrams for mutants, showing variable cell fate defects in the elt-1(ku491) mutants during the L3 and L4 stages. **** indicates p-value < 0.0001. Data for L1 to L3 stages and summary statistics of data for all developmental stages are shown in S1 Fig. Fig. 2. elt-1/(ku491) mutant animals have defective adult alae formation. 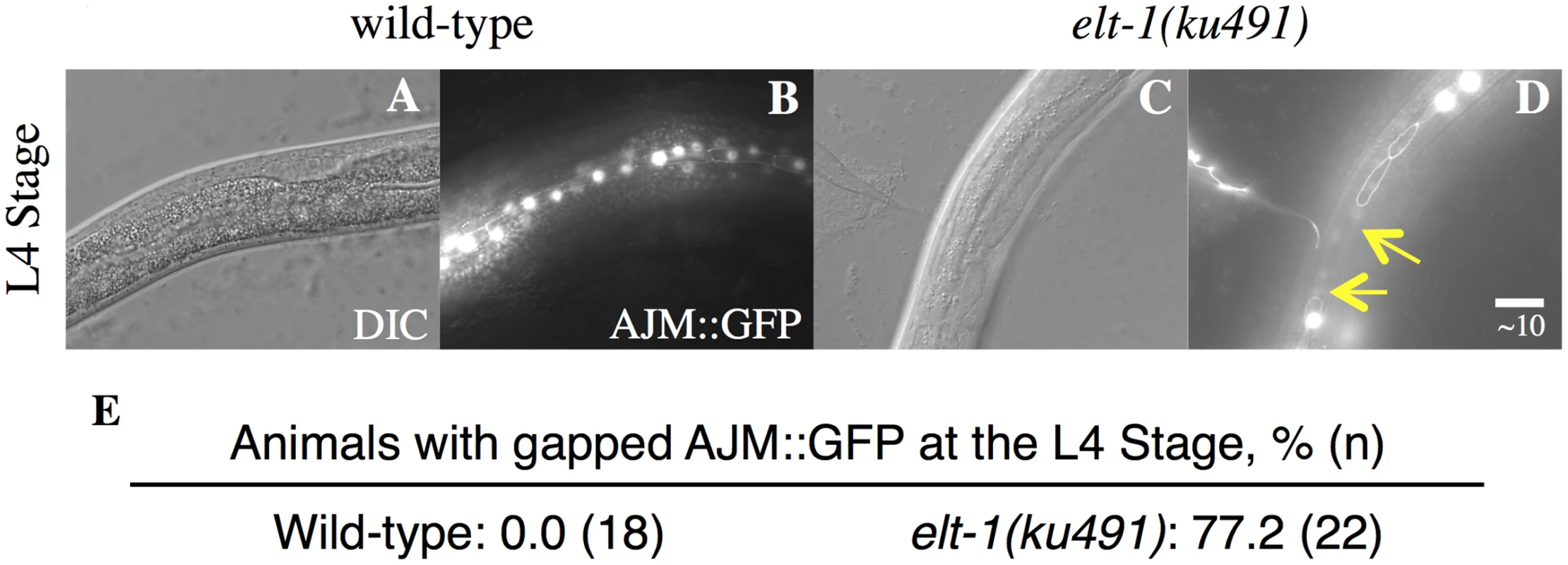
A-D, DIC and GFP fluorescence images showing adherins junctions of L4 animals with indicated genotypes. Arrow in D marks the gap of AJM::GFP fluorescence. E, Percentage of L4 animals with AJM::GFP fluorescence gaps in animals with the indicated genotypes. Tab. 1. elt-1 mutants have an L4-stage bursting vulva and defective alae formation and elt-1(ku491) is a partial loss-of-function allele. 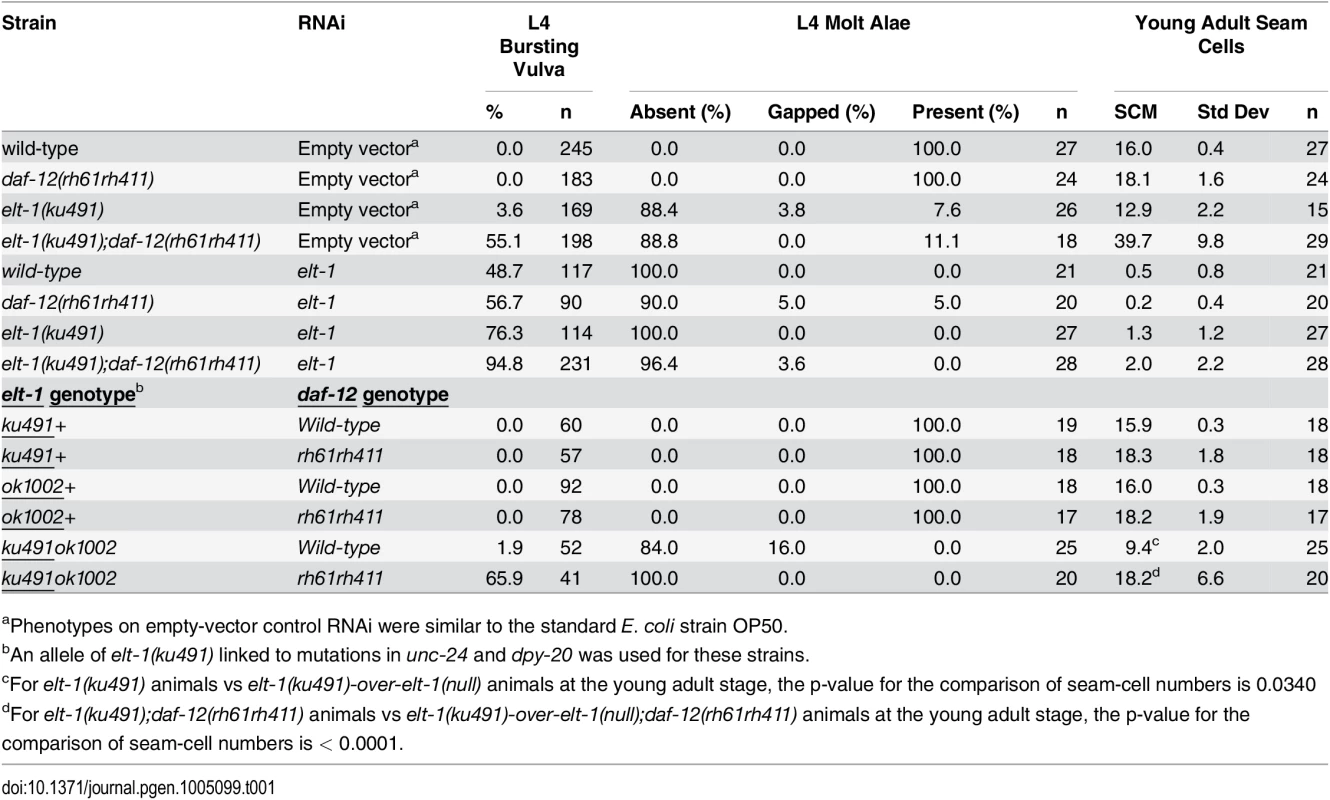
aPhenotypes on empty-vector control RNAi were similar to the standard E. coli strain OP50. In addition to the heterochronic phenotypes that are only present when daf-12 is null, elt-1/GATA(ku491) animals have daf-12(null)-independent defects in adult alae formation at the L4 molt and in the maintenance of seam cells during post-embryonic development (Table 1). These data are consistent with previous studies that have used post-embryonic RNAi against elt-1/GATA to show that it is required for the maintenance of seam-cell cell identities during post-embryonic development and for the formation of adult alae at the L4 molt [35–37]. The timing of premature differentiation of seam-cells in elt-1(ku491) single mutants and of excessive seam-cell divisions in the elt-1(ku491);daf-12(rh61rh411) double-mutants is variable but primarily during the L4 stage (Fig. 1L). Additionally, no supernumerary molts were observed, and male elt-1/GATA(ku491) single-mutant animals are able to cross-fertilize hermaphrodites. Among seam-cell nuclei that formed after seam-cell fusion into the hypodermal syncytium, we did not determine the proportion due to duplicate nuclei within fused cells versus in cells with completed divisions. Proliferation of seam cells and the L4 bursting vulva phenotype in elt-1(ku491); daf-12(null) double-mutant animals are novel phenotypes for elt-1/GATA and indicate that it is a heterochronic gene.
The elt-1/GATA(ku491) mutant allele is a partial loss-of-function allele
As shown in Table 1, the elt-1(ku491) mutation is fully recessive. Animals with the elt-1(ku491) mutation in trans to a null allele, elt-1(ok1002), have an equivalent or stronger phenotype for alae formation defects and L4 burst vulva when compared to elt-1/GATA(ku491) homozgotes. These data indicate that the elt-1(ku491) mutation is likely a partial loss-of-function mutation that compromises the role of the ELT-1 protein in post-embryonic developmental timing and adult alae formation, while leaving it competent to function in the embryonic specification of the hypodermis. Interestingly, there is reduced seam-cell proliferation in elt-1(ku491)-over-elt-1(null); daf-12(rh61rh411) animals compared to elt-1(ku491); daf-12(rh61rh411) animals. This is likely due to allelic haploinsufficiency of elt-1(ku491) for the maintenance of post-embryonic seam cell fate as, when daf-12 is wild-type, elt-1(ku491)-over-elt-1(null) animals also have a decrease in the numbers of seam-cells compared to elt-1(ku491) single-mutants animals, and elt-1 has previously been shown to be required for the post-embryonic maintenance of seam-cell cell fate [35–37].
The elt-1/GATA(ku491) mutant allele contains a C-to-T substitution in the 48th base-pair of exon 5, causing a proline-to-serine missense mutation at amino-acid residue 298 of the ELT-1 protein isoform A. ELT-1 has previously been shown to contain two conserved Zinc-finger DNA binding domains, each of which contains a single C-X2-C-X17-C-X2-C motif [38]; proline298 is 6 amino acid residues C-terminal to the second cysteine of the N-terminal Zn-finger DNA binding domain. This proline specifically, and the N-terminal Zinc-finger DNA binding domain overall, is conserved among worms, fish, mice, and humans (S2 Fig). Examining the structure of the murine GATA1 Zn-finger DNA-binding domains (Protein Databank accession number 3VD6) [39], we found that the amino acid residue conserved with C. elegans ELT-1 proline298 is located within a hairpin fold that bring the four cysteine residues of the N-terminal Zn-finger domain near to the required Zinc molecule (S3 Fig). This suggests that the elt-1(ku491) mutation could potentially alter the secondary structure of the N-terminal Zn-finger domain in the ELT-1 protein by interfering with the folding required for Zinc binding, causing its functional inactivation. This presumably does not have an effect on the ability of the C-terminal domain to recognize its target sequences as the elt-1(ku491) mutant has a non-null phenotype.
Five alleles of elt-1 were obtained from the million mutation project and examined in the presence of the daf-12(rh61rh411) mutation (S1 Table); two showed very mild increases in the number of seam-cells when compared to daf-12(rh61rh411) animals, but none showed an L4 bursting vulva phenotype or defective alae formation. These alleles contain mutations with mild effects on the ELT-1 protein, as listed in S1 Table, and have no previously described phenotype, so they likely are minor mutations that do not substantially interfere with the normal function of ELT-1, unlike the proline298-to-serine mutation present in elt-1(ku491).
In sum, the elt-1/GATA(ku491) allele significantly reduces the normal function of ELT-1/GATA during post-embryonic development, likely by disrupting the DNA binding ability of its N-terminal Zn-finger domain.
elt-1/GATA likely acts upstream of key heterochronic genes to control developmental timing
The well-described heterochronic gene network controls developmental timing in C. elegans [8]. To assess a possible genetic relationship between elt-1/GATA and key genes in the heterochronic gene network, the phenotype of elt-1(ku491); daf-12(rh61rh411) double-mutant animals was examined while the expression of several key heterochronic genes were each reduced by feeding RNAi, applied starting at the L1 stage. The results of this interaction analysis (Table 2) shows that the heterochronic phenotypes of elt-1/GATA(ku491); daf-12(rh61rh411) double-mutant animals requires normal activity of the products of the heterochronic genes lin-14, lin-28, hbl-1, lin-41, lin-42, and mab-10, as RNAi of these genes significantly reduced both the high seam cell number and busting vulva phenotypes. Additionally, these phenotypes were enhanced by knockdown of the lin-46 gene, and the seam-cell proliferation phenotype was not affected by knockdown of the lin-29 gene. Knockdown of ceh-16, which is involved in the regulation of seam-cell fate during post-embryonic development [40,41], suppressed both the high seam cell number and busting-vulva phenotypes, while RNAi of either kin-20 or dre-1, which are both involved in the promotion of late larval fates [42,43], each suppressed the seam-cell proliferation phenotype but not the bursting-vulva phenotype.
Tab. 2. Epistasis analysis of elt-1(ku491); daf-12(rh61rh411) mutant phenotypes with RNAi of other heterochronic genes. 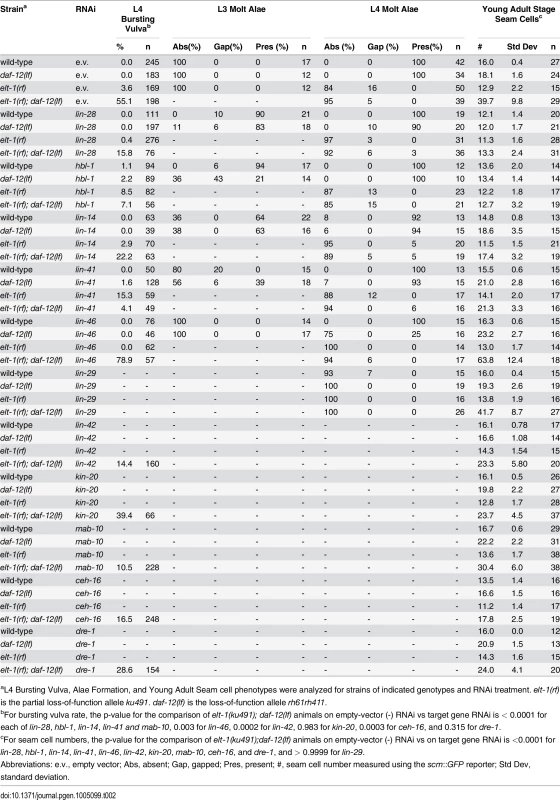
aL4 Bursting Vulva, Alae Formation, and Young Adult Seam cell phenotypes were analyzed for strains of indicated genotypes and RNAi treatment. elt-1(rf) is the partial loss-of-function allele ku491. daf-12(lf) is the loss-of-function allele rh61rh411. Interestingly, the daf-12(null)-independent defect in the formation of adult alae seen in elt-1/GATA(ku491) single-mutant animals was not affected by knock-down of any of the genes examined. These results indicate that the defect in developmental timing in elt-1(ku491); daf-12(rh61rh411) double-mutant animals is likely to be within or upstream of the part of the heterochronic gene network that controls late larval stages, while the daf-12(null)-independent defects of elt-1(ku491) animals are independent of the heterochronic gene network.
elt-1/GATA mutant animals have defective down-regulation of lin-41 during the L4 stage
As shown in Figs. 1, 2, S1, and Table 1, the heterochronic phenotypes of the elt-1/GATA(ku491); daf-12(rh61rh411) double-mutant animals are during late stages of post-embryonic development, L4 and Young Adulthood. The LIN-28 and HBL-1 proteins are known to be down-regulated at the L2 molt and during the L3 stage, respectively [8], which is mostly prior to the emergence of the heterochronic phenotypes of elt-1(ku491); daf-12(rh61rh411) double-mutant animals. The expression level of these genes’ mRNA at the L4 stage was found to be normally down-regulated in elt-1/GATA(ku491); daf-12(rh61rh411) double-mutant animals (S4 Fig). Therefore, altered expression of lin-28 and hbl-1 is unlikely to be responsible for the phenotypes of elt-1(ku491); daf-12(rh61rh411) double-mutants.
The LIN-41 protein is also highly expressed during larval development, but in contrast to LIN-28 and HBL-1, it is down-regulated primarily during the L4 stage by the LET-7 miRNA [3,13]. The heterochronic phenotypes of elt-1/GATA(ku491); daf-12(rh61rh411) double-mutant animals are at the same developmental stage as when LIN-41 is normally down-regulated, so the dynamics of lin-41 gene expression were examined in these mutant animals. As shown in Fig. 3A, elt-1/GATA(ku491); daf-12(rh61rh411) double-mutant animals fail to down-regulate the level of the lin-41 mRNA, in contrast to animals that are wild-type or carry either mutation individually. Summary descriptive statistics and statistical analysis for all mRNA qPCR results are shown in S4 Fig.
Fig. 3. elt-1(ku491);daf-12(rh61rh411) double-mutant animals fail to down-regulate the heterochronic gene lin-41 during development. 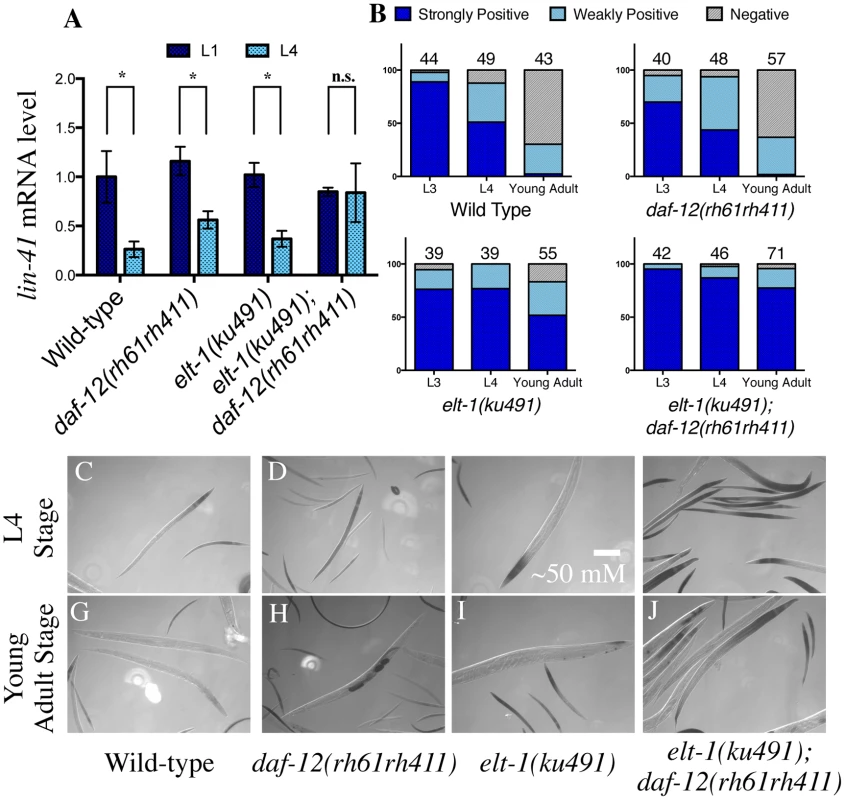
A, Lin-41 mRNA is down-regulated normally during L4 in wild-type and single-mutant animals as measured with RT-qPCR, but elt-1(ku491);daf-12(rh61rh411) double-mutant animals continue to express it at a level indistinguishable from their L1 level. Detailed descriptive statistics and p-values in S4 Fig. B, elt-1(ku491);daf-12(rh61rh411) double-mutant animals fail to down-regulate a lin-41 3’UTR reporter. The number scored per genotype and stage are noted above each column. Representative images of X-gal stain shown at L4 (C-F) and Young Adult (G-J) stages. To further examine the regulation of lin-41 mRNA during L4 in elt-1(ku491); daf-12(rh61rh411) double-mutant animals, the integrated transgene pkIs2084 was obtained and crossed into the mutant strains; pkIs2084 contains the beta-galactosidase gene under the control of the pan-hypodermal col-10 promoter and the lin-41 3’ untranslated region (3’UTR). This reporter has previously been shown to be down regulated in wild-type animals from the L3 stage to the L4 and young adult stages, and that this down regulation requires both LET-7 miRNA and the cofactors needed for miRNA-induced gene silencing (e.g., Argonaute) [3,44]. We found that elt-1/GATA(ku491); daf-12(rh61rh411) double-mutant animals fail to down-regulate the reporter correctly (Fig. 3B-J), indicating that those animals are defective in the negative regulation of the lin-41 3’UTR that normally occurs during the L4 stage. These results suggest that elt-1 normally contributes to developmental timing, at least in part, by promoting the down-regulation of lin-41 expression during L4, and that it may do so by promoting the expression of LET-7.
elt-1/GATA normally promotes the expression of the developmental timing miRNAs LET-7, miR-48, miR-84, and miR-241
The LET-7 family of miRNAs (miR-48, miR-84, and miR-241) have previously been shown to be expressed during or near the L2 molt to promote developmental progression [27,28], while LET-7 is expressed primarily during L4 and required for the L4-to-Adult transition, largely by down-regulating lin-41 mRNA [3,13]. The expression of the LET-7, miR-48, miR-84, and miR-241 miRNAs was therefore examined during the L4 stage in elt-1(ku491); daf-12(rh61rh411) double-mutant animals using RT-qPCR. As shown in Fig. 4A, elt-1(ku491); daf-12(rh61rh411) double-mutant animals have deficient expression of LET-7 and each of the LET-7 family of miRNAs (miR-48, miR-84, and Mir-241). The heterochronic miRNA LIN-4 was also analyzed, although its expression is initiated at early larval stages to drive the L1-to-L2 transition, and was therefore expected to be normal in these mutants. Summary descriptive statistics and statistical analysis results are in S2 Table.
Fig. 4. LET-7 family miRNAs are decreased in elt-1(ku491);daf-12(rh61rh411) double-mutant animals and ELT-1 binds to the let-7 promoter in vivo. 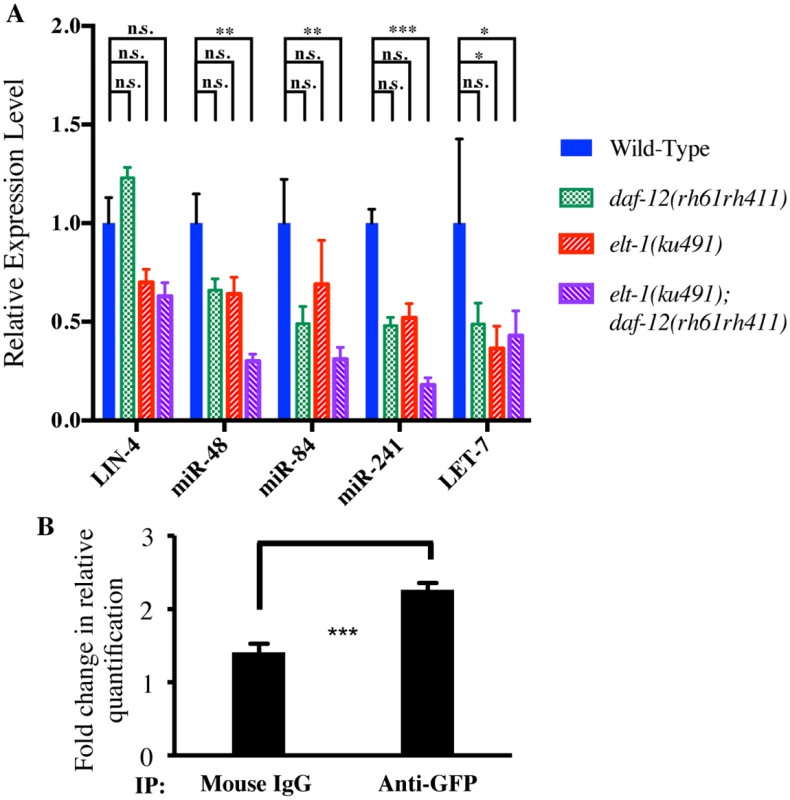
A. ELT-1 and DAF-12 redundantly regulate LET-7, miR-48, miR-84, and miR-241. The expression level of target miRNAs was determined by RT-qPCR using synchronous L4 animals. Graphs show mean ± standard error. Detailed descriptive statistics and p-values are listed in S2 Table. B, ChIP-qPCR data showing enrichment of ELT-1 binding to a region in the let-7 promoter. Samples from a strain expressing an ELT-1::GFP fusion transgene were subject to immunoprecipitation using either control IgG or anti-GFP antibody. The primers were specific to a region 1.7kb upstream of the let-7 transcription start site (Ch. X, 14747074 to 14747179). Graphs show mean ± standard error of triplicate experiments. ELT-1 also binds to promoters of other let-7 family genes (See Table 3). p-values: *, < 0.05; **, < 0.01; ***, < 0.001; ****, < 0.0001. Tab. 3. Analysis of modENCODE ChIP-Sequencing with ELT-1::GFP for selected genes. 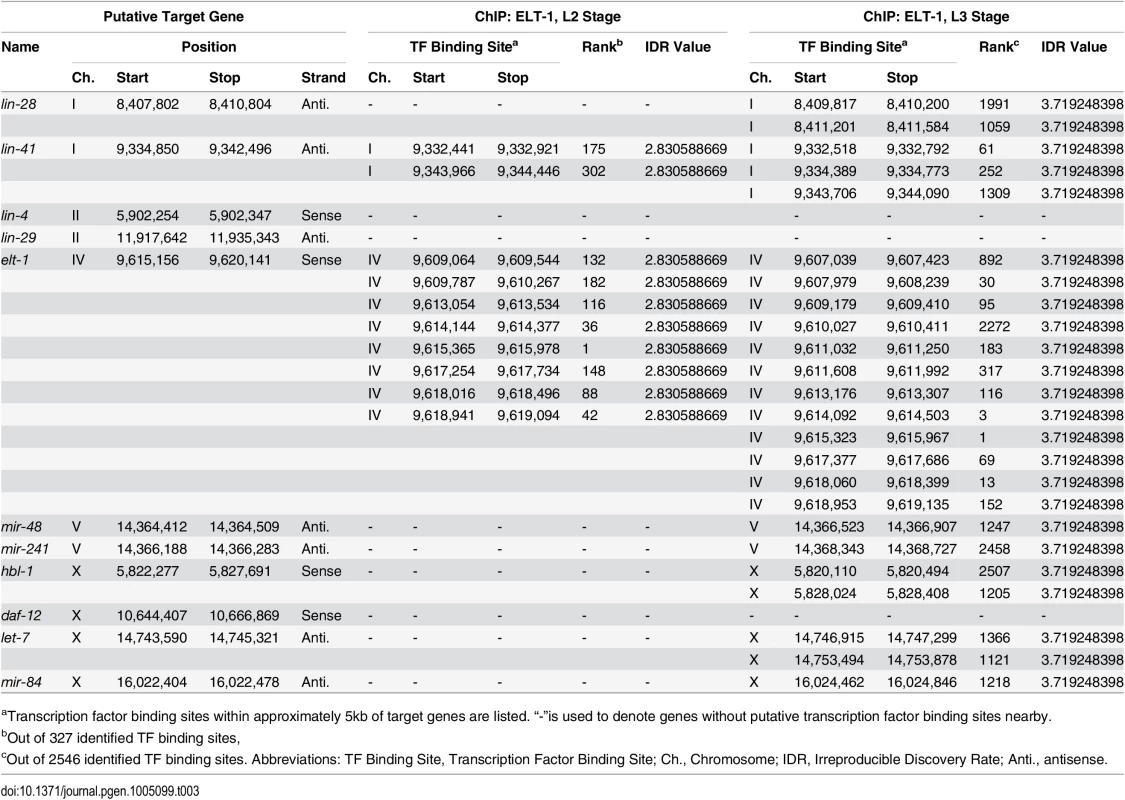
aTranscription factor binding sites within approximately 5kb of target genes are listed. “-”is used to denote genes without putative transcription factor binding sites nearby. The ELT-1 protein binds near the sequences encoding the let-7 miRNA family in vivo
A recent study examined the binding sites of a wide range of C. elegans transcription factors, including of ELT-1, using Chromatin Immunoprecipitation followed by high-throughput sequencing [45]. Examining their peak-calls near selected heterochronic genes (summarized in Table 3), we identified that ELT-1 binds to sites in the likely promoter regions for the DNA sequences encoding the let-7 family miRNAs during the L3 stage, while binding was not detected during the L2 stage. L4 stage-specific ChIP of ELT-1 was not included in this report [44]. To partially replicate their genome-wide ChIP-seq data for ELT-1, we performed qPCR following ChIP using L3-L4 stage-enriched worms. We designed qPCR primers based on 9 putative GATA transcription-factor binding sites within three kilobases 5’ of the transcription start site of let-7 gene. Among them, one primer set showed statistically-significant enrichment of ELT-1 binding (Fig. 4B). The genomic region corresponding to this primer set (Ch. X: 14,747,074 to 14,747,179; ~1.7 kb upstream of the transcription start site) overlapped with a binding site found in the modENCODE ChIP-seq data (Ch. X: 14,746,915 to 14,747,299; Table 3), supporting their finding that ELT-1 directly regulates the transcription of the let-7 gene.
Discussion
The study of postembryonic developmental timing in C. elegans has made important contributions to our understanding of the mechanisms of temporal developmental control in multicellular animals, including the initial discovery of miRNAs and of their role in the temporal regulation of key heterochronic genes’ expression [1–3,5]. Given that stage-specific expressions of these miRNAs controls the dynamic state of the heterochronic gene network, understanding the regulation of the expression of these miRNAs is an important problem with significant gaps in our current understanding [7]. In this study, we used a genetic enhancer screen to identify the GATA transcription factor ELT-1 as a new heterochronic gene and have shown that it contributes to developmental timing by providing positive regulation of the expression of the developmental timing miRNAs LET-7, miR-48, miR-84, and miR-241.
In C. elegans, the GATA transcription factor elt-1 has previously been shown to be required for formation of the hypodermis during embryonic development [46] and for the maintenance of cell fate in the seam-cell lineage and adult alae formation during post-embryonic development [35–37]. In this paper, analysis of a non-null allele of elt-1 identified from a random mutagenesis screen demonstrates that elt-1 is heterochronic gene that acts in parallel to the nuclear-hormone receptor daf-12 to provide essential regulation of late-larval stage-specific cell fates. Therefore, a genetic screen in a sensitized background with isolation of a partial loss-of-function allele allowed us to genetically separate the post-embryonic roles for elt-1 from its role in embryonic development; the role of elt-1/GATA in developmental timing was previously masked due to both pleiotropism and genetic redundancy.
The phenotype of elt-1(ku491); daf-12(rh61rh411) double-mutant animals is during late developmental stages, with seam cell proliferation during the L4 and Young Adult stages and an L4 bursting vulva phenotype (Figs. 1–2, Table 1). Epistasis analysis (Table 2) shows that the heterochronic phenotypes of elt-1(ku491); daf-12(rh61rh411) mutants require the function of the heterochronic gene network. In addition, the seam-cell proliferation defects, but not bursting-vulva phenotype, can be suppressed by knock-down of genes previously shown to regulate seam-cell maintenance or fate downstream of the heterochronic gene network [40–42]. For genes with partial suppression, such as mab-10 and kin-20, this would seem most likely due to premature adoption of later cell fates (suppressing the seam-cell phenotype) but without precocious expression of LET-7 family miRNAs (to suppress the bursting-vulva phenotype). These data suggest that the molecular defect in elt-1(ku491); daf-12(rh61rh411) double-mutant animals may be in the expression of an L4-specific regulatory factor. During the L4 stage, the major target of heterochronic miRs is the lin-41 mRNA [3,8], and the down regulation of lin-41 mRNA that occurs during that stage has previously been shown to require LET-7 [13]. Indeed, the RT-qPCR analysis of lin-41 mRNA levels during L4 presented here (Fig. 3A) is consistent with a defect in elt-1(ku491); daf-12(rh61rh411) double-mutant animals at the level of an L4-stage-specific regulatory factor that negatively regulates the lin-41 mRNA.
This L4-stage-specific regulatory factor may, in fact, be the LET-7 miRNA, as elt-1(ku491); daf-12(rh61rh411) double-mutant animals fail to down-regulate the lacZ::lin-41 3’UTR reporter during L4 (Fig. 3B-J) and have decreased expression of the LET-7 miR as measured by RT-qPCR (Fig. 4A). The regulation of lin-41 mRNA by LET-7 has previously been shown to be essential for L4-stage-specific developmental progression [3] and the phenotype of elt-1(ku491); daf-12(rh61rh411) double-mutant animals is consistent with that expected from reduced expression of LET-7. These data indicate that elt-1 promotes LET-7 expression during the L4 stage, a novel and unexpected finding. While the LET-7 miR qPCR data (Fig. 4A) are limited by noise likely intrinsic to the time of the measurement, all of the data, including the phenotypes, mRNA qPCR and lacZ staining, are consistent with the interpretation that elt-1 and daf-12 each provide redundant regulation of LET-7 that is required for its L4-stage-specific expression.
However, the decreased expression of LET-7 alone is unlikely to be the sole cause of the developmental timing phenotypes (Figs. 1, 2A, S1, and Table 1) or defective L4-stage down-regulation of lin-41 mRNA seen in the elt-1(ku491); daf-12(rh61rh411) double-mutant animals (Fig. 3A-J), as LET-7 is expressed at a similarly-decreased level in the elt-1(ku491); daf-12(rh61rh411) double mutants as in each of the single-mutant strains (Fig. 4A), which lack strong developmental timing phenotypes and correctly down-regulate lin-41 mRNA (Fig. 3A-B). However, the three LET-7 family miRNAs (miR-48, miR-84, and miR-241) all have a statistically-significant decrease in their expression during the L4 stage in the elt-1(ku491); daf-12(rh61rh411) double-mutant animals but not in either single-mutant, which likely accounts for the differences in the phenotypes and data. This suggests that both daf-12 and elt-1 promote the expression of miR-48, miR-84, and miR-241, but that this regulation is highly redundant, so that either transcription factor alone is sufficient to promote sufficient expression of the miRs to prevent a gross phenotype in the single-mutant strains, despite defective expression of LET-7 in those single-mutant animals. Fig. 5 is a proposed model for the role of elt-1 in the regulation miRNA expression in the heterochronic gene network. These data are also consistent with previous studies showing that daf-12 regulates developmental progression [25,26] and the expression of miR-48, miR-84, miR-241 and LET-7 at the L2 molt [28] and L3 stages [27].
Fig. 5. elt-1/GATA promotes the expression of multiple miRNAs that have key roles in the developmental timing regulatory pathway. 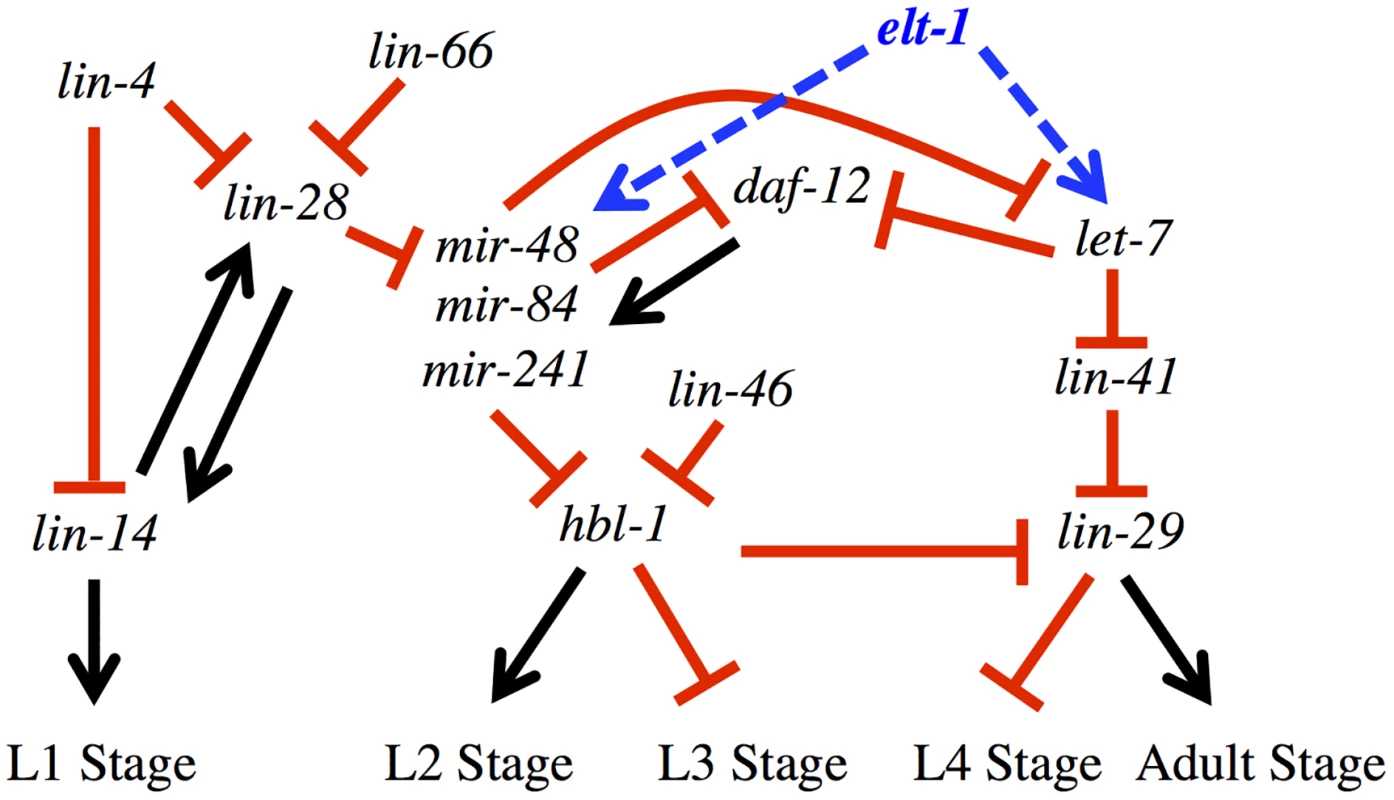
Proposed role for elt-1/GATA in the heterochronic gene regulatory network. Arrows and T-bars indicated positive and negative regulatory relationships, respectively. Model adapted from Resnick, McCulloch, and Rougvie (2010) [7]. A recent study of transcription factor binding sites in C. elegans included ELT-1 [45], and analysis of their data (Table 3) shows that the ELT-1 protein binds the likely promoter region of the DNA sequences encoding all members of the LET-7 miRNA family (mir-48, mir-84, mir-241, let-7), supporting the idea that ELT-1 directly regulates the transcription of the let-7 family miRNAs during late larval development. Our independent analysis confirmed ELT-1 binding in the let-7 promoter region (Fig. 4B). This binding site is 1.6kb upstream of the previously known temporal regulatory element (TRE) [32], so it would be of interest to determine the relationship between the ELT-1 binding site and the TRE in the regulation of let-7 expression.
ELT-1 also has binding sites near the lin-41, lin-28, and hbl-1 genes during the L3 stage, but it is unclear whether these sites are functionally significant, as lin-28 and hbl-1 appear to be normally expressed in the elt-1(ku491) mutants, and the abnormal expression of lin-41 in elt-1 mutants appears to be due to defective down-regulation of its 3’UTR. Intriguingly, ELT-1 protein is expressed in the hypodermis during embryonic development [46] and seam cells during post-embryonic development [36,37], but it remains unclear why it only promotes the expression of LET-7 family miRNAs during late larval stages, rather than throughout development. Perhaps it interacts with other stage-specific transcription factors, undergoes stage-specific post-translational modifications, or its binding sites near those genes are masked or unmasked in a stage-specific manner.
In summary, the elt-1/GATA(ku491) allele described in this study has uncovered a function for elt-1 in regulating miRNA expression and developmental timing that was previously masked by pleiotropism and genetic redundancy. This forcefully supports the idea that the robust expression of key developmental timing genes comes from regulation by parallel and redundant regulatory mechanisms. Similar mechanisms of robustness may also be important in regulating miRNA expression in other organisms during critical developmental transitions, such as in the differentiation of stem cells and in the maintenance of differentiated cell states.
Materials and Methods
Nematode methods
Worms were maintained at 20°C and handled as previously described [47]. Additional information can be found in the S1 Text.
Phenotype assessment
Phenotypes were scored, and feeding RNAi was performed starting at the L1 stage, as previously described [1,2,33,48]. Statistical comparisons of seam cell phenotypes were performed with Prism 6 using one-way ANOVAs with p-values calculated with Bonferroni’s multiple comparisons method. For L4 bursting vulva rate, data was analyzed with Prism 6 and p-values were calculated by 2-tailed binomial t-test. Additional information can be found in the S1 Text.
Quantitative measurement of mRNA and microRNA expression levels
Stage-specific samples were prepared by picking individual worms from mixed-stage plates based on gross appearance and vulval morphology; 50–100 animals were collected per sample. RT-qPCR was performed as previously described [33,49,50] with normalization to eef-2. Expression of miRNAs was measured from the same RNA samples using TaqMan miRNA assay kits (Invitrogen Corp.) with normalization to the snoRNA U18, as recommended by the manufacturer. Statistical analysis was performed with Prism 6 (GraphPad) using 2-way ANOVA with p-values calculated using Sidak’s multiple comparisons test. Additional information can be found in the S1 Text.
Expression of an in vivo reporter of regulation on the lin-41 3’ untranslated region
The pkIs2084 integrated reporter [3,44] was obtained and crossed into the indicated strains. Staining for lacZ activity was performed as described [51]; saturated staining at any point in the animal was scored as strong positive, visible but unsaturated staining as weak positive, and undetectable staining as negative.
Analysis of modENCODE ELT-1::GFP ChIP-sequencing data
modENCODE data sets with stage-specific ChIP-sequencing of an ELT-1::GFP array in strain OP354 at the L2 and L3 stage (modENCODE data coordinating center identifying numbers 4632 and 3843, respectively) were obtained from the website (listed below) and examined for transcription factor sites near selected genes [45,52,53]. The Blacklist Filtered Peak Calls file was used for analysis and is available online at https://www.encodeproject.org/comparative/regulation/#Wormset7.
Chromatin Immunoprecipitation (ChIP) analysis
OP354 strain (unc-119(tm4063); wgIs354 [elt-1::TY1::EGFP::3xFLAG + unc-119(+)]) was synchronized by bleaching and collected at the L3-L4 stages. The ChIP experiment was performed as described previously [54] with minor modifications. Briefly, paraformaldehyde-fixed chromatin was immunoprecipitated with either mouse IgG (Jackson immunoresearch) complexed with Protein G beads (GE healthcare) or TrapA GFP beads (Chromotek). Following extraction of the immunoprecipitated DNA, qPCR was performed according to the manufacturer’s instruction (Bioneer). Primers specific to let-7 promoter region are as follows (5’-3’): forward primer, TCTCACTGTGTGTCAGCCG, and reverse primer, TGCTGACGTACTACCGGTGC5. The result was normalized to the level of 3’Untranlsated Region of let-7 gene completed from the same immune complexes using the following primers (5’-TCGATCTCTGTCCGCTTTGAAAC-3’, 5’-CAGGAGGTGAAGAACGAGCA-3’).
Supporting Information
Zdroje
1. Wightman B, Ha I, Ruvkun G (1993) Posttranscriptional regulation of the heterochronic gene lin-14 by lin-4 mediates temporal pattern formation in C. elegans. Cell 75 : 855–862. 8252622
2. Lee RC, Feinbaum RL, Ambros V (1993) The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 75 : 843–854. 8252621
3. Reinhart BJ, Slack FJ, Basson M, Pasquinelli a E, Bettinger JC, et al. (2000) The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans. Nature 403 : 901–906. 10706289
4. Lin S-Y, Johnson SM, Abraham M, Vella MC, Pasquinelli A, et al. (2003) The C elegans hunchback homolog, hbl-1, controls temporal patterning and is a probable microRNA target. Dev Cell 4 : 639–650. 12737800
5. Moss EG, Lee RC, Ambros V (1997) The cold shock domain protein LIN-28 controls developmental timing in C. elegans and is regulated by the lin-4 RNA. Cell 88 : 637–646. 9054503
6. Ambros V (2000) Control of developmental timing in Caenorhabditis elegans. Curr Opin Genet Dev 10 : 428–433. 10889059
7. Resnick TD, McCulloch K a, Rougvie AE (2010) miRNAs give worms the time of their lives: small RNAs and temporal control in Caenorhabditis elegans. Dev Dyn 239 : 1477–1489. doi: 10.1002/dvdy.22260 20232378
8. Ambros V (2011) MicroRNAs and developmental timing. Curr Opin Genet Dev 21 : 511–517. doi: 10.1016/j.gde.2011.04.003 21530229
9. Piskounova E, Polytarchou C, Thornton JE, LaPierre RJ, Pothoulakis C, et al. (2011) Lin28A and Lin28B inhibit let-7 microRNA biogenesis by distinct mechanisms. Cell 147 : 1066–1079. doi: 10.1016/j.cell.2011.10.039 22118463
10. Heo I, Joo C, Cho J, Ha M, Han J, et al. (2008) Lin28 mediates the terminal uridylation of let-7 precursor MicroRNA. Mol Cell 32 : 276–284. doi: 10.1016/j.molcel.2008.09.014 18951094
11. Van Wynsberghe PM, Kai ZS, Massirer KB, Burton VH, Yeo GW, et al. (2011) LIN-28 co-transcriptionally binds primary let-7 to regulate miRNA maturation in Caenorhabditis elegans. Nat Struct Mol Biol 18 : 302–308. doi: 10.1038/nsmb.1986 21297634
12. Vella MC, Choi E-Y, Lin S-Y, Reinert K, Slack FJ (2004) The C. elegans microRNA let-7 binds to imperfect let-7 complementary sites from the lin-41 3’UTR. Genes Dev 18 : 132–137. 14729570
13. Slack FJ, Basson M, Liu Z, Ambros V, Horvitz HR, et al. (2000) The lin-41 RBCC gene acts in the C. elegans heterochronic pathway between the let-7 regulatory RNA and the LIN-29 transcription factor. Mol Cell 5 : 659–669. 10882102
14. Yu F, Yao H, Zhu P, Zhang X, Pan Q, et al. (2007) let-7 regulates self renewal and tumorigenicity of breast cancer cells. Cell 131 : 1109–1123. 18083101
15. Roush S, Slack FJ (2008) The let-7 family of microRNAs. Trends Cell Biol 18 : 505–516. doi: 10.1016/j.tcb.2008.07.007 18774294
16. Tocchini C, Keusch JJ, Miller SB, Finger S, Gut H, et al. (2014) The TRIM-NHL protein LIN-41 controls the onset of developmental plasticity in Caenorhabditis elegans. PLoS Genet 10: e1004533. doi: 10.1371/journal.pgen.1004533 25167051
17. Worringer KA, Rand TA, Hayashi Y, Sami S, Takahashi K, et al. (2014) The let-7/LIN-41 pathway regulates reprogramming to human induced pluripotent stem cells by controlling expression of prodifferentiation genes. Cell Stem Cell 14 : 40–52. doi: 10.1016/j.stem.2013.11.001 24239284
18. Yu J, Vodyanik M a, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, et al. (2007) Induced pluripotent stem cell lines derived from human somatic cells. Science 318 : 1917–1920. 18029452
19. Daley GQ (2012) Cellular alchemy and the golden age of reprogramming. Cell 151 : 1151–1154. doi: 10.1016/j.cell.2012.11.016 23217698
20. Madison BB, Liu Q, Zhong X, Hahn CM, Lin N, et al. (2013) LIN28B promotes growth and tumorigenesis of the intestinal epithelium via Let-7. Genes Dev 27 : 2233–2245. doi: 10.1101/gad.224659.113 24142874
21. Shyh-Chang N, Zhu H, Yvanka de Soysa T, Shinoda G, Seligson MT, et al. (2013) Lin28 enhances tissue repair by reprogramming cellular metabolism. Cell 155 : 778–792. doi: 10.1016/j.cell.2013.09.059 24209617
22. Viswanathan SR, Powers JT, Einhorn W, Hoshida Y, Ng TL, et al. (2009) Lin28 promotes transformation and is associated with advanced human malignancies. Nat Genet 41 : 843–848. doi: 10.1038/ng.392 19483683
23. Urbach A, Yermalovich A, Zhang J, Spina CS, Zhu H, et al. (2014) Lin28 sustains early renal progenitors and induces Wilms tumor. Genes Dev 28 : 971–982. doi: 10.1101/gad.237149.113 24732380
24. Motola DL, Cummins CL, Rottiers V, Sharma KK, Li T, et al. (2006) Identification of ligands for DAF-12 that govern dauer formation and reproduction in C. elegans. Cell 124 : 1209–1223. 16529801
25. Antebi A, Culotti JG, Hedgecock EM (1998) daf-12 regulates developmental age and the dauer alternative in Caenorhabditis elegans. Development 125 : 1191–1205. 9477318
26. Antebi A, Yeh WH, Tait D, Hedgecock EM, Riddle DL (2000) daf-12 encodes a nuclear receptor that regulates the dauer diapause and developmental age in C. elegans. Genes Dev 14 : 1512–1527. 10859169
27. Bethke A, Fielenbach N, Wang Z, Mangelsdorf DJ, Antebi A (2009) Nuclear hormone receptor regulation of microRNAs controls developmental progression. Science 324 : 95–98. doi: 10.1126/science.1164899 19342589
28. Hammell CM, Karp X, Ambros V (2009) A feedback circuit involving let-7-family miRNAs and DAF-12 integrates environmental signals and developmental timing in Caenorhabditis elegans. Proc Natl Acad Sci U S A 106 : 18668–18673. doi: 10.1073/pnas.0908131106 19828440
29. Hochbaum D, Zhang Y, Stuckenholz C, Labhart P, Alexiadis V, et al. (2011) DAF-12 regulates a connected network of genes to ensure robust developmental decisions. PLoS Genet 7: e1002179. doi: 10.1371/journal.pgen.1002179 21814518
30. Abbott AL, Alvarez-Saavedra E, Miska EA, Lau NC, Bartel DP, et al. (2005) The let-7 MicroRNA family members mir-48, mir-84, and mir-241 function together to regulate developmental timing in Caenorhabditis elegans. Dev Cell 9 : 403–414. 16139228
31. Zisoulis DG, Kai ZS, Chang RK, Pasquinelli AE (2012) Autoregulation of microRNA biogenesis by let-7 and Argonaute. Nature 486 : 541–544. doi: 10.1038/nature11134 22722835
32. Johnson SM, Lin SY, Slack FJ (2003) The time of appearance of the C. elegans let-7 microRNA is transcriptionally controlled utilizing a temporal regulatory element in its promoter. Dev Biol 259 : 364–379. 12871707
33. Morita K, Han M (2006) Multiple mechanisms are involved in regulating the expression of the developmental timing regulator lin-28 in Caenorhabditis elegans. EMBO J 25 : 5794–5804. 17139256
34. Perales R, King DM, Aguirre-Chen C, Hammell CM (2014) LIN-42, the Caenorhabditis elegans PERIOD homolog, negatively regulates microRNA transcription. PLoS Genet 10: e1004486. doi: 10.1371/journal.pgen.1004486 25032706
35. Brabin C, Appleford PJ, Woollard A (2011) The Caenorhabditis elegans GATA factor ELT-1 works through the cell proliferation regulator BRO-1 and the Fusogen EFF-1 to maintain the seam stem-like fate. PLoS Genet 7: e1002200. doi: 10.1371/journal.pgen.1002200 21829390
36. Gilleard JS, McGhee JD (2001) Activation of hypodermal differentiation in the Caenorhabditis elegans embryo by GATA transcription factors ELT-1 and ELT-3. Mol Cell Biol 21 : 2533–2544. 11259601
37. Smith J a, McGarr P, Gilleard JS (2005) The Caenorhabditis elegans GATA factor elt-1 is essential for differentiation and maintenance of hypodermal seam cells and for normal locomotion. J Cell Sci 118 : 5709–5719. 16303852
38. Spieth J, Shim YH, Lea K, Conrad R, Blumenthal T (1991) elt-1, an embryonically expressed Caenorhabditis elegans gene homologous to the GATA transcription factor family. Mol Cell Biol 11 : 4651–4659. 1875944
39. Wilkinson-White LE, Ripin N, Jacques DA, Guss JM, Matthews JM (2012) Both Zn Fingers of GATA1 Bound to Palindromic DNA Recognition Site, P21 Crystal Form: PDB ID: 3VD6. Available: http://www.rcsb.org/pdb/.
40. Cassata G, Shemer G, Morandi P, Donhauser R, Podbilewicz B, et al. (2005) ceh-16/engrailed patterns the embryonic epidermis of Caenorhabditis elegans. Development 132 : 739–749. 15659483
41. Huang X, Tian E, Xu Y, Zhang H (2009) The C. elegans engrailed homolog ceh-16 regulates the self-renewal expansion division of stem cell-like seam cells. Dev Biol 333 : 337–347. doi: 10.1016/j.ydbio.2009.07.005 19607822
42. Banerjee D, Kwok A, Lin S-Y, Slack FJ (2005) Developmental timing in C. elegans is regulated by kin-20 and tim-1, homologs of core circadian clock genes. Dev Cell 8 : 287–295. 15691769
43. Fielenbach N, Guardavaccaro D, Neubert K, Chan T, Li D, et al. (2007) DRE-1: an evolutionarily conserved F box protein that regulates C. elegans developmental age. Dev Cell 12 : 443–455. 17336909
44. Denli AM, Tops BBJ, Plasterk RH a, Ketting RF, Hannon GJ (2004) Processing of primary microRNAs by the Microprocessor complex. Nature 432 : 231–235. 15531879
45. Araya CL, Kawli T, Kundaje A, Jiang L, Wu B, et al. (2014) Regulatory analysis of the C. elegans genome with spatiotemporal resolution. Nature 512 : 400–405. doi: 10.1038/nature13497 25164749
46. Page BD, Zhang W, Steward K, Blumenthal T, Priess JR (1997) ELT-1, a GATA-like transcription factor, is required for epidermal cell fates in Caenorhabditis elegans embryos. Genes Dev 11 : 1651–1661. 9224715
47. Brenner S (1974) The Genetics of Caenorhabditis elegans. Genetics 77 : 71–94. 4366476
48. Kamath RS, Martinez-Campos M, Zipperlen P, Fraser AG, Ahringer J (2001) Effectiveness of specific RNA-mediated interference through ingested double-stranded RNA in Caenorhabditis elegans. Genome Biol 2.
49. Bagga S, Bracht J, Hunter S, Massirer K, Holtz J, et al. (2005) Regulation by let-7 and lin-4 miRNAs results in target mRNA degradation. Cell 122 : 553–563. 16122423
50. Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25 : 402–408. 11846609
51. Fire A (1992) Histochemical techniques for locating Escherichia coli beta-galactosidase activity in transgenic organisms. Genet Anal Tech Appl 9 : 151–158. 1296710
52. Contrino S, Smith RN, Butano D, Carr A, Hu F, et al. (2012) modMine: flexible access to modENCODE data. Nucleic Acids Res 40: D1082–D1088. doi: 10.1093/nar/gkr921 22080565
53. Gerstein MB, Lu ZJ, Van Nostrand EL, Cheng C, Arshinoff BI, et al. (2010) Integrative analysis of the Caenorhabditis elegans genome by the modENCODE project. Science 330 : 1775–1787. doi: 10.1126/science.1196914 21177976
54. Mukhopadhyay A, Deplancke B, Walhout AJM, Tissenbaum HA (2008) Chromatin immunoprecipitation (ChIP) coupled to detection by quantitative real-time PCR to study transcription factor binding to DNA in Caenorhabditis elegans. Nat Protoc 3 : 698–709. doi: 10.1038/nprot.2008.38 18388953
Štítky
Genetika Reprodukční medicína
Článek NLRC5 Exclusively Transactivates MHC Class I and Related Genes through a Distinctive SXY ModuleČlánek Inhibition of Telomere Recombination by Inactivation of KEOPS Subunit Cgi121 Promotes Cell LongevityČlánek HOMER2, a Stereociliary Scaffolding Protein, Is Essential for Normal Hearing in Humans and MiceČlánek LRGUK-1 Is Required for Basal Body and Manchette Function during Spermatogenesis and Male FertilityČlánek Systems Biology of Tissue-Specific Response to Reveals Differentiated Apoptosis in the Tick VectorČlánek Phenotype Specific Analyses Reveal Distinct Regulatory Mechanism for Chronically Activated p53Článek The Nuclear Receptor DAF-12 Regulates Nutrient Metabolism and Reproductive Growth in NematodesČlánek The ATM Signaling Cascade Promotes Recombination-Dependent Pachytene Arrest in Mouse SpermatocytesČlánek The Small Protein MntS and Exporter MntP Optimize the Intracellular Concentration of Manganese
Článek vyšel v časopisePLOS Genetics
Nejčtenější tento týden
2015 Číslo 3
-
Všechny články tohoto čísla
- NLRC5 Exclusively Transactivates MHC Class I and Related Genes through a Distinctive SXY Module
- Licensing of Primordial Germ Cells for Gametogenesis Depends on Genital Ridge Signaling
- A Genomic Duplication is Associated with Ectopic Eomesodermin Expression in the Embryonic Chicken Comb and Two Duplex-comb Phenotypes
- Genome-wide Association Study and Meta-Analysis Identify as Genome-wide Significant Susceptibility Gene for Bladder Exstrophy
- Mutations of Human , Encoding the Mitochondrial Asparaginyl-tRNA Synthetase, Cause Nonsyndromic Deafness and Leigh Syndrome
- Exome Sequencing in an Admixed Isolated Population Indicates Variants Confer a Risk for Specific Language Impairment
- Genome-Wide Association Studies in Dogs and Humans Identify as a Risk Variant for Cleft Lip and Palate
- Rapid Evolution of Recombinant for Xylose Fermentation through Formation of Extra-chromosomal Circular DNA
- The Ribosome Biogenesis Factor Nol11 Is Required for Optimal rDNA Transcription and Craniofacial Development in
- Methyl Farnesoate Plays a Dual Role in Regulating Metamorphosis
- Maternal Co-ordinate Gene Regulation and Axis Polarity in the Scuttle Fly
- Maternal Filaggrin Mutations Increase the Risk of Atopic Dermatitis in Children: An Effect Independent of Mutation Inheritance
- Inhibition of Telomere Recombination by Inactivation of KEOPS Subunit Cgi121 Promotes Cell Longevity
- Clonality and Evolutionary History of Rhabdomyosarcoma
- HOMER2, a Stereociliary Scaffolding Protein, Is Essential for Normal Hearing in Humans and Mice
- Methylation-Sensitive Expression of a DNA Demethylase Gene Serves As an Epigenetic Rheostat
- BREVIPEDICELLUS Interacts with the SWI2/SNF2 Chromatin Remodeling ATPase BRAHMA to Regulate and Expression in Control of Inflorescence Architecture
- Seizures Are Regulated by Ubiquitin-specific Peptidase 9 X-linked (USP9X), a De-Ubiquitinase
- The Fun30 Chromatin Remodeler Fft3 Controls Nuclear Organization and Chromatin Structure of Insulators and Subtelomeres in Fission Yeast
- A Cascade of Iron-Containing Proteins Governs the Genetic Iron Starvation Response to Promote Iron Uptake and Inhibit Iron Storage in Fission Yeast
- Mutation in MRPS34 Compromises Protein Synthesis and Causes Mitochondrial Dysfunction
- LRGUK-1 Is Required for Basal Body and Manchette Function during Spermatogenesis and Male Fertility
- Cis-Regulatory Mechanisms for Robust Olfactory Sensory Neuron Class-restricted Odorant Receptor Gene Expression in
- Effects on Murine Behavior and Lifespan of Selectively Decreasing Expression of Mutant Huntingtin Allele by Supt4h Knockdown
- HDAC4-Myogenin Axis As an Important Marker of HD-Related Skeletal Muscle Atrophy
- A Conserved Domain in the Scc3 Subunit of Cohesin Mediates the Interaction with Both Mcd1 and the Cohesin Loader Complex
- Selective and Genetic Constraints on Pneumococcal Serotype Switching
- Bacterial Infection Drives the Expression Dynamics of microRNAs and Their isomiRs
- The GATA Factor Regulates . Developmental Timing by Promoting Expression of the Family MicroRNAs
- Accumulation of Glucosylceramide in the Absence of the Beta-Glucosidase GBA2 Alters Cytoskeletal Dynamics
- Reproductive Isolation of Hybrid Populations Driven by Genetic Incompatibilities
- The Contribution of Alu Elements to Mutagenic DNA Double-Strand Break Repair
- Systems Biology of Tissue-Specific Response to Reveals Differentiated Apoptosis in the Tick Vector
- Tfap2a Promotes Specification and Maturation of Neurons in the Inner Ear through Modulation of Bmp, Fgf and Notch Signaling
- The Lysine Acetyltransferase Activator Brpf1 Governs Dentate Gyrus Development through Neural Stem Cells and Progenitors
- PHABULOSA Controls the Quiescent Center-Independent Root Meristem Activities in
- DNA Polymerase ζ-Dependent Lesion Bypass in Is Accompanied by Error-Prone Copying of Long Stretches of Adjacent DNA
- Examining the Evolution of the Regulatory Circuit Controlling Secondary Metabolism and Development in the Fungal Genus
- Zinc Finger Independent Genome-Wide Binding of Sp2 Potentiates Recruitment of Histone-Fold Protein Nf-y Distinguishing It from Sp1 and Sp3
- GAGA Factor Maintains Nucleosome-Free Regions and Has a Role in RNA Polymerase II Recruitment to Promoters
- Neurospora Importin α Is Required for Normal Heterochromatic Formation and DNA Methylation
- Ccr4-Not Regulates RNA Polymerase I Transcription and Couples Nutrient Signaling to the Control of Ribosomal RNA Biogenesis
- Phenotype Specific Analyses Reveal Distinct Regulatory Mechanism for Chronically Activated p53
- A Systems-Level Interrogation Identifies Regulators of Blood Cell Number and Survival
- Morphological Mutations: Lessons from the Cockscomb
- Genetic Interaction Mapping Reveals a Role for the SWI/SNF Nucleosome Remodeler in Spliceosome Activation in Fission Yeast
- The Role of China in the Global Spread of the Current Cholera Pandemic
- The Nuclear Receptor DAF-12 Regulates Nutrient Metabolism and Reproductive Growth in Nematodes
- A Zinc Finger Motif-Containing Protein Is Essential for Chloroplast RNA Editing
- Resistance to Gray Leaf Spot of Maize: Genetic Architecture and Mechanisms Elucidated through Nested Association Mapping and Near-Isogenic Line Analysis
- Small Regulatory RNA-Induced Growth Rate Heterogeneity of
- Mitochondrial Dysfunction Reveals the Role of mRNA Poly(A) Tail Regulation in Oculopharyngeal Muscular Dystrophy Pathogenesis
- Complex Genomic Rearrangements at the Locus Include Triplication and Quadruplication
- Male-Biased Aganglionic Megacolon in the TashT Mouse Line Due to Perturbation of Silencer Elements in a Large Gene Desert of Chromosome 10
- Sex Ratio Meiotic Drive as a Plausible Evolutionary Mechanism for Hybrid Male Sterility
- Tertiary siRNAs Mediate Paramutation in .
- RECG Maintains Plastid and Mitochondrial Genome Stability by Suppressing Extensive Recombination between Short Dispersed Repeats
- Escape from X Inactivation Varies in Mouse Tissues
- Opposite Phenotypes of Muscle Strength and Locomotor Function in Mouse Models of Partial Trisomy and Monosomy 21 for the Proximal Region
- Glycosyl Phosphatidylinositol Anchor Biosynthesis Is Essential for Maintaining Epithelial Integrity during Embryogenesis
- Hyperdiverse Gene Cluster in Snail Host Conveys Resistance to Human Schistosome Parasites
- The Class Homeodomain Factors and Cooperate in . Embryonic Progenitor Cells to Regulate Robust Development
- Recombination between Homologous Chromosomes Induced by Unrepaired UV-Generated DNA Damage Requires Mus81p and Is Suppressed by Mms2p
- Synergistic Interactions between Orthologues of Genes Spanned by Human CNVs Support Multiple-Hit Models of Autism
- Gene Networks Underlying Convergent and Pleiotropic Phenotypes in a Large and Systematically-Phenotyped Cohort with Heterogeneous Developmental Disorders
- The ATM Signaling Cascade Promotes Recombination-Dependent Pachytene Arrest in Mouse Spermatocytes
- Combinatorial Control of Light Induced Chromatin Remodeling and Gene Activation in
- Linking Aβ42-Induced Hyperexcitability to Neurodegeneration, Learning and Motor Deficits, and a Shorter Lifespan in an Alzheimer’s Model
- The Complex Contributions of Genetics and Nutrition to Immunity in
- NatB Domain-Containing CRA-1 Antagonizes Hydrolase ACER-1 Linking Acetyl-CoA Metabolism to the Initiation of Recombination during . Meiosis
- Transcriptomic Profiling of Reveals Reprogramming of the Crp Regulon by Temperature and Uncovers Crp as a Master Regulator of Small RNAs
- Osteopetrorickets due to Snx10 Deficiency in Mice Results from Both Failed Osteoclast Activity and Loss of Gastric Acid-Dependent Calcium Absorption
- A Genomic Portrait of Haplotype Diversity and Signatures of Selection in Indigenous Southern African Populations
- Sequence Features and Transcriptional Stalling within Centromere DNA Promote Establishment of CENP-A Chromatin
- Inhibits Neuromuscular Junction Growth by Downregulating the BMP Receptor Thickveins
- Replicative DNA Polymerase δ but Not ε Proofreads Errors in and in
- Unsaturation of Very-Long-Chain Ceramides Protects Plant from Hypoxia-Induced Damages by Modulating Ethylene Signaling in
- The Small Protein MntS and Exporter MntP Optimize the Intracellular Concentration of Manganese
- A Meta-analysis of Gene Expression Signatures of Blood Pressure and Hypertension
- Pervasive Variation of Transcription Factor Orthologs Contributes to Regulatory Network Evolution
- Network Analyses Reveal Novel Aspects of ALS Pathogenesis
- A Role for the Budding Yeast Separase, Esp1, in Ty1 Element Retrotransposition
- Nab3 Facilitates the Function of the TRAMP Complex in RNA Processing via Recruitment of Rrp6 Independent of Nrd1
- A RecA Protein Surface Required for Activation of DNA Polymerase V
- PLOS Genetics
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Clonality and Evolutionary History of Rhabdomyosarcoma
- Morphological Mutations: Lessons from the Cockscomb
- Maternal Filaggrin Mutations Increase the Risk of Atopic Dermatitis in Children: An Effect Independent of Mutation Inheritance
- Transcriptomic Profiling of Reveals Reprogramming of the Crp Regulon by Temperature and Uncovers Crp as a Master Regulator of Small RNAs
Přihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



