-
Články
Top novinky
Reklama- Vzdělávání
- Časopisy
Top články
Nové číslo
- Témata
Top novinky
Reklama- Videa
- Podcasty
Nové podcasty
Reklama- Práce v oboru
Doporučené pozice
Reklama- Praktické
Top novinky
ReklamaSialidases Affect the Host Cell Adherence and Epsilon Toxin-Induced Cytotoxicity of Type D Strain CN3718
Clostridium perfringens type B or D isolates, which cause enterotoxemias or enteritis in livestock, produce epsilon toxin (ETX). ETX is exceptionally potent, earning it a listing as a CDC class B select toxin. Most C. perfringens strains also express up to three different sialidases, although the possible contributions of those enzymes to type B or D pathogenesis remain unclear. Type D isolate CN3718 was found to carry two genes (nanI and nanJ) encoding secreted sialidases and one gene (nanH) encoding a cytoplasmic sialidase. Construction in CN3718 of single nanI, nanJ and nanH null mutants, as well as a nanI/nanJ double null mutant and a triple sialidase null mutant, identified NanI as the major secreted sialidase of this strain. Pretreating MDCK cells with NanI sialidase, or with culture supernatants of BMC206 (an isogenic CN3718 etx null mutant that still produces sialidases) enhanced the subsequent binding and cytotoxic effects of purified ETX. Complementation of BMC207 (an etx/nanH/nanI/nanJ null mutant) showed this effect is mainly attributable to NanI production. Contact between BMC206 and certain mammalian cells (e.g., enterocyte-like Caco-2 cells) resulted in more rapid sialidase production and this effect involved increased transcription of BMC206 nanI gene. BMC206 was shown to adhere to some (e.g. Caco-2 cells), but not all mammalian cells, and this effect was dependent upon sialidase, particularly NanI, expression. Finally, the sialidase activity of NanI (but not NanJ or NanH) could be enhanced by trypsin. Collectively these in vitro findings suggest that, during type D disease originating in the intestines, trypsin may activate NanI, which (in turn) could contribute to intestinal colonization by C. perfringens type D isolates and also increase ETX action.
Published in the journal: . PLoS Pathog 7(12): e32767. doi:10.1371/journal.ppat.1002429
Category: Research Article
doi: https://doi.org/10.1371/journal.ppat.1002429Summary
Clostridium perfringens type B or D isolates, which cause enterotoxemias or enteritis in livestock, produce epsilon toxin (ETX). ETX is exceptionally potent, earning it a listing as a CDC class B select toxin. Most C. perfringens strains also express up to three different sialidases, although the possible contributions of those enzymes to type B or D pathogenesis remain unclear. Type D isolate CN3718 was found to carry two genes (nanI and nanJ) encoding secreted sialidases and one gene (nanH) encoding a cytoplasmic sialidase. Construction in CN3718 of single nanI, nanJ and nanH null mutants, as well as a nanI/nanJ double null mutant and a triple sialidase null mutant, identified NanI as the major secreted sialidase of this strain. Pretreating MDCK cells with NanI sialidase, or with culture supernatants of BMC206 (an isogenic CN3718 etx null mutant that still produces sialidases) enhanced the subsequent binding and cytotoxic effects of purified ETX. Complementation of BMC207 (an etx/nanH/nanI/nanJ null mutant) showed this effect is mainly attributable to NanI production. Contact between BMC206 and certain mammalian cells (e.g., enterocyte-like Caco-2 cells) resulted in more rapid sialidase production and this effect involved increased transcription of BMC206 nanI gene. BMC206 was shown to adhere to some (e.g. Caco-2 cells), but not all mammalian cells, and this effect was dependent upon sialidase, particularly NanI, expression. Finally, the sialidase activity of NanI (but not NanJ or NanH) could be enhanced by trypsin. Collectively these in vitro findings suggest that, during type D disease originating in the intestines, trypsin may activate NanI, which (in turn) could contribute to intestinal colonization by C. perfringens type D isolates and also increase ETX action.
Introduction
Clostridium perfringens, a Gram-positive, spore-forming anaerobe, is an important pathogen of both humans (causing, for example, gas gangrene and type A human food poisoning) and livestock (causing severe enterotoxemias and enteritis) [1]. The virulence of this bacterium is largely attributable to its ability to express a plethora of potent toxins. However, while C. perfringens can produce >15 different toxins, individual strains express only portions of this toxin arsenal [1]-[3]. Therefore, based upon production of four typing toxins (α, β, ε, and ι), isolates of this organism are commonly classified into five toxinotypes (type A through E) [4].
By definition, C. perfringens type D isolates must produce alpha and epsilon toxins, while type B isolates must express alpha, beta and epsilon toxin [4]. Beyond those typing toxins, type D and type B isolates commonly produce additional toxins, e.g., perfringolysin O, enterotoxin, TpeL or beta2 toxin [5]-[7]. Type B and D isolates cause enterotoxemias in livestock that initiate with toxin production in the intestines, followed by absorption of those toxins into the circulation to affect other internal organs, such as the brain and kidneys. Type D isolates can also cause acute or chronic enteritis in goats [1], [8], [9].
Epsilon toxin (ETX) is considered important for the virulence of both type B and type D isolates [5], [6]. This CDC class B select toxin, which ranks as the third most-potent clostridial toxin after the botulinum toxins and tetanus toxin, belongs to the aerolysin family of pore-forming toxins [9], [10]. ETX is synthesized and secreted as an inactive prototoxin of 311 amino acids (32.7 kDa). In the animal intestines, the prototoxin can be proteolytically activated to the fully-active toxin (274 amino acids) by trypsin and chymotrypsin [11].
To date, only a few ETX-sensitive cultured cell lines have been identified, including MDCK cells, mpkCCDc14 cells, and human leiomyoblastoma (G402) cells [11]. The mechanism of ETX action on MDCK cells is still under active study but first involves the binding of this toxin to unidentified protein receptors on the MDCK cell membrane surface. The bound toxin then uses lipid rafts to form a heptameric prepore complex on the membrane surface [12], [13]. When this prepore complex inserts into the cell membrane, an active pore is created that causes, or strongly contributes to, MDCK cell death [9].
Genome sequencing has revealed that C. perfringens strains typically possess three sialidase-encoding genes, named nanH, nanI and nanJ, which are located on a conserved region of the chromosome [14]-[16]. The nanH gene product, which is not secreted, is the ∼43 kDa NanH sialidase. The nanI gene product is the secreted ∼77 kDa NanI sialidase, while the nanJ gene product is the secreted ∼129 kDa NanJ sialidase. The nanH ORF shares only 19% nucleotide sequence identity with nanI and nanJ, but the ORFs encoding the two larger exoenzymes are more closely related, sharing 57% nucleotide sequence identity [17].
Sialidase (neuraminidases) production by some pathogenic bacteria has been implicated in their virulence. For example, Vibrio cholerae sialidase enhances the activity of cholera toxin by modifying surface gangliosides to create additional toxin receptors and thereby increase toxin binding levels [18]. Sialidases can also apparently contribute to virulence in other ways besides enhancing toxin binding. For example, neuramindases are thought to assist Streptococcus pneumoniae pathogenesis by providing nutrients for growth, assisting in biofilm formation, and enhancing colonization by exposing adhesion sites for this bacterium in the airways [19].
Possible sialidase contributions to C. perfringens virulence have received only limited attention. A recent study [20] used mutants to evaluate the potential pathogencity contributions of NanI and NanJ sialidase when C. perfringens type A strain 13 causes clostridial myonecrosis (gas gangrene). That study [20] found sialidases can enhance alpha-toxin-mediated cytotoxic effects in vitro, but also reported that sialidase production is not necessary for strain 13 to cause gas gangrene in a mouse model. Possible virulence contributions of sialidases when other C. perfringens strains, such as type D strains, cause disease originating in the intestines has received even less study. It was reported that sialidases can enhance ETX cytotoxicity towards MDCK cells [21], but an apparently contradictory conclusion was reached by another study determining that treatment of synaptosomal membrane fractions with sialidases lowered ETX binding levels [22].
The current study has used both biochemical and isogenic mutant approaches to better evaluate the possible sialidase enhancement of in vitro ETX action. In addition, a possible role for sialidases in facilitating C. perfringens cell adhesion to host cells was examined for the first time. Results of these in vitro studies suggest that sialidases could contribute to type D pathogenesis via at least two distinct mechanisms.
Results
Pretreatment of MDCK cells with purified C. perfringens NanI sialidase enhances ETX cytotoxicity, binding and complex formation
To clarify whether C. perfringens sialidases can enhance ETX cytotoxicity in sensitive mammalian cells, MDCK cells were pretreated with purified C. perfringens NanI sialidase and, after washing, those cells were challenged with purified ETX. The results showed that sialidase pretreatment substantially increased ETX-induced cytotoxicity (Figure 1A). This enhancement was stronger using a 0.005 U/ml vs. a 0.001 U/ml dose of sialidase for the pretreatment. However, while enhancement of ETX cytotoxicity increased up to a 90 min sialidase pretreatment using the 0.001 U/ml NanI dose, the higher 0.005 U/ml NanI dose caused no further increase in cytotoxicity beyond a 60 min pretreatment (Figure 1A). Sialidase treatment alone, or treatment with prototoxin, did not increase cytotoxicity above background levels (data not shown).
Fig. 1. Pre-treatment of MDCK cells with purified NanI enhances ETX binding and cytotoxicity. 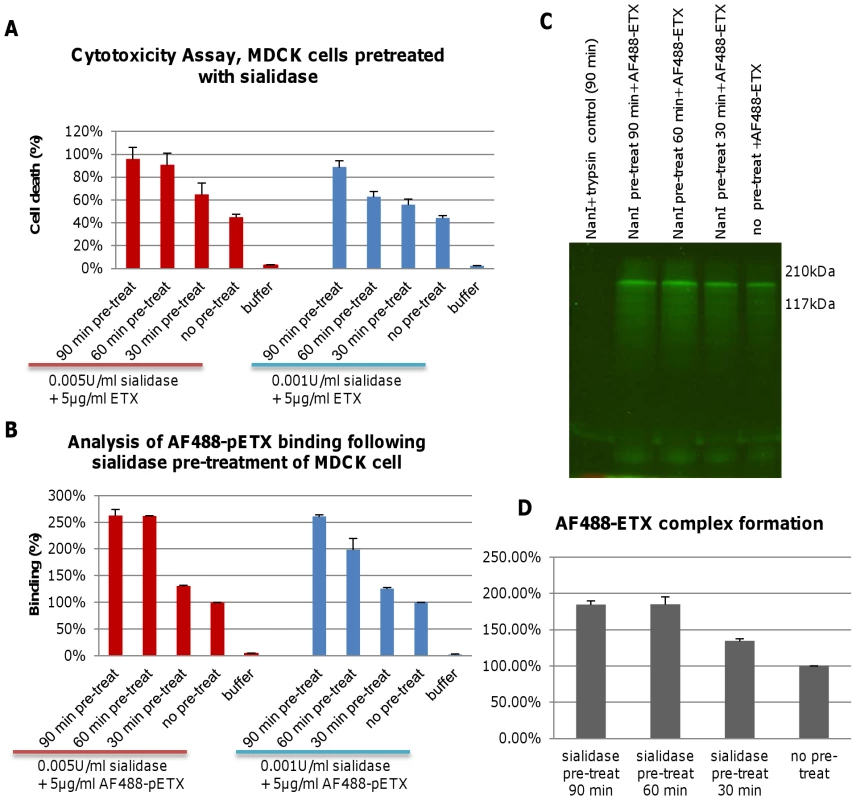
Panel A, cytotoxicity assay of MDCK cells pretreated for the specified times with either 0.001 U/ml or 0.005 U/ml of NanI sialidase and then challenged with 5 µg/ml of purified ETX. Panel B, AF488-pETX binding following the specified sialidase pre-treatment of MDCK cells. AF488-pETX (5 µg/ml) binding to MDCK cells in the absence of any pre-treatment with sialidase was considered 100% binding. Panel C, pre-treatment with purified NanI sialidase enhances AF488-ETX complex formation in, MDCK cells. Lane 1, sialidase and trypsin controls; Lane 2-4, ETX complex formation after sialidase pre-treatment of MDCK cells for 30, 60 or 90 min, followed by treatment with 5 µg/ml of AF488-ETX. Lane 5, ETX complex formation by 5 µg/ml AF488-ETX-treated MDCK cells (no sialidase pretreatment); Molecular weight markers shown on the right. Panel D, quantitative comparison of the effects of NanI pretreatment on AF488-ETX complex formation. Gel regions from panel C experiments containing the ETX complex were scanned for fluorescence using a Typhoon scanner. The results shown in Figure 1 show the average of two (panel D) or three (panel A and B) repetitions; the error bars indicate the standard deviation (S.D.). Experiments were then performed to explore the mechanistic basis behind the sialidase-induced enhancement of ETX cytotoxicity for MDCK cells that was shown in Figure 1A. First, MDCK cells were pretreated with purified C. perfringens NanI sialidase and, after washing, those cells were incubated with Alexa Fluor 488-labeled epsilon prototoxin (AF488-pETX), which was used since it binds similarly as active ETX to MDCK cells, yet causes no cytotoxicity [23]. Results from this experiment (Figure 1B) showed that pretreating MDCK cells with either 0.001 U/ml or 0.005 U/ml of purified NanI sialidase substantially increased subsequent AF488-pETX binding levels. This enhancement of toxin binding increased with longer sialidase pretreatment time, although no further increase in AF488-pETX binding was noted after a 60 min sialidase pretreatment beyond the 0.005 U/ml sialidase dose (Figure 1B).
An experiment then assessed the ability of sialidase pretreatment to increase formation of the ETX oligomeric complex that is considered responsible for pore formation-induced ETX cytotoxicity [9]. SDS-PAGE analyses of MDCK cell lysates treated with 5 µg/ml of Alexa Fluor 488-labeled ETX (AF488-ETX) detected an increased formation of the ETX oligomeric complex after NanI pretreatment (Figure 1C and 1D). This effect increased up to a 60 min pretreatment with 0.005 U/ml of NanI. Lastly, similar NanI pretreatment also increased MDCK cell binding and complex formation using a 10 µg/ml dose of AF488-pETX or AF488-ETX, respectively (data not shown).
Construction and genotypic characterization of isogenic CN3718 sialidase mutants
The Figure 1 results indicated that pretreating MDCK cells with purified C. perfringens NanI sialidase can enhance ETX cytotoxicity by increasing toxin binding and thus, complex formation. Therefore, we next sought to assess whether sialidases also contribute to ETX-induced cytotoxicity when these glycoside hydrolyases are expressed at natural levels and in the natural presence of other C. perfringens exoproducts, including other toxins. To initiate this work, we first surveyed sialidase activity in 6 h and overnight supernatants from cultures of various C. perfringens isolates (Figure 2). This survey detected considerable strain-to-strain variations in supernatant sialidase activity and also identified type D strain CN3718 as a moderately high sialidase producer. Since CN3718 is also transformable and produces ETX (see below), it was chosen for construction of sialidase mutants.
Fig. 2. Sialidase activity in different type C. perfringens strains. 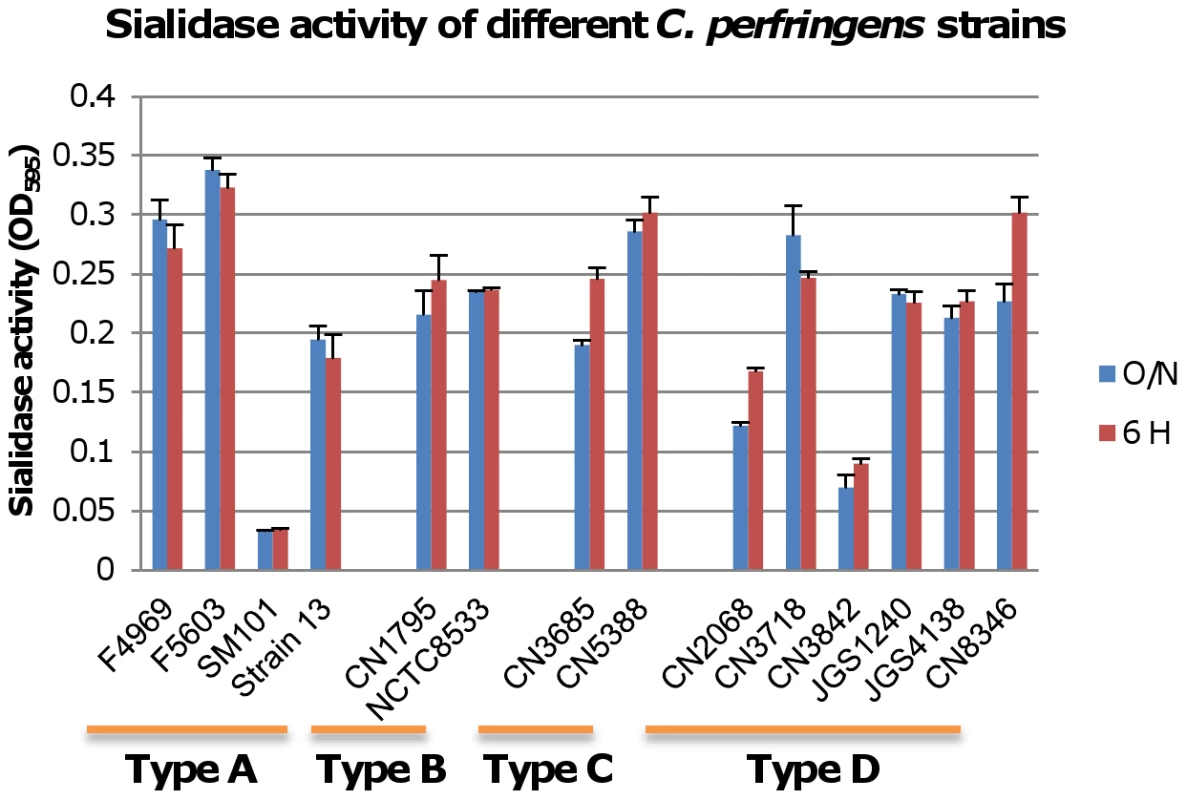
Sialidase activity was measured in supernatants from overnight and 6 h TH cultures of the specified C. perfringens strains, including type A strains F4969, F5603, SM101and Strain13; type B strains CN1795 and NCTC8533; type C strains CN3685 and CN5385 and type D strains CN2068, CN3718, CN3842, JGS1240, JGS4138 and CN8346. Results shown are the average of three repetitions; the error bars indicate the standard deviation (S.D.). The origin and typing of these strains has been described previously [14], [16], [33]-[36]. PCR analyses first determined that CN3718 carries all three identified C. perfringens sialidase genes (data not shown), including nanI and nanJ, which encode the secreted NanI and NanJ sialidases, and nanH, which encodes the NanH sialidase that lacks a signal peptide and thus localizes in the cytoplasm. Therefore, the current study used a Clostridium-modified Targetron insertional mutagenesis method [24] to construct isogenic single nanH, nanI, and nanJ null mutants, a nanI/nanJ double null mutant, and a nanH/nanI/nanJ triple null mutant in a CN3718 background.
The identity of the CN3718 nanJ, nanI and nanH single null mutants (named BMC201, BMC202 and BMC203, respectively) was first demonstrated by PCR using primers specific for internal nanJ ORF, nanI ORF or nanH ORF sequences. Using DNA from wild-type CN3718, these internal PCR primers specifically amplified the expected PCR products of 306 bp for nanJ, 467 bp for nanI, and 285 bp for nanH (data not shown). However, consistent with the insertion of an ∼900 bp intron into the target ORF, the same primers amplified PCR products of ∼1200 bp, ∼1400 bp, and ∼1200 bp for the nanJ, nanI and nanH null mutants, respectively (data not shown). Those PCR assays also confirmed the identity of a nanJ/I double null mutant (BMC204) and a nanJ/I/H triple null mutant (BMC205) constructed by Targetron technology (data not shown). The nanJ/I double null mutant PCR supported amplification of two larger bands, which matched the product sizes of a disrupted nanJ gene and nanI gene, along with a small band matching the ∼285 bp product size for the wild-type nanH gene (data not shown). Using BMC205 DNA, PCR amplified larger bands for all three sialidase ORFs (data not shown).
After curing the intron delivery plasmids from the BMC201, BMC202, BMC203, BMC204 or BMC205 sialidase null mutants, DNA from the wild-type strain and each null mutant was subjected to Southern blot analysis (Figure 3A) using an intron-specific probe [25]. No probe hybridization to wild-type DNA was detected, as expected. In contrast, the presence of a single intron insertion was visible on this Southern blot using DNA from each single sialidase null mutant. In addition, two intron insertions were detected using DNA from the BMC204 nanI/J double null mutant and three intron insertions were noted using DNA from the BMC205 nanI/J/H triple null mutant.
Fig. 3. Intron-based mutagenesis to create isogenic CN3718 sialidase null mutants and characterization of those mutants. 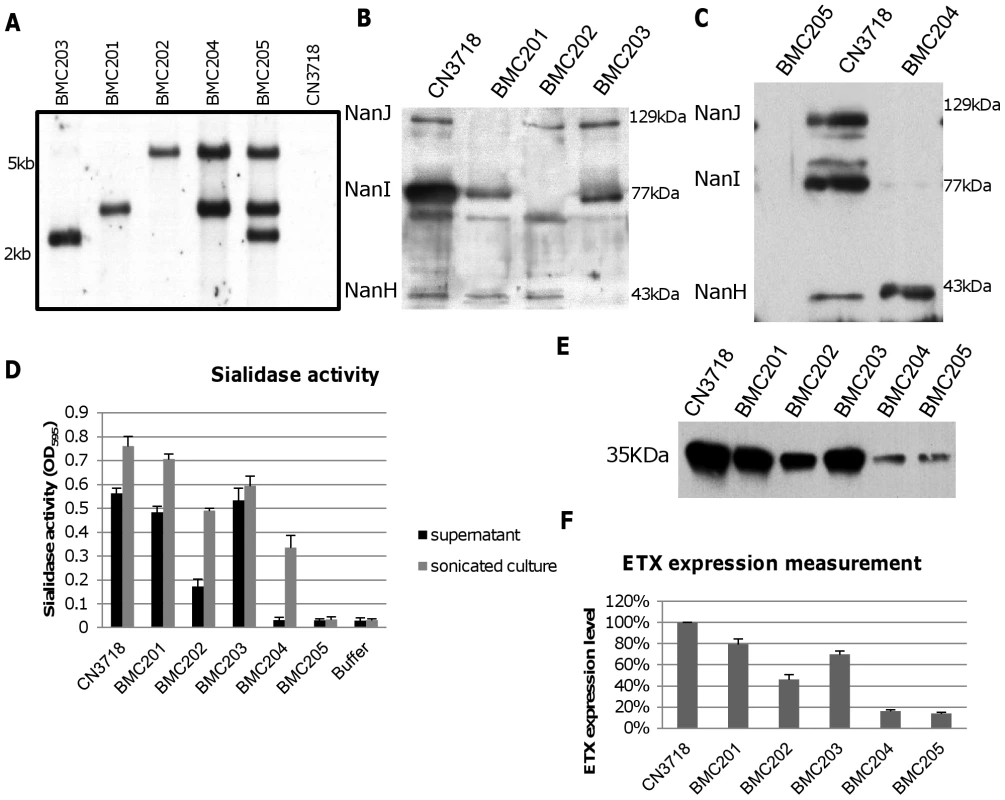
Panel A, Southern blot hybridization of an intron-specific probe with DNA from wild-type CN3718 or the sialidase null mutants (BMC201, BMC202, BMC203, BMC204 or BMC205). DNA from each strain was digested with BsrGI and electrophoresed on a 1% agarose gel prior to blotting and hybridization with an intron-specific probe. Size of DNA fragments, in kilobases (kb) is shown at left. Panel B, Western blot analyses for sialidase expression by wild-type CN3718 or the nanJ, nanI or nanH single null mutants named BMC201, BMC202, or BMC203, respectively. Size of proteins in kDa is shown at right. Panel C, Western blot analyses for sialidase expression by wild-type CN3718, the nanJ/nanI double null mutant (BMC204) or the nanJ/nanI/nanH triple null mutant (BMC205). Size of proteins in kDa is shown at right. Panel D, sialidase activity analyses for wild-type CN3718 or specified sialidase null mutant strains using 8 h TH culture supernatants or supernatants from 8 h TH sonicated cultures. Panel E, Western blot analyses for ETX expression by wild type CN3718 or sialidase single, double or triple null mutant strains (BMC201, BMC202, BMC203, BMC204 or BMC205). Size of prototoxin in kDa is shown at left. Panel F, Measurement of ETX expression by wild-type CN3718 or sialidase null mutant strains, based upon densitometric scanning of panel E gels. All the results show the average of three repetitions; the error bars indicate the standard deviation (S.D.). Phenotypic characterization of wild-type CN3718 and isogenic sialidase null mutants
To initiate a phenotypic characterization, vegetative growth of the isogenic mutants was compared in Todd Hewitt (TH) medium. After similar inoculation into TH medium, the vegetative growth rate of the isogenic mutants was nearly identical to the CN3718 parent (data not shown). Western blots then compared sialidase expression between CN3718 and the isogenic single, double and triple sialidase null mutants. These analyses demonstrated that sonicated 8 h TH cultures of the wild-type strain contain the 129 kDa NanJ, the 77 kDa NanI and the 43 kDa NanH (Figure 3B). In contrast, the BMC201 null mutant only produced NanI and NanH, the BMC202 null mutant only produced NanJ and NanH, and the BMC203 null mutant produced only NanI and NanJ (Figure 3B). Similar Western blotting of sonicated cultures (Figure 3C) showed that the BMC204 nanI/J double null mutant only produced the 43 kDa NanH, while the BMC205 nanJ/I/H triple null mutant did not produce any sialidase protein.
To evaluate the effects of mutating each sialidase gene on the total sialidase activity of CN3718, sialidase activity assays were carried out using either 8 h TH culture supernatants (to measure exosialidase activity) or sonicated 8 h TH culture supernatants (to measure total sialidase activity) from the wild-type parent and each null mutant strain. Collectively, the results (Figure 3D) indicated that the exosialidase activity measured in supernatants from the wild-type strain is mainly attributable to NanI. The Figure 3D results also confirmed that NanH predominantly accumulates in the cytoplasm since 8 h supernatants of the BMC204 nanI/J double mutant exhibited almost no sialidase activity, while the sonicated culture supernatant of this double mutant possessed significant sialidase activity. Lastly, this analysis demonstrated that CN3718 produces only the three recognized sialidases since the supernatant and sonicated culture of the BMC205 nanI/J/H triple null mutant lacked sialidase activity.
Since a major goal of the current study was to evaluate a possible relationship between sialidases and ETX action, ETX expression was measured for wild-type CN3718 and the isogenic sialidase null mutants. Surprisingly, each single sialidase mutant, but especially the BMC202 nanI null mutant, showed decreased ETX expression compared to the wild-type parent (Figure 3E and 3F). ETX expression levels by the BMC204 nanI/J double null mutant and the BMC205 nanI/J/H triple mutant decreased further compared against ETX production levels by wild-type CN3718 (Figure 3E and 3F).
Construction and genotypic characterization of isogenic etx null mutants of CN3718 and BMC205
The considerable ETX expression differences between wild-type strain CN3718 and the sialidase mutants shown in Figure 3E and 3F precluded using those strains for comparative ETX cytotoxicity experiments aimed at evaluating sialidase contributions to ETX action under natural sialidase expression levels and in the presence of natural levels of other secreted C. perfringens products. To overcome this complication, etx null mutations were engineered into the CN3718 wild-type strain and the BMC205 triple sialidase null mutant strain to create BMC206 and BMC207, respectively. The availability of these two etx mutants would allow later comparative cytotoxicity experiments using equivalent amounts of ETX added to supernatants of these strains. These etx mutants were constructed by a Targetron insertional mutagenesis approach. PCR confirmed the presence of an intron insertion into the etx gene (Figure 4A). Using DNA from wild-type CN3718, internal etx PCR primers specifically amplified a PCR product of 117 bp. However, consistent with a 900 bp intron insertion into the etx ORF, the same primers amplified a PCR product of ∼1000 bp using DNA from the putative etx mutants of both BMC206 and BMC207. PCR approaches also confirmed that BMC207 maintains intron insertions in its nanI, nanJ and nanH ORFs (Figure 4B).
Fig. 4. Intron-based mutagenesis to create etx null mutants of wild-type CN3718 and BMC205. 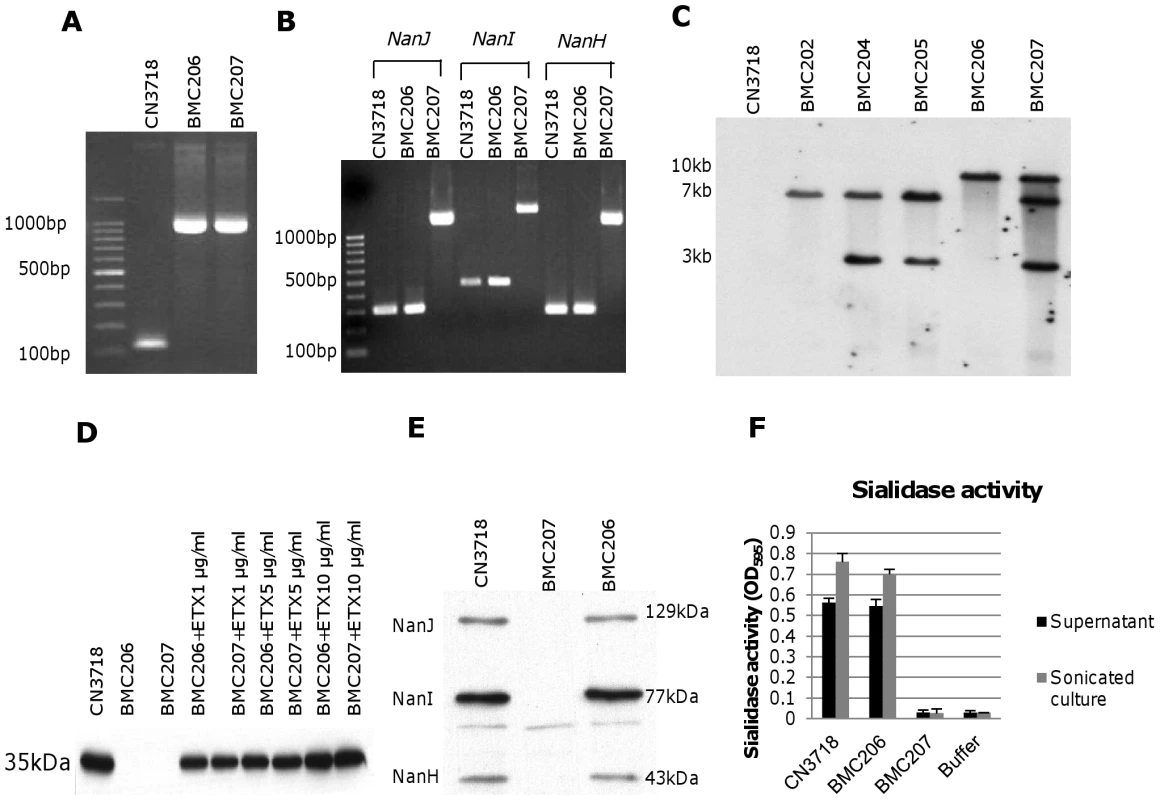
Panel A, PCR confirmation of construction of isogenic etx null mutants of wild-type CN3718 (creating BMC206) and BMC205 (creating BMC207). Using DNA isolated from wild-type CN3718, a PCR assay amplified a 117 bp product using internal etx primers but an ∼1.0 kb product, consistent with insertion of a 0.9 kb intron, using template DNA isolated from BMC206 or BMC207 etx null mutants. Lane 1, 100 bp molecular size markers. Panel B, PCR assays for nanI, nanJ or nanH confirming that BMC206 carries the wild-type nanI, nanJ or nanH genes, but BMC207 has a 0.9 kb intron insertions in its nanI, nanJ and nanH null mutant genes. Lane 1, 100 bp molecular size markers. Panel C, Southern blot hybridization of an intron-specific probe with DNA from wild-type CN3718, BMC202, BMC204, BM205, BMC206 or BMC207. DNA from each strain was digested with EcoRI and electrophoresed on a 1% agarose gel prior to blotting and hybridization with an intron-specific probe. Size of DNA fragments, in kilobases (kb), is shown at left. Panel D, ETX Western blot analyses of supernatants from wild-type CN3718 or the BMC206 and BMC207 etx null mutants, without or with supplementation with 1 µg/ml, 5 µg/ml or 10 µg/ml of ETX. Size of prototoxin in kDa is shown at left. Panel E, Western blot analyses for sialidase expression by wild-type CN3718, BMC206 or BMC207. Size of proteins in kDa is shown at right. Panel F, sialidase activity analyses for wild-type CN3718 or the BMC206 and BMC207 etx null mutant strains using 8 h TH culture supernatants or supernatants from sonicated 8 h TH cultures. After curing the intron delivery plasmids from these mutants, DNA was isolated from the two putative etx null mutants and subjected to Southern blotting using an intron-specific probe (Figure 4C). This analysis detected no probe hybridization with wild-type CN3718 DNA, as expected. As no suitable enzyme was identified that could separate the four intron copies present in BMC207, EcoRI was used to distinguish the presence of the etx intron from the other three sialidase introns, whose presence in the BMC205 mutant had already been demonstrated (Figure 3A). By this approach, the presence of the etx intron was clearly shown in BMC206 and BMC207 by Southern blotting (Figure 4C).
Phenotypic characterization of isogenic BMC206 and BMC207 null mutants
To initiate phenotypic comparisons, the growth rates of the BMC206 and BMC207 etx null mutants were first determined to be similar to wild-type CN3718 in TH medium (data not shown). Western blotting then confirmed the lack of ETX expression by these etx null mutants (Figure 4D). For later experiments it was important to demonstrate that culture supernatants could be supplemented with purified ETX to achieve equivalent ETX levels. This was confirmed by adding 1, 5 or 10 µg/ml of ETX to supernatants of the two etx null mutants (Figure 4D).
Sialidase Western blotting and sialidase activity analyses (Figure 4E and 4F) were next performed using cultures or sonicated cultures (to release NanH) of wild-type CN3718, BMC206 and BMC207 to evaluate whether introducing an etx gene mutation had affected sialidase expression. Western blot results demonstrated comparable levels of NanJ, NanI and NanH production by wild-type CN3718 and BMC206 when grown for 8 h in TH. However, as expected, production of these three sialidases was absent from similar cultures of BMC207 (Figure 4E). Measurement of sialidase activity in sonicated cultures (Figure 4F) further confirmed those Western blotting results. The sialidase activity detected in TH sonicated lysates or TH culture supernatants of CN3718 vs. BMC206 was very similar. However, no sialidase activity was measured in similarly prepared BMC207 sonicated lysates or culture supernatants.
Construction and characterization of nanJ, nanI and nanH complementing strains of BMC207
For preparing a nanI complementing strain of BMC207, we first constructed pJIR750Icomp, which consists of the CN3718 nanI ORF, 500 bp of upstream sequence and 300 bp of downstream sequence cloned into the C. perfringens/E. coli shuttle plasmid pJIR750. A nanJ complementing plasmid, named pJIR751Jcomp, was similarly prepared by PCR-amplifying a product corresponding to the nanJ ORF, 1000 bp of upstream sequence and 400 bp of downstream sequence, and then cloning that PCR product into the C. perfringens/E. coli shuttle plasmid pJIR751. Finally, by the same method, a nanH complementing plasmid named pJIR751Hcomp was prepared that contains 500 bp of upstream sequence, the nanH ORF and 300 bp of downstream sequence cloned into pJIR751. The three sialidase complementing plasmids were then individually transformed into BMC207 by electroporation. PCR confirmed the resultant transformants contained nanI, nanJ or nanH wild-type genes (Figure 5A). Using primers to internal sequences of each sialidase gene and BMC207 DNA, the nanI, nanJ or nanH PCR products amplified large bands indicating an intron insertion. In contrast, using DNA from the three complementing strains, the PCR products amplified were of smaller size, consistent with their containing a wild-type sialidase gene, which is preferentially amplified over the larger intron-disrupted sialidase gene also present in these complementing strains.
Fig. 5. Characterization of sialidase complementing strains. 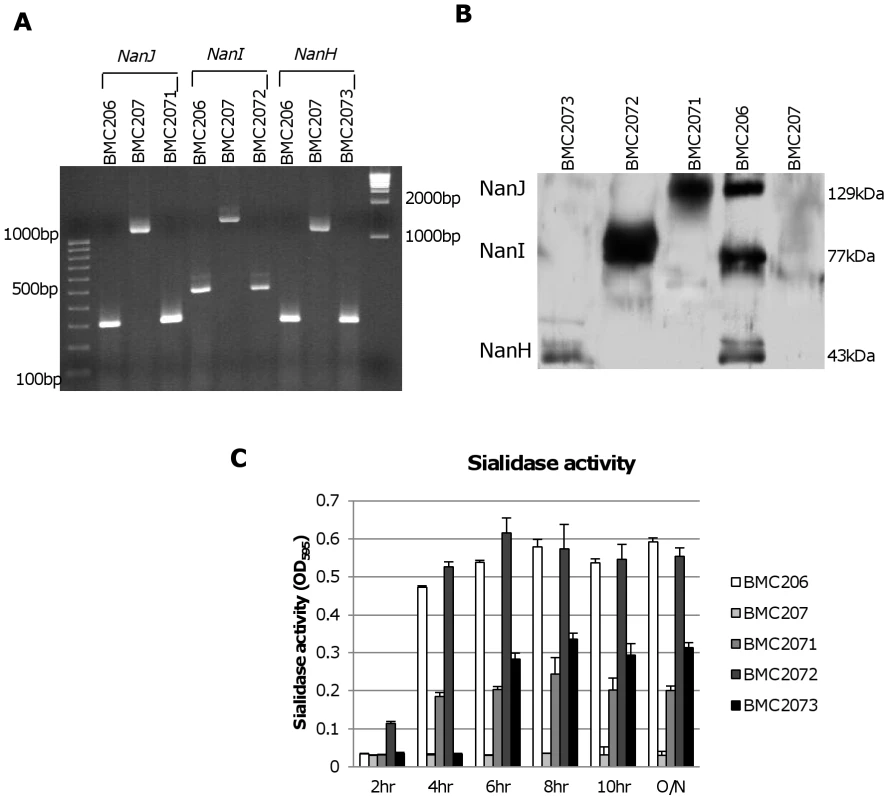
Panel A, PCR confirmation of the construction of isogenic sialidase null mutant and complementing strains. Using DNA isolated from BMC206, PCR assays using internal sialidase gene primers amplified the expected wild-type nanJ, nanI and nanH products of 306 bp, 467 bp and 285 bp, respectively. Using template DNA isolated from BMC207, which has a 900 bp intron insertion in each sialidase gene, the same PCR assays amplified larger products of 1206 bp, 1367 and 1185 for nanJ, nanI and nanH, respectively. With the same assay, DNA from each complementing strain yielded the same wild-type sized PCR products as amplified from BMC206. Lines 1 and 11, 100 bp or 1 kb molecular weight markers, respectively. Panel B, Western blot analyses for sialidase expression by complementing strains BMC2071 (for nanJ complementation), BMC2072 (for nanI complementation) and BMC2073 (for nanH complementation) using 8 h TH sonicated cultures. Size of protein in kDa is shown at right. Panel C, sialidase activity detected in supernatants of BMC206, BMC207, BMC2071, BMC2072 or BMC2073 after growth in TH medium for 2 h, 4 h, 6 h, 8 h, 10 h or overnight. Western blot analyses then assessed sialidase expression by the complementing strains (Figure 5B). As expected, BMC206 produced the ∼43 kDa NanH, the ∼77 kDa NanI and the ∼130 kDa NanJ. In contrast, the BMC207 null mutant expressed no sialidase proteins. These Western blots also showed that the nanH complementing strain BMC2073 only produced NanH, the nanI complementing strain BMC2072 only made NanI, and the nanJ complementing strain BMC2071 only expressed NanJ (Figure 5B).
The sialidase activity of the complementing strains was also assessed (Figure 5C). When sialidase activity was measured in 2 h, 4 h, 6 h, 8 h, 10 h or overnight TH cultures, the nanI complementing strain BMC2072 started expressing NanI as early as 2 h in TH culture. In contrast, BMC206 and the nanJ complementing strain began producing sialidase at 4 h. Wild-type CN3718 also expressed sialidase starting at 4 h (data not shown). The NanI complementing strain BMC2072 produced more sialidase activity than other complementing strains. Since the BMC2072 nanI single complementing strain already expressed more sialidase activity than either CN3718 or BMC206, and also due to technical limitations, no double or triple sialidase complementing strains were prepared.
Culture supernatants containing NanI enhance epsilon toxin binding to MDCK cells
The availability of sialidase mutants and complementing strains allowed us to evaluate whether specific sialidases can enhance ETX binding to MDCK cells at natural sialidase expression levels and in the background of other secreted C. perfringens products. The same 5 µg/ml amount of AF488-pETX was added to concentrated supernatants from BMC206, the BMC207 triple sialidase null mutant strain, or the three BMC207 sialidase complementing strains (i.e. BMC2071, BMC2072 and BMC2073). Note that, i) a natural 1× concentration of C. perfringens culture supernatant was finally present in these MDCK cell cultures, ii) a 5 µg/ml ETX concentration corresponds to a typical natural supernatant concentration of this toxin for ETX-producing C. perfringens strains [5], [6] and iii) these binding experiments were performed at 37°C to obtain the enzymatic effects of sialidases (when present), but previous studies [13] have shown that prototoxin binding to MDCK cells is equivalent at 4°C or 37°C.
When these mixtures were added to MDCK cells, the fluorescent readings showed that the BMC207 supernatant sample supported less AF488-pETX binding to MDCK cells compared against the BMC206 supernatant (Figure 6). Supernatant from the BMC2072 nanI complementing strain exhibited even greater levels of AF488-pETX binding than did BMC206 supernatant after a similar supplementation with the labeled prototoxin. In contrast, a smaller increase in AF488-pETX binding was noted using supernatants of the nanJ or nanH complementing strains supplemented with labeled prototoxin.
Fig. 6. Binding of the Alex 488-labeled prototoxin to MDCK cells. 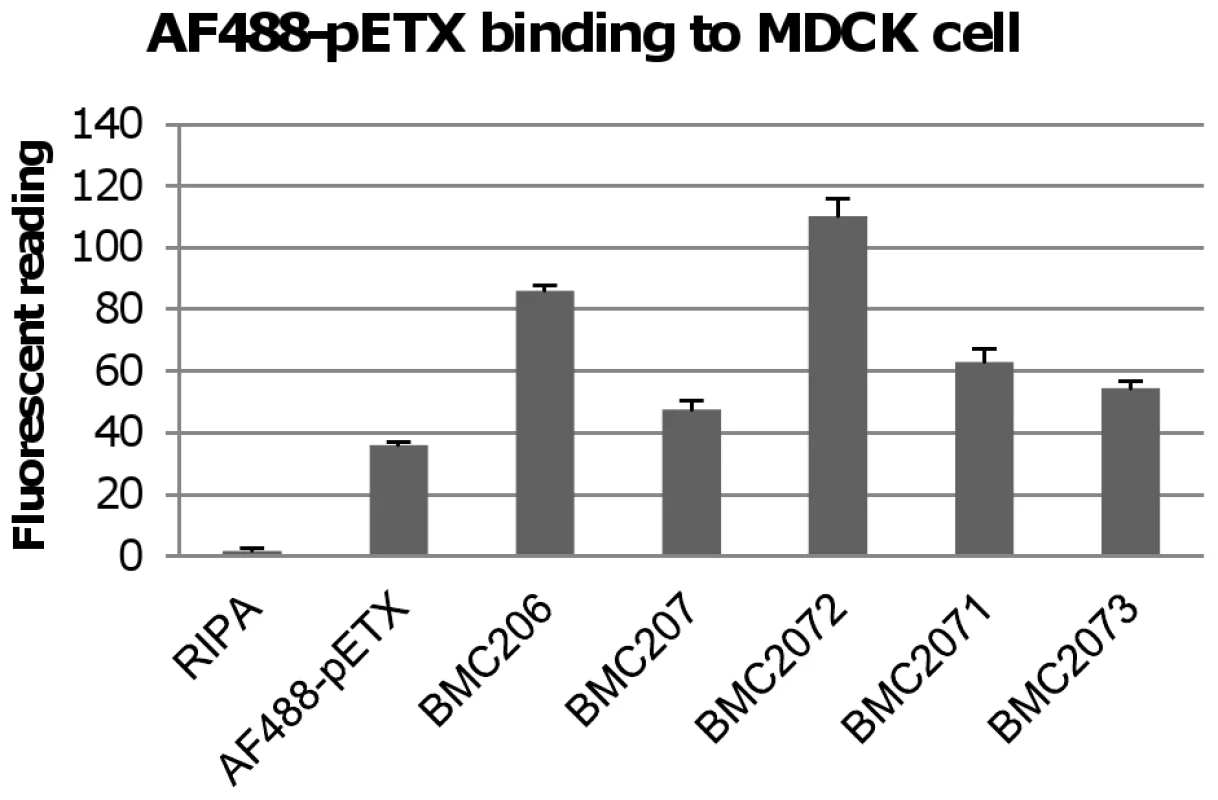
The left bar shows fluorescence for a negative control sample using MDCK cells lysed with RIPA buffer but not treated with labeled prototoxin. Another control shown included fluorescence for MDCK cells treated only with labeled prototoxin binding (no C. perfringens culture supernatant added). BMC206, BMC207, BMC2071, BMC2072, BMC2073 samples show fluorescence measured when labeled prototoxin (5 µg/ml) was added to the indicated TH supernatant before MDCK cell treatment. The results show the average of three repetitions; the error bars indicate standard errors of the means. NanI enhances epsilon toxin-mediated cytotoxicity in MDCK cells
Figure 1 results demonstrated that pretreating MDCK cells with purified C. perfringens NanI sialidase increases subsequent ETX cytotoxicity. Results shown in Figure 7A confirmed this conclusion and demonstrated that this effect is not due to direct sialidase-induced cytotoxicity.
Fig. 7. Sialidases increase cytotoxicity for MDCK cells. 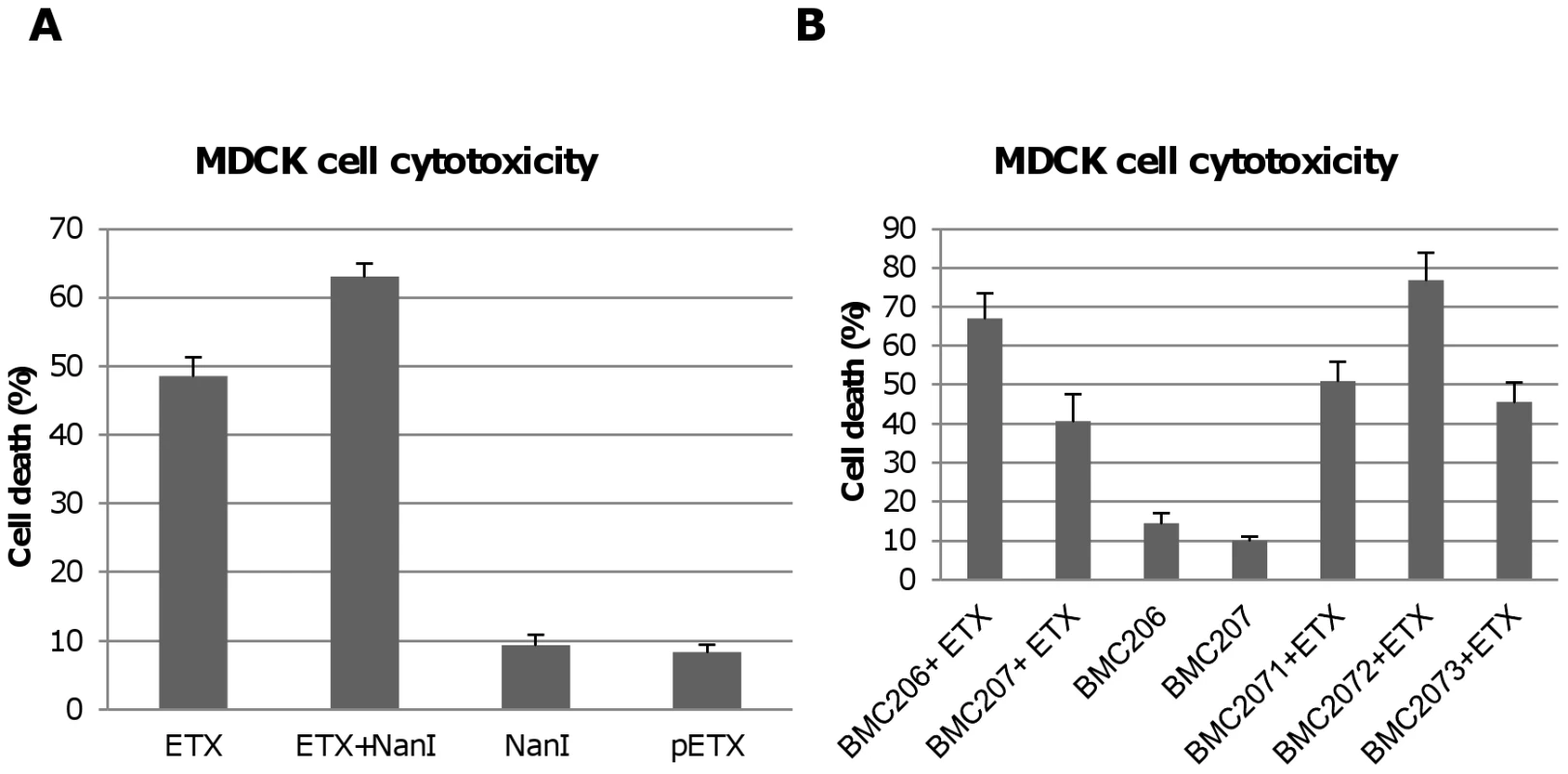
Panel A, percent of dead MDCK cells after treatment at 37°C for 1 h with 5 µg/ml ETX, 5 µg/ml pETX, 0.005 U/ml NanI sialidase or a mixture of 5 µg/ml ETX and 0.005 U/ml NanI sialidase, Panel B, percent of dead MDCK cells after the cells were treated for 1 h at 37°C with 5 µg/ml of ETX dissolved in BMC206, BMC207, BMC2071, BMC2072 or BMC2073 culture supernatants. BMC206 and BMC207 culture supernatants without ETX supplementation are shown for comparison. The results shown represent the average of three repetitions; the error bars indicate standard errors of the means. The Figure 6 binding results suggested that, compared against supernatants from BMC207, increased MDCK cell cytotoxicity should also be observed using supernatants from BMC206 or the nanI complementing strain BMC2072 when those supernatants were supplemented with ETX. Verifying this suggestion, the ETX-supplemented culture supernatants of BMC206 strain caused about 30% more MDCK cell cytotoxicity than did similarly ETX-supplemented BMC207 supernatants (Figure 7B). Furthermore, complementation with the wild-type nanI gene substantially increased the cytotoxic effects of the sialidase-deficient BMC207 mutant, while complementation of the BMC207 mutant with the wild-type nanJ or nanH genes increased cytotoxicity to a lesser extent.
NanI can be activated by trypsin
During natural disease, ETX is secreted into the animal intestines as an inactive prototoxin of ∼33 kDa, which can then be activated by intestinal proteases such as trypsin or chymotrypsin [11]. Interestingly, when CN3718 wild-type supernatants were trypsin-treated to activate prototoxin (data not shown), overall sialidase activity in these supernatants also significantly increased. This result was confirmed by treating purified C. perfringens NanI with trypsin. As shown in Figure 8A, the trypsin-treated NanI possessed significantly increased sialidase activity compared against the same amount of non-trypsin-treated NanI. When these samples were subjected to Western blotting, the results indicated that most of the trypsin-treated NanI ran as a slightly smaller protein than native NanI, supporting proteolytic processing (Figure 8B). In addition, chymotrypsin also activated NanI sialidase activity (data not shown).
Fig. 8. Effects of trypsin treatment on NanI sialidase. 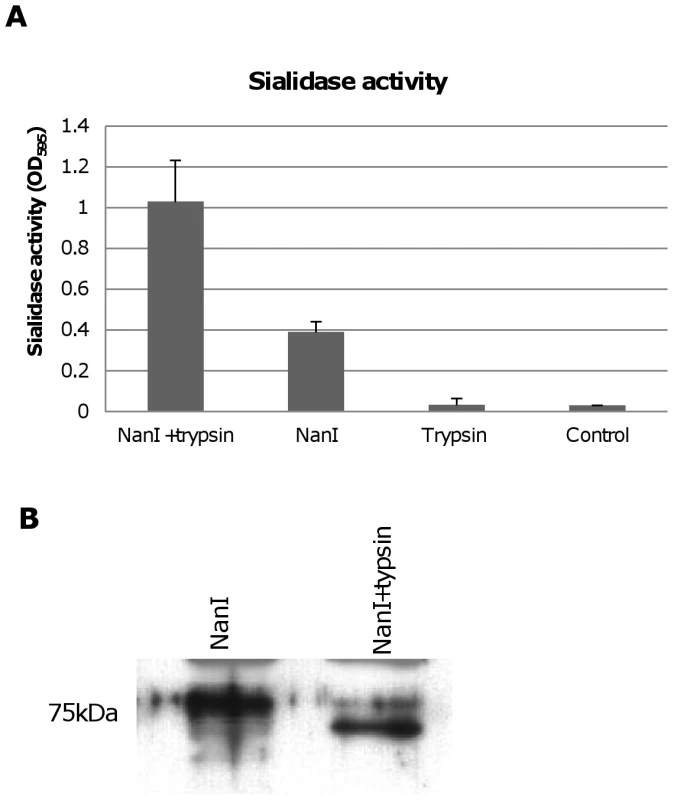
Panel A, Sialidase enzymatic activity was measured for 0.005 U/ml of purified NanI sialidase or 0.005 U/ml of purified NanI sialidase that had been treated for 1 h with 15 µg/ml of trypsin (note: following this treatment, trypsin activity was blocked using trypsin inhibitor). Also shown is sialidase activity measured for 15 µg/ml of trypsin (with trypsin inhibitor but no NanI sialidase added) and the buffer control. The results shown represent the average of three representations; the error bars indicate standard errors of the means. Panel B, Western blot comparative size analysis of NanI sialidase and trypsin-treated NanI sialidase. Size of protein in kDa is shown at left. Trypsin activation of NanI can enhance cytotoxicity
A further experiment then compared ETX cytotoxicity in the presence of purified NanI sialidase that had, or had not, been trypsin-activated prior to mixing with purified active ETX. After blocking trypsin activity from the sialidase sample with trypsin inhibitor, an enhancement of ETX cytotoxicity was observed using the trypsin-activated sialidase compared against the same amount of sialidase that had not been trypsin-activated (Figure 9A). Trypsin-activated sialidase alone caused no increase in cytotoxicity above background.
Fig. 9. Trypsin-activation of NanI sialidase further enhances ETX-induced MDCK cell cytotoxicity. 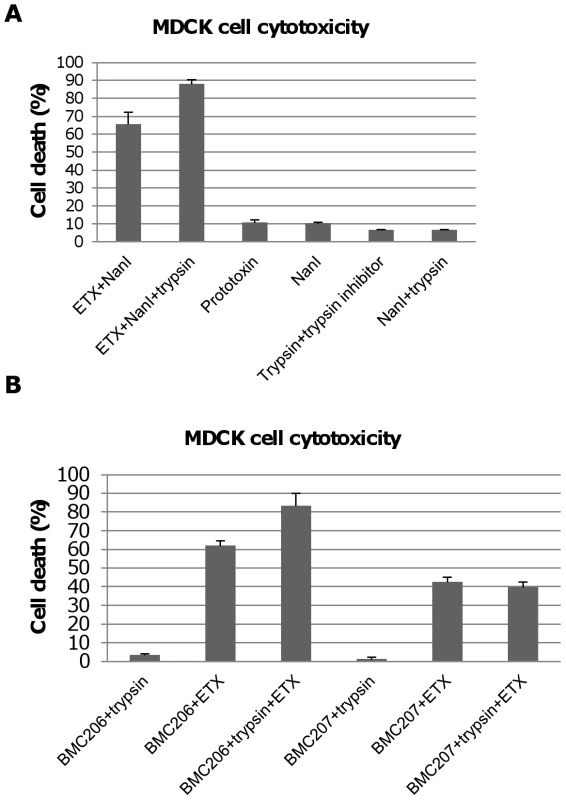
Panel A, percent of dead MDCK cells after treatment for 1 h at 37°C with 5 µg/ml of ETX in the presence of 0.005 U/ml sialidase or 5 µg/ml ETX in the presence of trypsin-treated 0.005 U NanI sialidase (note that trypsin activity in this sample was blocked with trypsin inhibitor before it was added to MDCK cells). Controls shown include: Prototoxin, NanI (no ETX), the trypsin and trypsin inhibitor mixture (no ETX or sialidase), and NanI with trypsin/trypsin inhibitor (no ETX). The results shown represent the average of three repetitions; the error bars indicate standard errors of the means. Panel B, percent of dead MDCK cells after treatment for with 5 µg/ml of ETX dissolved in BMC206 or BMC207 supernatants that had or had not been trypsin-treated for 1 h at 37°C (note: after this treatment, trypsin activity in this sample was blocked using trypsin inhibitor before the sample was added to MDCK cells). Effects of trypsin-treated BMC206 or BMC207 supernatants (no ETX addition) are shown as controls. The results shown represent the average of three repetitions; the error bars indicate standard errors of the means. Similar experiments using concentrated C. perfringens culture supernatants showed that trypsin activation of these supernatants enhanced MDCK cell cytotoxicity by about 25% for BMC206 (Figure 9B). Further supporting the involvement of sialidases in this trypsin-induced increase in the cytotoxic properties of BMC206, similar trypsin treatment of BMC207 supernatants did not increase ETX-induced cytotoxicity (Figure 9B).
The Figure 8A results indicated that NanI can be activated by trypsin. To assess whether the other two C. perfringens sialidase (NanJ and NanH) can also be trypsin-activated, the supernatants of nanI, nanJ and nanH complementing strains were similarly treated with trypsin and their sialidase activity was measured. The results obtained indicated that only the sialidase activity of NanI is enhanced by trypsin (Figure 10A). In contrast, NanJ and NanH sialidase activity slightly decreased after similar trypsin treatment (Figure 10A). Consistent with this result, trypsin increased the MDCK cell cytotoxicity of supernatants containing only NanI, but not supernatants containing only NanJ or NanH (Figure 10B).
Fig. 10. Sialidase activity and cytotoxicity for MDCK cells caused by culture supernatants from complementing strains. 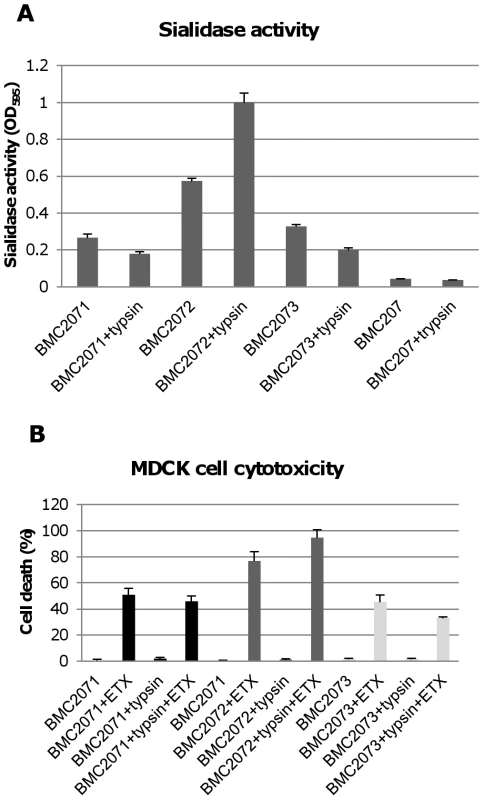
Panel A, sialidase activity detected in 8 h TH culture supernatants with or without trypsin treatment (note: following trypsin treatment, trypsin activity was blocked with trypsin inhibitor before the sample was assayed for sialidase activity). Effects of BMC207 and trypsin-treated BMC207 8 h TH culture supernatants are shown as controls. The results shown represent the average of three repetitions; the error bars indicate standard errors of the means. Panel B, percent of dead MDCK cells after treatment for 1 h at 37°C with 5 µg/ml of ETX added to 8 h TH culture supernatants of BMC2071, BMC2072 or BMC2073; % dead MDCK cells after treatment for 1 h with 5 µg/ml of ETX added to 8 h trypsin-treated TH culture supernatants of BMC2071, BMC2072 or BMC2073 (after this treatment, trypsin activity was blocked with trypsin inhibitor before addition of the sample to MDCK cells). Also shown are the cytotoxic effects after 1 h on MDCK cells of BMC2071, BMC2072 or BMC2073 8 h TH culture supernatants without ETX supplementation, as well as trypsin treated BMC2071, BMC2072 or BMC2073 8 h TH culture supernatants without ETX supplementation. The results shown represent the average of three repetitions; the error bars indicate standard errors of the means. Contact with certain mammalian cells upregulates CN3718 exosialidase activity
During natural enterotoxemias, type D strains remain present in the intestines, where they contact host enterocytes [8]. Therefore, an experiment examined whether contact with cultured mammalian cells for 6 hr (including intestinal Caco-2 and HT-29 cells as well as MDCK, Vero and NT6 fibroblasts) might affect the exosialidase activity of BMC206. This study revealed that contact between BMC206 and Caco-2, HT-29, or MDCK cells resulted in increased culture supernatant sialidase activity, although this effect was weaker using MDCK cells. In the absence of BMC206 infection, supernatant sialidase activity for Caco-2, HT-29 and MDCK cell cultures did not increase above background (Figure 11A), suggesting that the increased culture supernatant sialidase activity measured in the BMC206-infected cultures was not from secreted mammalian sialidase activity. The results also detected no culture supernatant sialidase activity upon infection of these mammalian cells with the triple sialidase null mutant BMC207. In contrast to the stimulation of sialidase activity observed using Caco-2, HT-29 or MDCK cells, exosialidase activity did not increase after BMC206 infection of Vero cell or NT6 fibroblast cultures (Figure 11A).
Fig. 11. Sialidase activity upon contact with mammalian cells. 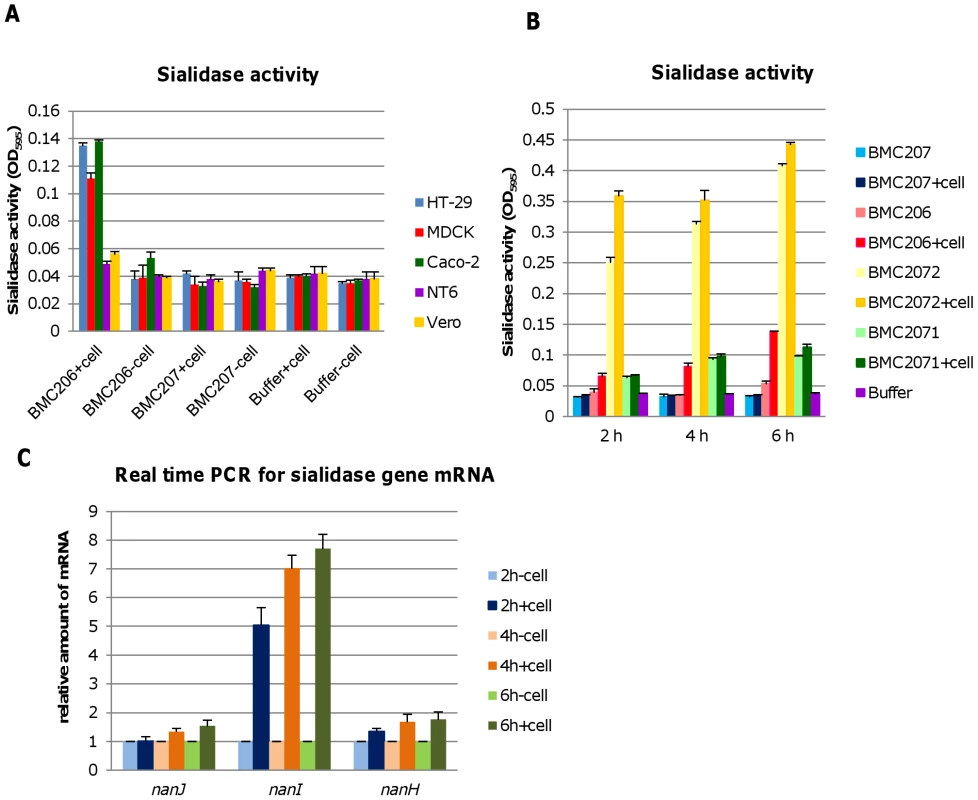
Panel A, Sialidase activity detected after washed BMC206 and BMC207 cells were suspended in HBSS and added to tissue culture wells that did or did not contain monolayers of HT-29, MDCK, Caco-2, NT6 or Vero cells. Buffer w/o cells as negative control. The results show average of three representations; the error bars indicate standard errors of the means. Panel B, Sialidase activity detected after washed BMC206, BMC207, BMC2071 or BMC2072 cells were suspended in HBSS and added to tissue culture wells that did or did not contain monolayers of Caco-2 cells for 2, 4 or 6 h. The results shown represent the average of three repetitions; the error bars indicate standard errors of the means. Panel C, Real time PCR for sialidase gene mRNA expression (nanJ, nanI and nanH), when the washed bacteria were added to tissue culture wells that did or did not contain Caco-2 cells, followed by incubation for 2, 4 or 6 h. The results show the average of three representations; the error bars indicate standard errors of the means. The nature of this host cell-induced increase in sialidase activity was further investigated using a Caco-2 cell infection model (Figure 11B). This study detected no culture supernatant sialidase activity upon infection of the triple null mutant BMC207 with Caco-2 cells up to 6 h, consistent with the increased sialidase activity detected upon BMC206 infection of Caco-2 cells involving upregulated sialidase expression by BMC206 upon Caco-2 cell contact. Results obtained using the BMC2072 NanI complementing strain suggested this upregulation of sialidase activity upon contact of BMC206 with Caco-2 cells primarily involves increased NanI production (Figure 11B). To definitively resolve whether contact with Caco-2 cells upregulates NanI expression by BMC206, quantitative RT-PCR studies were performed (Figure 11C). Those studies clearly demonstrated a substantial increase in nanI transcription in the presence of Caco-2 cells.
NanI increases C. perfringens attachment to some mammalian cells
When causing disease originating in the intestines, C. perfringens type D vegetative cells likely adhere to intestinal tissue to colonize and sustain toxin production [8]. Relatively little is known about C. perfringens adherence to host cells, particularly enterocytes during enteritis or enterotoxemia. However, since enterocytes are coated with a variety of sialic acid-containing glycoconjugates, sialic acid residues could conceivably modulate attachment of these bacteria to the intestines.
Therefore, experiments tested whether BMC206 or BMC207 can attach to cultured mammalian cells, including Caco-2 cells, HT-29 cells, MDCK cells, Vero cells and NT6 fibroblasts. As shown in Figure 12A and 12B, BMC206 attached well to Caco-2 and HT-29 cells, but substantially less well to the other surveyed host cells. BMC206 attachment to Caco-2 and HT-29 cells involved sialidase production since BMC207 failed to attach to those two mammalian cell lines (Figure 12A and 12B).
Fig. 12. Attachment of C. perfringens to mammalian cells. 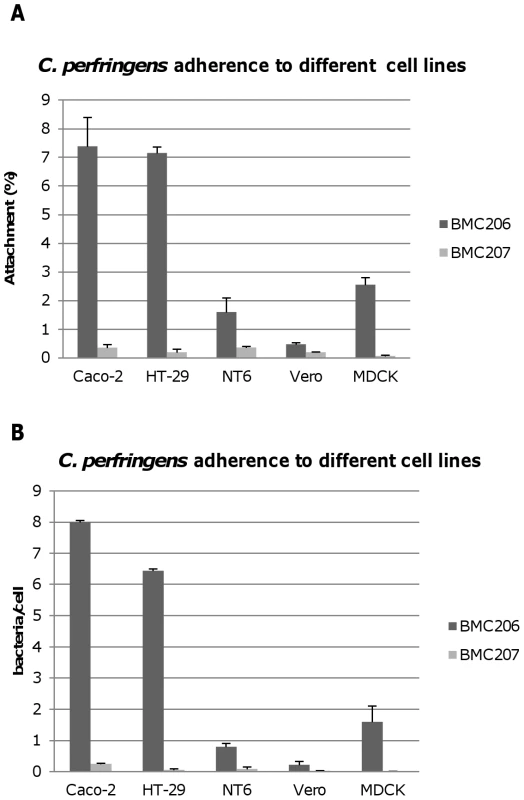
Caco-2, HT-29, NT6, Vero or MDCK cells monolayers were incubated for 2 h with the indicated bacteria at 37°C under anaerobic conditions. Monolayers were then washed three times with HBSS buffer, lysed in distilled water and total bacteria were plated onto BHI agar plates for counting. Panel A, Attachment levels expressed as the percentage of attached bacteria relative to the total number of input bacteria. The results shown represent the average of three repetitions; the error bars indicate standard errors of the means. Panel B, Attachment levels expressed as the bacteria attached per mammalian cell. The results shown represent the average of three repetitions; the error bars indicate standard errors of the means. Additional studies were performed with the human enterocyte-like Caco-2 cell line as an in vitro model to examine whether sialidases play a role in CN3718 adherence to intestinal cells (Figure 13A and 13B). As already established (Figure 12A), BMC206 attached well to Caco-2 cells. In contrast, there was little or no attachment of BMC207 to these host cells under the same experimental conditions. However, the BMC2072 nanI complementing strain exhibited Caco-2 cell adhesion levels equal to, or exceeding those of, BMC206 (Figure 13A and 13B). In contrast, the Caco-2 adhesion ability of the nanJ and nanH complementing strains was not substantially increased over the attachment of BMC207 (Figure 13A and 13B).
Fig. 13. Attachment of C. perfringens to Caco-2 cells. 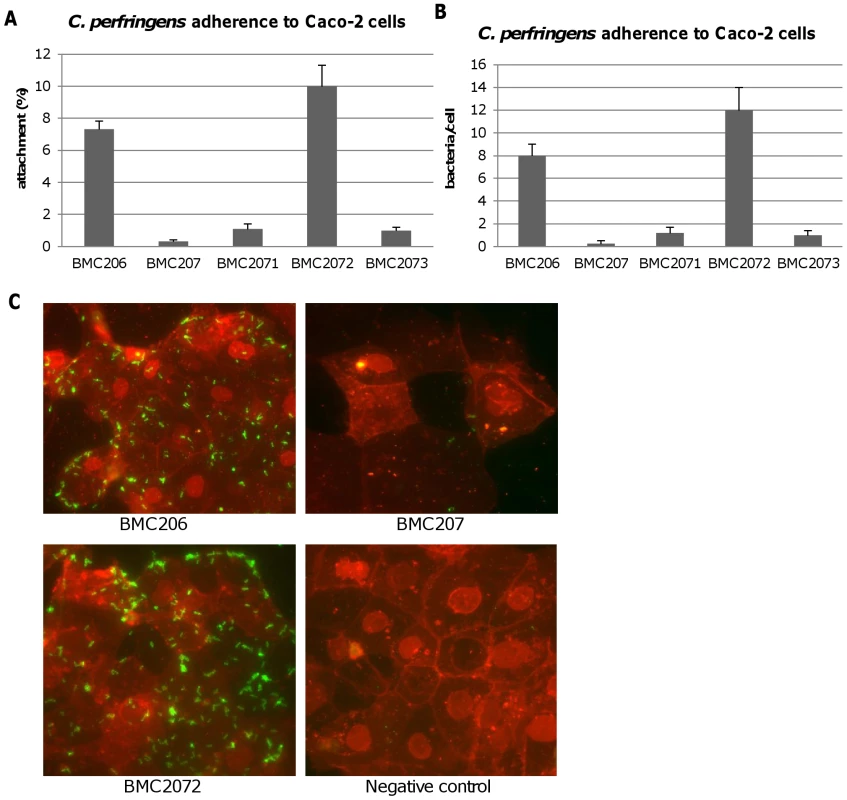
Caco-2 cell monolayers were incubated for 2 h with bacteria at 37°C under anaerobic conditions. Monolayers were then washed three times with HBSS buffer, lysed in distilled water and total bacteria were plated onto BHI agar plates for counting. Panel A, Attachment levels expressed as the percentage of attached bacteria relative to the total number of input bacteria. The results shown represent the average of three repetitions; the error bars indicate standard errors of the means. Panel B, Attachment levels expressed as the bacteria attached per mammalian cell. The results shown represent the average of three repetitions; the error bars indicate standard errors of the means. Panel C, C. perfringens BMC206, BMC207 and BMC2072 attached to Caco-2 cells as detected by fluorescence microscopy (600×) as described in the Materials and Methods. Green: bacteria; Red: Caco-2 cells. A role for NanI sialidase in modifying the Caco-2 cell surface to increase the adherence of CN3718 vegetative cells to these host cells was further supported by results of an experiment where Caco-2 cells were pretreated with purified NanI. This pretreatment substantially increased the Caco-2 cell adherence of the BMC207 mutant (data not shown).
Phase-contrast and immunofluorescence microscopy analyses (Figure 13C) confirmed the strong adhesion of BMC206 or BMC2072 vegetative cells to Caco-2 cells. This microscopy detected many adherent bacteria attached to Caco-2 cells, often along their edges. In contrast, few (if any) BMC207 cells were adherent to Caco-2 cells.
Discussion
A recent study [20] showed that a nanI mutation nearly completely abolished the sialidase activity of C. perfringens type A strain 13, which produces NanI and NanJ. However, unlike strain 13, many C. perfringens strains can also produce NanH [14]. Using a combination of nanI, nanJ or nanH single null mutants, a nanI/nanJ double null mutant, and a sialidases triple null mutant, the current study evaluated the relative contribution of each sialidase to the exosialidase activity and total sialidase activity of C. perfringens type D strain CN3718. These analyses revealed that, using the sialidase assay conditions employed in this study, i) CN3718 produces all three recognized C. perfringens sialidases but no additional unknown sialidases, ii) NanI is responsible for most exosialidase activity of this strain and iii) NanH is also a major contributor to the total sialidase activity possessed by CN3718. Furthermore, by demonstrating that CN3718 produces all three sialidases, the current results argue against proposals [17] that only myonecrosis strains of C. perfringens produce all three sialidases. Bioinformatics analyses of database sequences support carriage of three sialidase genes as the norm for C. perfringens strains, regardless of origin. In addition to our current CN3718 findings, analysis of the 8 completed or partially-completed C. perfringens genomes revealed that all three sialidase genes are present in 6 of those 8 isolates, which include several isolates from humans or livestock suffering from C. perfringens disease originating in the intestines. The two exceptions are strain 13 and SM101 that, respectively, lack nanH or both nanI and nanJ [14], [16].
Previous studies with other bacterial pathogens showed that sialidases can contribute to virulence in several ways, e.g., by exposing sites to facilitate more bacterial adhesion or toxin binding to host cells [19], [26]. Our results indicated that NanI sialidase increases the ETX sensitivity of MDCK cells. The mechanism of this enhancement was shown, for the first time, to involve an increase in toxin binding, suggesting NanI exposes additional ETX receptors on the host cell surface. Alternatively, NanI could modify host cell surface nonreceptors so they acquire ETX binding ability, similar to the situation in Vibrio cholerae, where a sialidase modifies glycolipids to increase cholera toxin binding [18]. We also demonstrated that increased ETX binding to NanI-exposed MDCK cells leads to more ETX complex formation, which translates to greater pore formation and host cell death [23]. Overall, these conclusions are consistent with, and explain for the first time, observations from a previous study reporting that C. perfringens sialidases can increase ETX cytotoxicity towards MDCK cells [21]. They are also consistent with previous results showing that soluble sialic acid cannot inhibit ETX binding to, or action on, MDCK cells [21]. However, the current results apparently contrast with another study [22] reporting that sialidases lower ETX binding to brain synaptosomal membrane vesicles. It is not immediately clear whether the varying conclusions amongst these studies are attributable to using MDCK cells vs. synaptosomal membranes or involve some other explanation.
Sialidases assist the ability of some pathogens to adhere to host cells or tissues. For example, sialidase contributions to adherence and colonization are well-documented for S. pneumoniae [19]. However, prior to the current work, only one study had addressed C. perfringens vegetative cell adherence to the mammalian intestinal epithelium, despite the likely importance of this process for pathogenesis; that earlier study suggested a putative C. perfringens collagen adhesion protein (CNA) might contribute to porcine enteritis by promoting adhesion of this bacterium to damaged intestinal tissue [27]. Our study now reports the first evidence that adherence of a C. perfringens strain to certain host cells, including enterocyte-like Caco-2 cells, is facilitated by sialidases, especially NanI. Inactivation of NanI sialidase production decreased adhesion of CN3718 strain by at least 20-fold and this effect was reversible by complementation. Furthermore, pretreating Caco-2 cells with purified NanI allowed adherence of the BMC207 mutant unable to produce any sialidase, indicating that NanI modifies the Caco-2 cell surface to render it favorable for CN3718 adherence. NanH and NanJ appeared to play a lesser role in facilitating C. perfringens adherence to Caco-2 cells based upon complementation results using the triple sialidase mutant. The varying ability of the three sialidases to enhance ETX binding or C. perfringens adherence likely reflects the reported enzymatic difference in sialidase kinetic properties and substrate specificities [17], although further comparative study of the properties of C. perfringens sialidases is warranted.
These in vitro findings open the possibility that sialidases, particularly NanI, contribute to virulence by facilitating C. perfringens adherence in the intestines, a hypothesis that should be tested in animals. In addition, future studies should identify the adhesins mediating CN3718 binding to certain host cells. Beyond the CNA study [27] mentioned in the preceding paragraph, the only other available information regarding molecular mechanisms of C. perfringens adhesion to host cells is a report implicating type IV pilin in type A strain 13 attachment to myoblasts [28]. Future studies will examine whether the type IV pilus or CNA protein contribute to CN3718 adherence to host cells.
A notable finding of the current work was that CN3718 adhesion is specific for certain mammalian cells. Notably, this strain adheres well to Caco-2 and HT-29 cells, both of which are intestinal cell lines. Much lower attachment of this strain was detected using cell lines of nonintestinal origin, including MDCK cells (canine kidney cells), Vero cells (African green monkey kidney cells) or NT6 rat fibroblasts cells. Furthermore, contact of CN3718 with the intestinal cells lines, and MDCK cells to a lesser degree, also stimulated nanI transcription. Collectively, these results suggest that CN3718, a type D disease strain, is well-adapted for sensing the presence of enterocytes and then responding by producing more sialidase to facilitate its specific attachment to these cells. However, a survey including many additional cell lines and C. perfringens strains are needed to test this hypothesis.
The current study also suggests that trypsin could be a greater contributor to type D disease than previously appreciated [29]. It is well established that, following secretion, the inactive ETX prototoxin must be activated by protease-induced removal of residues from the N - and C-terminus [29]. This prototoxin activation normally occurs in the gut of infected animals, where it likely involves intestinal proteases, including trypsin. Interestingly, the current study found that trypsin proteolytically activates NanI as well as ETX, thereby increasing sialidase activity. Since we also showed that trypsin activation of NanI enhances ETX binding and cytotoxicity towards MDCK cells, trypsin-activated NanI could similarly increase ETX action in vivo. Furthermore, since NanI was also determined to be important for CN3718 adherence to Caco-2 cells, trypsin activation of NanI could also contribute to type D disease by promoting C. perfringens colonization in the intestines. Chymotrypsin also activated NanI sialidase activity (data not shown), so that intestinal protease could further enhance disease.
Another possible virulence role for sialidases is suggested by our observation that various CN3718 sialidase mutants, particularly nanI mutants, exhibited significantly decreased ETX production levels despite wild-type growth properties (data not shown). This result might indicate that sialic acid signals ETX production, but initial attempts to prove this hypothesis have yielded inconclusive results (data not shown). Interestingly, previous studies showed that inactivating the nanI gene in strain 13 also affects toxin production levels [20]. However, for that strain, the nanI mutant showed increased production levels of alpha-toxin and perfringolysin O. Whether the increased production of alpha toxin and perfringolysin O by strain 13 involves a sialic acid signal should also be evaluated in the future.
While our findings suggest NanI plays, at minimum, two virulence roles for CN3718, i.e., enhancing both ETX binding and attachment of this strain to enterocytes, it remains unclear why this strain also produces NanJ and NanH sialidases. As mentioned, NanH and NanI have different temperature optimums, kinetic properties and substrate specificities [17]; the enzymatic characteristics of NanJ, which was only discovered during genome sequencing studies [14] remains uncharacterized. Possibly NanH and NanJ are important for growth or survival in specific environments such as soil or sewage or in other infections. Furthermore, NanJ possesses additional domains of unknown function that are missing from NanI [17]; those additional NanJ domains might contribute to growth or virulence under conditions that remain to be identified.
In summary, our in vitro results suggest that, while NanI sialidase does not appear to be involved in the pathogenesis of type A isolates during myonecrosis [20], it could contribute to type D enterotoxaemia and enteritis in at least two ways. First, NanI (particularly after trypsin activation in the intestines) could increase ETX binding and complex formation, thereby potentiating ETX cytotoxicity. The potential relevance of this effect for damage to non-intestinal target organs, such as brain and kidneys, is unclear since it has not yet been evaluated whether NanI sialidase can be absorbed into the circulation during type D enterotoxemias. Similarly, ETX causes limited damage in the intestines of sheep [8], so sialidase potentiation of ETX may have less importance during ovine type D disease. However, sialidases could possibly increase ETX binding to the intestines of sheep or goats and thus facilitate absorption of this toxin into the circulation. Moreover, ETX does substantially damage the caprine gastrointestinal tract [8], [30], so, by enhancing ETX intestinal toxicity, trypsin-activated NanI could directly contribute to caprine type D enteritis. A second possible virulence contribution of NanI may be to increase C. perfringens vegetative cell adherence to the intestinal epithelium, thereby contributing to type D infections by facilitating colonization of ETX-producing bacteria in the intestines. Lastly, our results suggest that contact with intestinal cells may stimulate C. perfringens to produce more NanI, thereby further potentiating (particularly after trypsin and chymotrypsin activation) ETX effects and bacterial adherence during disease. The relevance of these in vitro findings for possible sialidase virulence contributions should now be tested experimentally in animal models.
Materials and Methods
Bacterial strains and medium
CN3718, an ETX-positive C. perfringens type D animal disease strain (Table 1), originated from the Burroughs-Wellcome collection and was obtained via Dr. R. G. Wilkinson [5]. NCTC8346, another type D animal disease isolate, was used for ETX toxin purification [5]. E.coli Top10 cells (Invitrogen) were used as the cloning host. Mutant, complementing strains, and plasmids used in this study are listed in Table 1.
Tab. 1. Strains, mutants and plasmids used in this study. 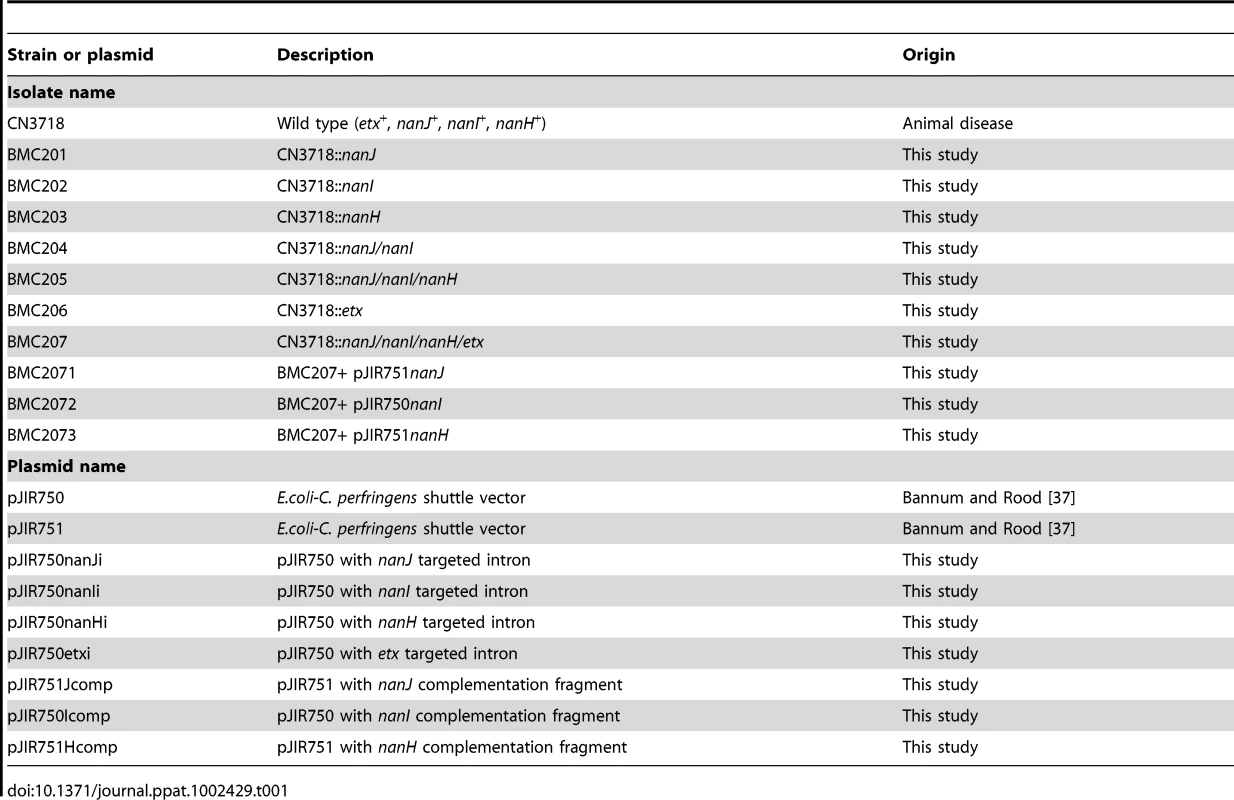
Media used in this study for culturing C. perfringens included FTG medium (fluid thioglycolate medium; Difco Laboratories); TH medium (Bacto Todd Hewitt Broth [Becton-Dickinson], with 0.1% sodium thioglycolate [Sigma Aldrich]); TGY medium (3% tryptic soy broth [Becton-Dickinson], 2% glucose [Fisher scientific], 1% yeast extract [Becton-Dickinson], and 0.1% sodium thioglycolate [Sigma Aldrich]) and BHI agar plates (brain heart infusion, Becton-Dickinson). For culturing E. coli, Luria-Bertani (LB) broth (1% tryptone [Becton-Dickinson], 0.5% yeast extract [Becton-Dickinson], 1% NaCl [Fisher scientific] and LB agar (1.5% agar [Becton-Dickinson]) were used. All antibiotics used in this study were purchased from the Sigma-Aldrich Chemical Company or Fisher Scientific Company.
Proteins and antibodies
Purified C. perfringens neuraminidase (NanI) was purchased from Roche Applied Science. ETX prototoxin was purified to homogeneity using a previously described method [5]. Following the method described by the manufacturer, the purified ETX prototoxin was fluorescently-labeled using an Alexa Fluor 488 protein labeling kit (Invitrogen), creating AF488-pETX. For ETX detection by Western blots, an ETX-specific monoclonal antibody (5B7; kindly provided by Paul Hauer, Center for Veterinary Biologics, Ames, Iowa) was used as primary antibody, followed by rabbit anti-mouse immunoglobulin G (IgG)-peroxidase conjugate (Sigma) as a secondary antibody. A polyclonal rabbit antibody against C. perfringens neuraminidases was purchased from Thermo Scientific.
Construction of nanJ-, nanI - and nanH-null mutant of C. perfringens CN3718
The nanJ, nanI and nanH genes of CN3718 were each inactivated by insertion of a group II intron via the Clostridium-modified TargeTron gene knock-out system [24]. Using intron insertion sites identified by the Sigma TargeTron algorithm, an ∼900 bp intron was targeted into the nanJ, nanI and nanH ORFs in a sense orientation. The intron insertion was targeted between nucleotides 657 and 658 of the nanJ ORF. The primers used for PCR targeting the nanJ intron were 657/658-IBS, 657/658-EBS1d and 657/658-EBS2 (Table 2). The intron insertion was targeted between nucleotides 730 and 731 of the nanI ORF using primers 730/731-IBS, 730/731-EBS1d and 730/731-EBS2 (Table 2). The intron insertion was targeted between nucleotides 707 and 708 of the nanH ORF. The primers used for PCR targeting the intron into nanH were 707/708-IBS, 707/708-EBS1d and 707/708-EBS2 (Table 2).
Tab. 2. Primers used in this study. 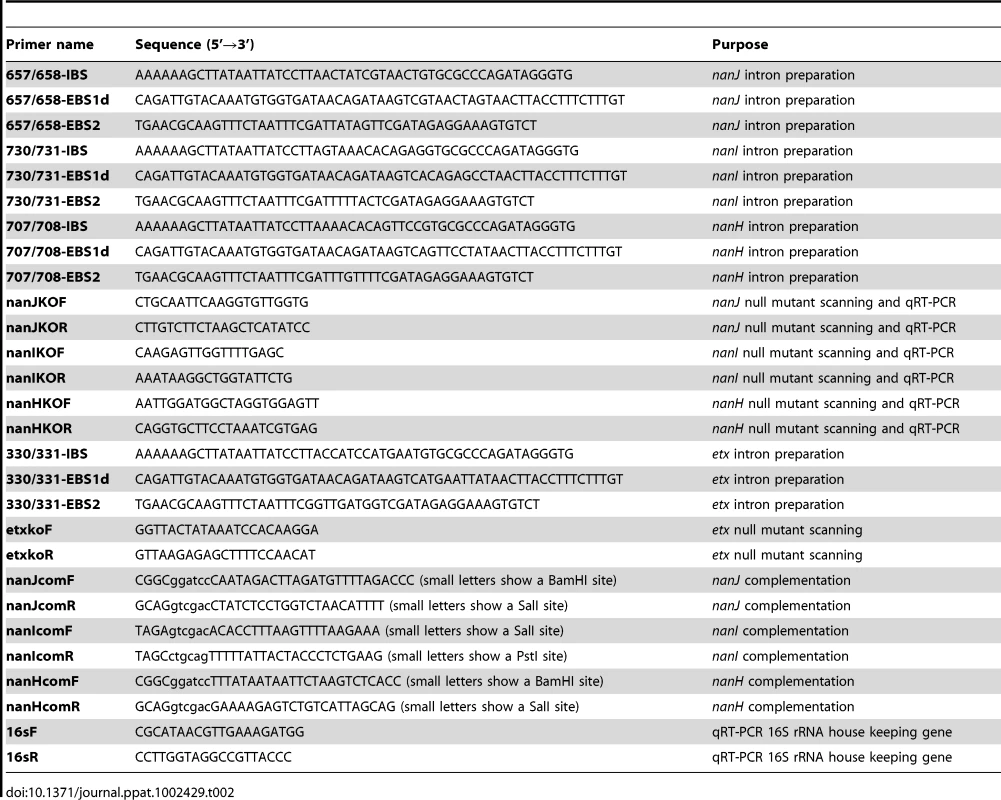
The 350 bp intron PCR products were inserted into pJIR750ai between the HindIII and BsrGI enzyme sites in order to construct nanJ, nanI and nanH-specific TargeTron mutagenesis plasmids. The resultant plasmids, named pJIR750nanJi, pJIR750nanIi and pJIR750nanHi respectively (Table 1), were individually electroporated into wild-type CN3718. This produced nanJ, nanI and nanH single null mutants named, respectively, BMC201, BMC202 and BMC203 (Table 1).
The CN3718 transformation efficiency was ∼240 transformants/µg plasmid DNA. Transformants were selected on BHI agar plates containing 15 µg/ml of chloramphenicol. Transformant colonies were then identified by colony PCR using primers nanJKOF and nanJKOR (Table 2) for screening nanJ-null mutants (BMC201), primers nanIKOF and nanIKOR (Table 2) for screening nanI-null mutants (BMC202), and primers nanHKOF and nanHKOR (Table 2) for screening nanH-null mutants (BMC203). Each reaction mixture was subjected to the following PCR amplification conditions: cycle 1, 95°C for 5 min; cycles 2 through 35, 95°C for 30 s, 55°C for 40 s, and 68°C for 90 s; and a final extension for 5 min at 68°C. An aliquot (20 µl) of each PCR sample was electrophoresed on a 1.5% agarose gel and then visualized by staining with ethidium bromide. The mutants were cured of the intron-carrying donor plasmid as described [25].
Construction of the CN3817 nanI/nanJ double null mutant BMC204
To prepare a nanI/nanJ double null mutant, pJIR750nanJi was electroporated into the BMC202 nanI null mutant strain, which had been cured of the pJIR750nanIi plasmid. Transformants were grown on BHI agar plates containing 15 µg/ml chloramphenicol and putative nanJ/nanI double null mutants were then identified by demonstrating the presence of introns in both the nanI and nanJ ORFs by PCR. The confirmed nanI/nanJ double null mutant (named BMC204, Table 1) was cured the intron-carrying donor plasmid pJIR750nanJi. The identity of this BMC204 nanI/nanJ double null mutant was then confirmed by Southern blotting, which demonstrated the presence of two introns in the mutant.
Construction of the BMC205 nanI/nanJ/nanH triple null mutant
To prepare a nanI/nanJ/nanH triple null mutant, pJIR750nanHi was electroporated into the BMC205 double nanI/nanJ null mutant strain and transformants were then grown on BHI agar plates containing 15 µg/ml of chloramphenicol. Putative nanH null mutants were screened by PCR, as described above. The presence of introns in the nanI, nanJ and nanH genes of this mutant (named BMC205, Table 1) was confirmed by PCR and Southern blotting, following curing of the intron-carrying donor plasmid pJIR750nanHi.
Inactivation of the etx gene in CN3817 and BMC205
An intron insertion, with a sense orientation, was targeted between nucleotides 330 and 331 of the etx ORF. The primers used for targeting this intron were 330/331-IBS, 330/331-EBS1d and 330/331-EBS2 (Table 2). The 350-bp PCR products were inserted into pJIR750ai between the HindIII and BsrGI enzyme sites in order to construct an etx-specific TargeTron plasmid. The resultant plasmid, named pJIR750etxi (Table 1), was electroporated into either wild-type CN3718 or BMC205 to inactivate their etx genes, which produced (respectively) etx null mutant strains named BMC206 and BMC207 (Table 1). The primers used for etx null mutant scanning were etxkoF and etxkoR (Table 2).
Southern hybridization analyses
DNA was isolated from wild-type CN3718, the five sialidase null mutants (including the three single null mutants BMC201, BMC202 and BMC203, the double nanJ/nanI null mutant strain BMC204, and the triple sialidase null mutant strain BMC205) and the two etx null mutants of CN3718 and BMC205 (including the BMC206 null mutant that produces all three sialidases and the BMC207 null mutant unable to produce any sialidase) using the MasterPureTM Gram-Positive DNA Purification Kit (Epicenter). Each DNA was then digested with BsrGI or EcoRI overnight at 37°C and run on a 1% agarose gel. After the alkali transfer to a nylon membrane (Roche), the blot was hybridized with a digoxigenin-labeled, intron-specific probe as described previously [25]. This intron-specific probe was prepared using the primers KO-IBS and KO-EBS1d [25] and a PCR DIG Labeling Kit (Roche Applied Science) according to the manufacturer’s instructions.
Complementation of the nanJ, nanI and nanH null mutants
DNA was isolated from CN3718 using the Master PureTM Gram-positive DNA purification kit. The primers nanJcomF and nanJcomR (Table 2) were used for nanJ complementing strain construction. The primers nanIcomF and nanIcomR (Table 2) were used for nanI complementing strain construction. The primers nanHcomF and nanHcomR (Table 2) were used for construction of a nanH complementing strain. The PCR reactions were set up as: 1 µl of each pair of primers (at a 0.5 µM final concentration), 1 µl of purified DNA template and 25 µl 2×Taq Long Range Mixture (NEB) were mixed together and ddH2O was added to reach a total volume of 50 µl. The reaction mixtures were then placed in a thermal cycler (Techne) and subjected to the following amplification conditions: 1 cycle of 95°C for 2 min, 35 cycles of 95°C for 30s, 55°C for 40s, and 65°C for 5 min, and a single extension of 65°C for 5 min. The resultant 4.9kb nanJ PCR product, 2.8 kb nanI PCR product, or 2.3 kb nanH PCR product were each separately cloned into the Invitrogen pCR2.1 TOPO vector according to the manufacturer’s instruction and inserts were then sequenced at the University of Pittsburgh Core Sequencing Facility. Using BamHI and SalI, the nanJ insert was removed from the TOPO vector and ligated into the pJIR751 C. perfringens/E. coli shuttle plasmid, forming a plasmid named pJIR751nanJcomp (Table 1). Using the same method, nanI and nanH inserts were separately cloned into pJIR750 and pJIR751 C. perfringens/E. coli shuttle plasmids, forming plasmids named pJIR750nanIcomp and pJIR751nanHcomp, respectively (Table 1). These plasmids were individually introduced, by standard electroporation techniques, into the BMC207 null mutant strain to create nanJ, nanI and nanH complementation strains named, respectively, BMC2071, BMC2072 and BMC2073 (Table 1).
Western blot analyses
A 0.2 ml aliquot of an overnight FTG culture of the wild-type, null mutants or complementing strains was inoculated into 10 ml of TH medium. To perform ETX and sialidase Western blots, supernatants of 8 h or overnight TH cultures were used. Samples were collected and each supernatant (or the sonicated whole culture) was mixed with SDS loading buffer and boiled for 5 min. Those mixtures were electrophoresed on a 12% polyacrylamide gel containing SDS for analyzing ETX or an 8% polyacrylamide gel containing SDS for analyzing sialidase proteins. The gels were then subjected to Western blotting using appropriate antibodies, as previously described [31].
Sialidase enzyme activity assay
To assay sialidase enzyme activity, a previously described protocol was used [20] Briefly, 20 µl of TH culture supernatant was added to 60 µl of 100 mM sodium acetate buffer (pH 7.2) in a microtiter tray. A 20 µl of aliquot 4 mM 5-bromo-4-chloro-3-indolyl-α-D-N-acetylneuraminic acid (Sigma) was then added, and the tray was incubated at 37°C for 30 min. The absorbance at 595 nm was then determined using a microplate reader (Bio-Rad).
Growth rate measurement for wild type, mutant and complementing strains
A 0.1 ml aliquot of an FTG overnight culture of the wild-type strain, null mutants or complementing strains was transferred to TH medium, which was grown at 37°C overnight. A 0.2 ml aliquot of each TH overnight culture was then inoculated into 10 ml of pre-warmed TH medium and the OD600 of those cultures was measured at 37°C over time to determine vegetative growth, as described previously [32].
Cell culture
Madin-Darby Canine Kidney (MDCK) epithelial cells were cultured in a 1∶1 (v/v) mix of Dulbecco’s Modified Eagle’s Medium (DMEM, Sigma) and Nutrient Mixture F12 HAM (Sigma), supplemented with 3% fetal bovine serum (Mediatech), 100 µg/ml penicillin/streptomycin (Sigma) and 1% glutamine (Sigma). Caco-2 cells were maintained in Eagle minimal essential medium (Sigma) supplemented with 10% fetal bovine serum (Mediatech), 1% nonessential amino acids (Sigma), and 100 µg/ml of penicillin/streptomycin. Vero cells were cultured in M199 medium (Sigma) supplemented with 5% fetal bovine serum and 100 µg/ml of penicillin/streptomycin. HT-29 cells were cultured in RPM1-164 medium (Sigma) supplemented with 10% fetal bovine serum and 100 µg/ml of penicillin/streptomycin. NT6 fibroblast cells were culture in Alpha-MEM (Modified Eagle Medium) supplemented with 7.5% fetal bovine serum, 2 mM L-glutamine, 1× Non Essential Amino Acids, 1 mM sodium pyruvate, and 100 µg/ml of penicillin/streptomycin. All cells were grown at 37°C with 5% atmospheric CO2.
Cytotoxicity assays
Prior to use in ETX cytotoxicity assays, purified ETX prototoxin was activated by trypsin. For this purpose, 50 µl aliquots of labeled or unlabeled prototoxin (2 mg/ml) were incubated with 12.5 µg of trypsin (Sigma)/µg prototoxin for 1 h at 37°C. After that incubation, trypsin inhibitor (Sigma) (1∶1 v/v) was added to remove trypsin activity. The same trypsin/trypsin inhibitor mix (no ETX) was used as a negative control for cytotoxicity assays. Confluent monolayer MDCK cells were incubated in the presence or absence of 5 µg/ml of activated ETX; in some experiments, that same amount of activated ETX was added to a 10 × concentrated TH supernatant culture for 1 hr at 37°C. After this incubation, cytotoxicity was measured using the LDH Cytotoxicity Detection Kit (Roche).
Trypsin or chymotrypsin treatment of NanI and culture supernatants
An aliquot of 0.005 U/ml NanI (Roche) or 8 h TH culture supernatants of BMC206, BMC207, BMC2071, BMC2072 or BMC2073 were incubated with 12.5 µg of trypsin (Sigma) or chymotrypsin (Sigma) for 1 h at 37°C. After that incubation with trypsin, trypsin inhibitor (Sigma) (1∶1 v/v) was added to remove trypsin activity from those samples. Sialidase activities were detected by the method described before [20].
ETX binding/complex formation in MDCK cells
Confluent monolayers of MDCK cells grown in 6 well cluster plates (Corning) were or were not pretreated with 0.005 U/ml or 0.001 U/ml of C. perfringens sialidase (Roche) for 30, 60, or 90 min. After this treatment, 5 µg/ml of AF488-ETX was added and the cells were then further incubated for 60 min at 37°C. Following this toxin challenge, the cells were harvested and washed twice with PBS buffer and the pellets were then resuspended in 50 µl of PBS. After treatment of those asample with 1 µl of Benzonase nuclease (Novagen) for 5 min at room temperature, 12 µl of 5×SDS loading buffer was added. These samples were then electrophoresed on a SDS-containing 8% polyacrylamide gel, in the dark. The resultant gel was imaged using a Typhoon 9400 variable mode imager (Amersham Biosciences), with fluorescence emission set to detect the Alexa Fluor 488 label using the green laser with wavelength 532 nm. For detection of the molecular weight markers, the red laser was used with a wavelength of 633 nm. Complex formation was then quantified using Imagequant version 5.2 (Molecular Dynamics).
To evaluate ETX binding, confluent monolayers of MDCK cells grown in 24 well cluster plates (Corning) were pretreated with a 0.005 U/ml or 0.001 U/ml of NanI sialidase at 37°C, and then challenged with 5 µg/ml of AF488-pETX suspended in 250 µl of buffer or culture supernatants (from BMC206, BMC207, BMC2071, BMC2072, BMC2073) that had been concentrated 10× using an Ultrafiltration centrifuge tube (Millipore). After 60 min at 37°C, the cells were harvested and washed twice in PBS. The washed pellets were then resuspended in 50 µl of RIPA buffer (Invitrogen) and added to 96-well plates, where fluorescence was quantified using a Multi-mode Microplate Reader (SynergyTM4, BioTek).
Analysis of sialidase production after C. perfringens strains contact Caco-2 cells, HT29 cells, NT6, Vero cells and MDCK cells
A 0.1 ml aliquot of an FTG overnight culture of BMC206, BMC207, BMC2071 or BMC2072 was transferred to TH medium, which was grown at 37°C overnight. A 0.1 ml aliquot of each TH overnight culture was then inoculated into fresh 10 ml TH medium. These overnight cultures were centrifuged and the pelleted cells were washed three times with HBSS buffer and then resuspended in HBSS buffer at 107/ml. Confluent monolayers of Caco-2 cells, MDCK cells, Vero cells, HT-29 cells, and NT-6 cells grown in 6 well cluster plates were washed three times with HBSS and then challenged with a 1 ml aliquot of the washed bacterial cell suspension. Following anaerobic incubation at 37°C, for 2, 4, or 6 h, the supernatants were removed, centrifuged and 60 µl supernatant aliquot were used to measure sialidase activity by the method described above, except the sample was incubated at room temperature overnight instead of 37°C for 30 min before the absorbance was read in order to increase assay sensitive. Control washed bacterial cell suspensions were treated similarly except for the absence of cells.
RNA isolation and qRT-PCR
As described previously [25], total RNA was extracted from 1 ml of a 2, 4, or 6 h culture of BMC206 that had or had not been in contact with Caco2 cells. All RNA samples were treated with DNAaseI (Promega). The purified RNA was quantified by absorbance at 260 nm and stored in a -80°C freezer.
qRT-PCR reactions were performed on the purified RNA samples using the iScriptTM one-step RT-PCR kit with SYBR green (Bio-Rad). Briefly, 100 ng of each RNA sample was reverse-transcribed to cDNA at 50°C for 10 min. That cDNA was then used as template for PCR reactions with primers 16sF and 16sR (Table 2) targeting the 16sRNA as a housekeeping gene; primers nanIKOF and nanIKOR targeting the nanI gene, primers nanJKOF and nanJKOR targeting the nanJ gene, or primers nanHKOF and nanHKOR targeting the nanH gene (Table 2). All reactions were performed at AB Appled Biosystems Step One plus Real-Time PCR system.
Attachment assays
A 1.5 ml aliquot of a TH overnight culture of BMC206, BMC207, BMC2071, BMC2072 or BMC2073 was centrifuged and the bacterial pellet was then washed three times with HBSS buffer. After resuspension of the washed pellet in 1.5 ml of HBSS buffer, these suspensions were serially diluted from 10-2 to 10-7 with sterile water and aliquots were spread onto BHI agar plates. Following an overnight anaerobic incubation at 37°C, the colonies arising on these plates were counted to determine the number of CFU added to the mammalian cell cultures. Monolayers of Caco-2, HT-29, Vero, MDCK or NT6 fibroblasts were then incubated under anaerobic conditions with a 100 µl aliquot of these washed bacteria for 2 h at 37°C. After this incubation, the monolayers were washed with HBSS three times and host cell-associated bacteria were retrieved by lysing the monolayers in distilled water. Aliquots of these suspensions were plated onto BHI agar plates. After overnight anaerobic incubation at 37°C, the colonies arising on the plates were used to calculate the number of adherent CFU.
Immunofluorescence microscopy
Caco-2 cells were grown to confluency in an 8-well chamber slide (Fisher). A 1 µl aliquot of a BMC206, BMC207 and BMC2072 overnight TH culture was added to each chamber of the slide, and the slide was then incubated at 37°C for 2 h. Cells were washed three times with HBSS buffer. The slides were fixed in 4% formaldehyde in HBSS for 30 min at room temperature. The fixed cells were incubated with a 1∶500 dilution of anti-C. perfringens rabbit polyclonal antibody (Genway) in HBSS with 10% BSA for 2 h at room temperature. Those cells were then washed three times with HBSS buffer and incubated with 1∶500 Alexa Fluor-488 goat anti-rabbit IgG (Invitrogen) in HBSS with 10% BSA for 1 h at room temperature. After 3 more washes with HBSS buffer, the cells were incubated with 1∶200 VybrantTM Cell-labeling solutions (Molecular Probes) for 30 min at room temperature. Following a final three washes with HBSS buffer, the chambers were removed, and a coverslip was mounted with Fluoro-Gel (Electron Microscopy Sciences). Imaging was performed on a Zeiss Axioskop 40 immunofluorescence microscope.
Zdroje
1. McClaneBA 2007 Clostridium perfringens. DoyleMPBeuchatLR Food Microbiology. 3rd edition Washington, D.C. ASM press 423 444
2. HathewayC 1990 Toxigenic clostridia. Clin Microb Rev 3 66 76
3. McDonelJL 1986 Toxins of Clostridium perfringens types A, B, C, D, and E. DornerFDrewsH Pharmacology of Bacterial Toxins Oxford Pergamon Press 477 517
4. PetitLGilbertMPopoffMR 1999 Clostridium perfringens: toxinotype and genotype. Trends Microbiol 7 104 110
5. SayeedSFernandez-MiyakawaMEFisherDJAdamsVPoonR 2005 Epsilon-toxin is required for most Clostridium perfringens type D vegetative culture supernatants to cause lethality in the mouse intravenous injection model. Infect Immun 73 7413 7421
6. Fernandez-MiyakawaMEFisherDJPoonRSayeedSAdamsV 2007 Both epsilon-toxin and beta-toxin are important for the lethal properties of Clostridium perfringens type B isolates in the mouse intravenous injection model. Infect Immun 75 1443 1452
7. RoodJI 1998 Virulence genes of Clostridium perfringens. Ann Review Microbiol 52 333 360
8. UzalFASongerG 2008 Diagnosis of Clostridium perfringens intestinal infections in sheep and goat. J Vet Diagn Invest 20 253 265
9. PopoffMR 2011 Epsilon toxin: a fascinating pore-forming toxin. FEBS J (in press) doi:10.1111/j.1742-4658.2011.08145.x
10. ColeARGibertMPopoffMMossDSTitballRW 2004 Clostridium perfringens epsilon-toxin shows structural similarity to the pore-forming toxin aerolysin. Nat Struct Mol Biol 11 797 798
11. Bokori-BrownMSavvaGCFernandes da CostaSPNaylorCEBasakAK 2011 Molecular basis of toxicity of Clostridium perfringens epsilon toxin. FEBS J (in press) doi:10.1111/j.1742-4658.2011.08140.x
12. MiyataSMinamiJTamiaEMatsushitaPShimamotoS 2002 Clostridium perfringens epsilon-toxin forms a hepatmeric pore within the detergent-insoluble microdomains of Madin-Darby canine kidney cells and rat synaptosomes. J Biol Chem 277 39463 39468
13. RobertsonSLLiJUzalFAMcClaneBA 2011 Evidence for a prepore stage in the action of Clostridium perfringens epsilon toxin. PLoS One 6 e22053
14. MyersGSRaskoDACheungJKRavelJSeshadriR 2006 Skewed genomic variability in strains of the toxigenic bacterial pathogen, Clostridium perfringens. Genome Res 16 1031 1040
15. LyristisMBryantAESloanJAwadMMNisbetIT 1994 Identification and molecular analysis of a locus that regulates extracellular toxin production in Clostridium perfringens. Molec Microbiol 12 761 777
16. ShimizuTOhtaniKHirakawaHOhshimaKYamashitaA 2002 Complete genome sequence of Clostridium perfringens, an anaerobic flesh-eater. Proc Natl Acad Sci U S A 99 996 1001
17. BorastonABFicko-BleanEHealeyM 2007 Carbohydrate recognition by a large sialidase toxin from Clostridium perfringens. Biochemistry 46 11352 11360
18. GalenJEKetleyJMFasanoARichardsonSHWassermanSS 1992 Role of Vibrio cholerae neuraminidase in the function of cholera toxin. Infect Immun 60 406 415
19. KingSJ 2010 Pneumococcal modification of host sugars: a major contributor to colonization of the human airway. Mol Oral Microbiol 25 15 24
20. ChiarezzaMLyrasDPidotSJFlore-DiazMAwadMM 2009 The NanI and NanJ sialidases of Clostridium perfringens are not essential for virulence. Infect Immun 77 4421 4428
21. ShimanotoSTamaiEMatsushitaOMinamiJOkabeA 2005 Changes in ganglioside content affect the binding of Clostridium perfringens epsilon-toxin to detergent-resistant membranes of Madin-Darby canine kidney cells. Microbiol Immunol 49 245 253
22. NagahamaMSakuraiJ 1992 High-affinity binding of Clostridium perfingens epsilon-toxin to rat brain. Infect Immun 60 1237 1240
23. PetitLGibertMGilletDLaurent-WinterCBoquetP 1997 Clostridium perfringens epsilon toxin acts on MDCK cells by forming a large membrane complex. J Bacteriol 179 6480 6487
24. ChenYMcClaneBAFisherDJRoodJIGuptaP 2005 Construction of an alpha toxin gene knockout mutant of Clostridium perfringens type A by use of a mobile group II intron. Appl Environ Microbiol 71 7542 7547
25. SayeedSUzalFAFisherDJSaputoJVidalJE 2008 Beta toxin is essential for the intestinal virulence of Clostridium perfringens type C disease isolate CN3685 in a rabbit ileal loop model. Molec Microbiol 67 15 30
26. HonmaKMishimaESharmaA 2010 Role of Tannerella forsythia NanH sialidase in epithelial cell attachment. Infect Immun: 79 393 401
27. JostBHBillingtonSJTrinhHTSongerJG 2006 Association of genes encoding beta2 toxin and a collagen binding protein in Clostridium perfringens isolates of porcine origin. Vet Microbiol 115 173 182
28. RodgersKArvisonCGMelvilleSB 2011 Expression of a Clostridium perfringens type IV pilin by Neisseria gonorrhoeae mediates adherence to muscle cells. Infect Immun 79 3096 3105
29. HunterSECClarkeINKelleyDCTitballRW 1992 Cloning and nucleotide sequencing of the Clostridium perfringens epsilon-toxin gene and its expression in Escherichia coli. Infect Immun 60 102 110
30. Fernandez-MiyakawaMEUzalFA 2003 The early effects of Clostridium perfringens type D epsilon toxin in ligated intestinal loops of goats and sheep. Vet Res Commun 27 231 241
31. Kokai-KunJFSongerJGCzeczulinJRChenFMcClaneBA 1994 Comparison of Western immunoblots and gene detection assays for identification of potentially enterotoxigenic isolates of Clostridium perfringens. J Clin Microbiol 32 2533 2539
32. LiJMcClaneBA 2006 Further comparison of temperature effects on growth and survival of Clostridium perfringens type A isolates carrying a chromosomal or plasmid-borne enterotoxin gene. Appl Environ Microbiol 72 4561 4568
33. MiyamotoKFisherDJLiJAkimotoSMcClaneBA 2006 Complete sequencing and diversity analysis of the enterotoxin-encoding plasmids in Clostridium perfringens type A non-food-borne human gastrointestinal disease isolates. J Bacteriol 188 1585 1598
34. SayeedSLiJMcClaneBA 2010 Characterization of virulence plasmid diversity among Clostridium perfringens type B isolates. Infect Immun 78 495 504
35. GurjarALiJMcClaneBA 2010 Characterization of toxin plasmids in Clostridium perfringens type C isolates. Infect Immun 78 4860 4869
36. SayeedSLiJMClaneBA 2007 Virulence plasmid diversity in Clostridium perfringens type D isolates. Infect Immun 75 2391 2398
37. BannamTLRoodJI 1993 Clostridium perfringens-Escherichia coli shuttle vectors that carry single antibiotic resistance determinants. Plasmid 29 223 235
Štítky
Hygiena a epidemiologie Infekční lékařství Laboratoř
Článek Genesis of Mammalian Prions: From Non-infectious Amyloid Fibrils to a Transmissible Prion DiseaseČlánek Role of Permissive Neuraminidase Mutations in Influenza A/Brisbane/59/2007-like (H1N1) VirusesČlánek Allelic Variation on Murine Chromosome 11 Modifies Host Inflammatory Responses and Resistance toČlánek Multifaceted Regulation of Translational Readthrough by RNA Replication Elements in a TombusvirusČlánek Latent KSHV Infection of Endothelial Cells Induces Integrin Beta3 to Activate Angiogenic PhenotypesČlánek Controlling Viral Immuno-Inflammatory Lesions by Modulating Aryl Hydrocarbon Receptor Signaling
Článek vyšel v časopisePLOS Pathogens
Nejčtenější tento týden
2011 Číslo 12- Farmakovigilanční studie perorálních antivirotik indikovaných v léčbě COVID-19
- Jak souvisí postcovidový syndrom s poškozením mozku?
- Měli bychom postcovidový syndrom léčit antidepresivy?
- 10 bodů k očkování proti COVID-19: stanovisko České společnosti alergologie a klinické imunologie ČLS JEP
-
Všechny články tohoto čísla
- Inhibition of Apoptosis and NF-κB Activation by Vaccinia Protein N1 Occur via Distinct Binding Surfaces and Make Different Contributions to Virulence
- Genesis of Mammalian Prions: From Non-infectious Amyloid Fibrils to a Transmissible Prion Disease
- Kaposi's Sarcoma Herpesvirus microRNAs Target Caspase 3 and Regulate Apoptosis
- Nutritional Immunology: A Multi-Dimensional Approach
- Role of Permissive Neuraminidase Mutations in Influenza A/Brisbane/59/2007-like (H1N1) Viruses
- Vaccinomics and Personalized Vaccinology: Is Science Leading Us Toward a New Path of Directed Vaccine Development and Discovery?
- Symbiont Infections Induce Strong Cytoplasmic Incompatibility in the Tsetse Fly
- Allelic Variation on Murine Chromosome 11 Modifies Host Inflammatory Responses and Resistance to
- Computational and Biochemical Analysis of the Effector AvrBs2 and Its Role in the Modulation of Type Three Effector Delivery
- Granzyme B Inhibits Vaccinia Virus Production through Proteolytic Cleavage of Eukaryotic Initiation Factor 4 Gamma 3
- Association of Activating KIR Copy Number Variation of NK Cells with Containment of SIV Replication in Rhesus Monkeys
- Fungal Virulence and Development Is Regulated by Alternative Pre-mRNA 3′End Processing in
- versus the Host: Remodeling of the Bacterial Outer Membrane Is Required for Survival in the Gastric Mucosa
- Follicular Dendritic Cell-Specific Prion Protein (PrP) Expression Alone Is Sufficient to Sustain Prion Infection in the Spleen
- Autophagy Protein Atg3 is Essential for Maintaining Mitochondrial Integrity and for Normal Intracellular Development of Tachyzoites
- Longevity and Composition of Cellular Immune Responses Following Experimental Malaria Infection in Humans
- Sequential Adaptive Mutations Enhance Efficient Vector Switching by Chikungunya Virus and Its Epidemic Emergence
- Acquisition of Pneumococci Specific Effector and Regulatory Cd4 T Cells Localising within Human Upper Respiratory-Tract Mucosal Lymphoid Tissue
- The Meaning of Death: Evolution and Ecology of Apoptosis in Protozoan Parasites
- Deficiency of a Niemann-Pick, Type C1-related Protein in Is Associated with Multiple Lipidoses and Increased Pathogenicity
- Feeding Cells Induced by Phytoparasitic Nematodes Require γ-Tubulin Ring Complex for Microtubule Reorganization
- Eight RGS and RGS-like Proteins Orchestrate Growth, Differentiation, and Pathogenicity of
- Prion Uptake in the Gut: Identification of the First Uptake and Replication Sites
- Nef Decreases HIV-1 Sensitivity to Neutralizing Antibodies that Target the Membrane-proximal External Region of TMgp41
- Multifaceted Regulation of Translational Readthrough by RNA Replication Elements in a Tombusvirus
- A Temporal Role Of Type I Interferon Signaling in CD8 T Cell Maturation during Acute West Nile Virus Infection
- The Membrane Fusion Step of Vaccinia Virus Entry Is Cooperatively Mediated by Multiple Viral Proteins and Host Cell Components
- HIV-1 Capsid-Cyclophilin Interactions Determine Nuclear Import Pathway, Integration Targeting and Replication Efficiency
- Neonatal CD8 T-cell Hierarchy Is Distinct from Adults and Is Influenced by Intrinsic T cell Properties in Respiratory Syncytial Virus Infected Mice
- Two Novel Transcriptional Regulators Are Essential for Infection-related Morphogenesis and Pathogenicity of the Rice Blast Fungus
- Five Questions about Non-Mevalonate Isoprenoid Biosynthesis
- The Human Cytomegalovirus UL11 Protein Interacts with the Receptor Tyrosine Phosphatase CD45, Resulting in Functional Paralysis of T Cells
- Wall Teichoic Acids of Limit Recognition by the Drosophila Peptidoglycan Recognition Protein-SA to Promote Pathogenicity
- A Novel Role for the NLRC4 Inflammasome in Mucosal Defenses against the Fungal Pathogen
- Inflammasome-dependent Pyroptosis and IL-18 Protect against Lung Infection while IL-1β Is Deleterious
- CNS Recruitment of CD8+ T Lymphocytes Specific for a Peripheral Virus Infection Triggers Neuropathogenesis during Polymicrobial Challenge
- Latent KSHV Infection of Endothelial Cells Induces Integrin Beta3 to Activate Angiogenic Phenotypes
- A Receptor-based Switch that Regulates Anthrax Toxin Pore Formation
- Targeting of Heparin-Binding Hemagglutinin to Mitochondria in Macrophages
- Chikungunya Virus Neutralization Antigens and Direct Cell-to-Cell Transmission Are Revealed by Human Antibody-Escape Mutants
- Ce-Duox1/BLI-3 Generated Reactive Oxygen Species Trigger Protective SKN-1 Activity via p38 MAPK Signaling during Infection in
- Structural Elucidation and Functional Characterization of the Effector Protein ATR13
- Controlling Viral Immuno-Inflammatory Lesions by Modulating Aryl Hydrocarbon Receptor Signaling
- SAMHD1-Deficient CD14+ Cells from Individuals with Aicardi-Goutières Syndrome Are Highly Susceptible to HIV-1 Infection
- Acid Stability of the Hemagglutinin Protein Regulates H5N1 Influenza Virus Pathogenicity
- Cryo Electron Tomography of Herpes Simplex Virus during Axonal Transport and Secondary Envelopment in Primary Neurons
- A Novel Human Cytomegalovirus Locus Modulates Cell Type-Specific Outcomes of Infection
- Juxtamembrane Shedding of AMA1 Is Sequence Independent and Essential, and Helps Evade Invasion-Inhibitory Antibodies
- Pathogenesis and Host Response in Syrian Hamsters following Intranasal Infection with Andes Virus
- IRGM Is a Common Target of RNA Viruses that Subvert the Autophagy Network
- Epstein-Barr Virus Evades CD4 T Cell Responses in Lytic Cycle through BZLF1-mediated Downregulation of CD74 and the Cooperation of vBcl-2
- Quantitative Multicolor Super-Resolution Microscopy Reveals Tetherin HIV-1 Interaction
- Late Repression of NF-κB Activity by Invasive but Not Non-Invasive Meningococcal Isolates Is Required to Display Apoptosis of Epithelial Cells
- Polar Flagellar Biosynthesis and a Regulator of Flagellar Number Influence Spatial Parameters of Cell Division in
- Epstein-Barr Virus Nuclear Antigen 3C Stabilizes Gemin3 to Block p53-mediated Apoptosis
- The Enteropathogenic (EPEC) Tir Effector Inhibits NF-κB Activity by Targeting TNFα Receptor-Associated Factors
- Toward an Integrated Model of Capsule Regulation in
- A Systematic Screen to Discover and Analyze Apicoplast Proteins Identifies a Conserved and Essential Protein Import Factor
- A Host Small GTP-binding Protein ARL8 Plays Crucial Roles in Tobamovirus RNA Replication
- Comparative Pathobiology of Fungal Pathogens of Plants and Animals
- Synergistic Roles of Eukaryotic Translation Elongation Factors 1Bγ and 1A in Stimulation of Tombusvirus Minus-Strand Synthesis
- Engineered Immunity to Infection
- Inflammatory Monocytes and Neutrophils Are Licensed to Kill during Memory Responses
- Sialidases Affect the Host Cell Adherence and Epsilon Toxin-Induced Cytotoxicity of Type D Strain CN3718
- Eurasian-Origin Gene Segments Contribute to the Transmissibility, Aerosol Release, and Morphology of the 2009 Pandemic H1N1 Influenza Virus
- SARS Coronavirus nsp1 Protein Induces Template-Dependent Endonucleolytic Cleavage of mRNAs: Viral mRNAs Are Resistant to nsp1-Induced RNA Cleavage
- Identification and Characterization of a Novel Non-Structural Protein of Bluetongue Virus
- Functional Analysis of the Kinome of the Wheat Scab Fungus
- Norovirus Regulation of the Innate Immune Response and Apoptosis Occurs via the Product of the Alternative Open Reading Frame 4
- PLOS Pathogens
- Archiv čísel
- Aktuální číslo
- Informace o časopisu
Nejčtenější v tomto čísle- Controlling Viral Immuno-Inflammatory Lesions by Modulating Aryl Hydrocarbon Receptor Signaling
- Fungal Virulence and Development Is Regulated by Alternative Pre-mRNA 3′End Processing in
- Cryo Electron Tomography of Herpes Simplex Virus during Axonal Transport and Secondary Envelopment in Primary Neurons
- Epstein-Barr Virus Nuclear Antigen 3C Stabilizes Gemin3 to Block p53-mediated Apoptosis
Přihlášení#ADS_BOTTOM_SCRIPTS#Zapomenuté hesloZadejte e-mailovou adresu, se kterou jste vytvářel(a) účet, budou Vám na ni zaslány informace k nastavení nového hesla.
- Vzdělávání



